Advanced Cabazitaxel Synthesis: Technical Breakthroughs for Commercial Scale-up
Advanced Cabazitaxel Synthesis: Technical Breakthroughs for Commercial Scale-up
The pharmaceutical landscape for oncology treatments continues to evolve, with Cabazitaxel standing out as a critical second-line therapy for metastatic prostate cancer. The patent CN102659721A introduces a transformative synthetic methodology that addresses long-standing challenges in the manufacturing of this complex taxane derivative. By utilizing 10-deacetylbaccatin III as the foundational starting material, this novel approach leverages a sophisticated sequence of selective hydroxyl protections and deprotections to achieve high purity and yield. This technical advancement is particularly significant for global supply chains seeking reliable Active Pharmaceutical Ingredients (APIs) suppliers who can deliver consistent quality at scale. The structural complexity of Cabazitaxel, as illustrated below, demands precise chemical manipulation to ensure therapeutic efficacy and safety.
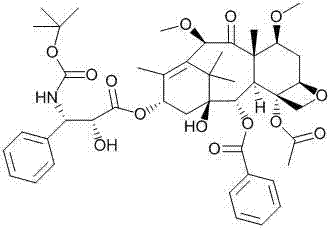
Traditional synthetic routes have often been plagued by inefficiencies that hinder large-scale production. The legacy methods, such as those disclosed in US5889043, typically involve introducing the expensive oxazolidine carboxylic acid side chain at an early stage or utilizing harsh conditions for methylation that compromise yield. These conventional approaches frequently suffer from low reaction efficiency and stringent condition requirements, making them less suitable for industrial suitability. Furthermore, the early introduction of the chiral side chain can lead to significant material wastage if subsequent steps on the core scaffold fail or produce impurities. This creates a bottleneck for procurement managers looking for cost reduction in pharmaceutical intermediates manufacturing, as the loss of high-value intermediates directly impacts the bottom line.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The historical context of taxane synthesis reveals a persistent struggle with regioselectivity and reaction harshness. In earlier methodologies, the methylation of the 7 and 10 hydroxyl groups often required aggressive reagents or conditions that could degrade the sensitive taxane ring system. Additionally, protecting group strategies were sometimes suboptimal, leading to mixtures of products that required extensive and costly purification processes. The use of triethylsilyl (TES) or trichloroethoxycarbonyl (Troc) groups in prior art, while effective, often resulted in lower yields during the protection and deprotection cycles. These inefficiencies accumulate over a multi-step synthesis, drastically reducing the overall throughput and increasing the environmental footprint due to solvent and reagent consumption. For supply chain heads, these factors translate into longer lead times and higher vulnerability to raw material shortages.
The Novel Approach
The methodology outlined in CN102659721A represents a paradigm shift by optimizing the order of operations to maximize efficiency. The core innovation lies in the sequential protection strategy: first selectively protecting the more reactive 7 and 10 hydroxyls, followed by the protection of the 13 hydroxyl. Crucially, the 7 and 10 protecting groups are then removed to allow for methylation under mild alkaline conditions before the final coupling with the side chain. This "late-stage coupling" strategy ensures that the expensive side chain is only introduced when the core scaffold is fully prepared and verified, minimizing financial risk. The reaction conditions are notably milder, utilizing standard reagents like sodium hydride and methyl iodide in controlled environments, which enhances operational safety and simplifies equipment requirements for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Selective Hydroxyl Protection and Methylation
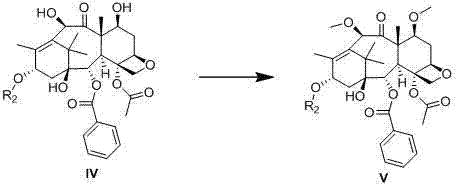
The success of this synthetic route hinges on the precise exploitation of the differing reactivities of the hydroxyl groups on the baccatin III core. The 7 and 10 hydroxyls exhibit higher nucleophilicity compared to the 13 hydroxyl, allowing for their selective protection using agents such as Troc, MEM, or silyl chlorides. This initial step creates a masked intermediate where the 13 position remains available for subsequent modification. Once the 13 hydroxyl is protected, the 7 and 10 groups are deprotected, exposing them for methylation. This specific sequence prevents the formation of unwanted byproducts that would arise from simultaneous protection attempts. The methylation step itself is conducted under nitrogen or argon shielding in organic solvents like THF, using sodium hydride as a base to generate the alkoxide, which then reacts with methyl iodide. This controlled environment ensures high conversion rates and minimizes side reactions.
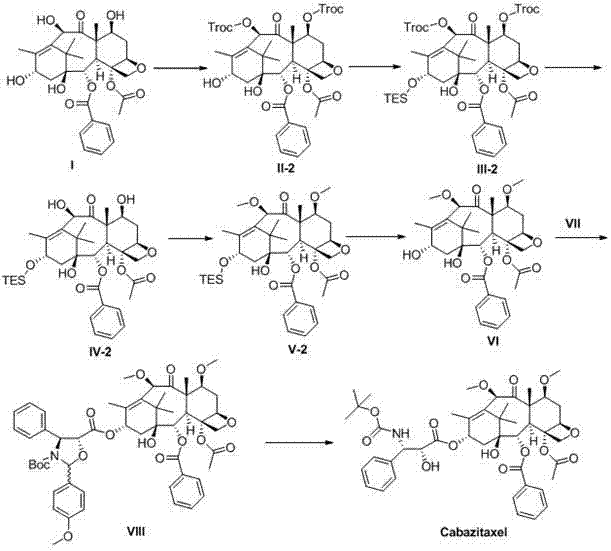
Following methylation, the 13 protecting group is removed to reveal the hydroxyl necessary for esterification with the side chain. The condensation reaction utilizes coupling agents like DCC in the presence of catalysts such as DMAP, facilitating the formation of the ester bond with high stereochemical fidelity. The final deprotection step removes any remaining temporary groups, yielding the target Cabazitaxel molecule. This mechanistic pathway is robust and reproducible, offering a clear advantage over routes that rely on Pummerer rearrangements or Raney nickel reductions, which can be unpredictable on a large scale. The ability to control the stereochemistry and purity at each stage is paramount for meeting the rigorous quality standards required by regulatory bodies for oncology drugs.
How to Synthesize Cabazitaxel Efficiently
The synthesis of Cabazitaxel via this patented method involves a series of carefully orchestrated chemical transformations designed to maximize yield and purity while minimizing waste. The process begins with the selective protection of the 10-deacetylbaccatin III core, followed by a strategic sequence of deprotection and methylation steps that prepare the molecule for the final coupling event. Each stage requires precise control of temperature, stoichiometry, and reaction time to ensure optimal outcomes. The detailed标准化 synthesis steps are provided in the guide below, offering a roadmap for technical teams to implement this efficient route in their own facilities.
- Perform selective protection on the 7 and 10 hydroxyl groups of 10-deacetylbaccatin III using agents like Troc or MEM to form the protected intermediate.
- Protect the 13-hydroxyl group, then remove the 7 and 10 protecting groups to expose them for subsequent methylation.
- Methylate the 7 and 10 positions, deprotect the 13-position, and condense with the oxazolidine carboxylic acid side chain to finalize the structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic method offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the significant optimization of resource utilization. By deferring the introduction of the costly oxazolidine carboxylic acid side chain until the final stages of the synthesis, manufacturers can avoid the financial loss associated with processing defective cores. This strategic timing acts as a risk mitigation tool, ensuring that high-value materials are not wasted on intermediates that may not meet specification. Furthermore, the use of readily available reagents and mild reaction conditions reduces the dependency on specialized or hazardous chemicals, simplifying the procurement process and enhancing workplace safety.
- Cost Reduction in Manufacturing: The streamlined protection and deprotection sequence eliminates the need for expensive and harsh reagents often required in traditional routes. By avoiding the early use of the chiral side chain, the process inherently reduces the cost of goods sold (COGS) by preventing the loss of this high-value component during earlier, lower-yield steps. The improved yields at each stage cumulatively result in a much higher overall output from the same amount of starting material, driving down the unit cost of production. Additionally, the simplified purification requirements reduce solvent consumption and waste disposal costs, contributing to a leaner and more economical manufacturing operation.
- Enhanced Supply Chain Reliability: The reliance on common chemical reagents such as methyl iodide, sodium hydride, and standard protecting group agents ensures a stable supply chain不受 limited by niche material availability. This robustness is critical for maintaining continuous production schedules and meeting delivery commitments to downstream partners. The method's adaptability to various protecting groups (e.g., Troc, MEM, Silyl) provides flexibility, allowing manufacturers to switch reagents based on market availability without compromising the integrity of the synthesis. This flexibility mitigates the risk of supply disruptions and supports a more resilient global distribution network for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions and straightforward workup procedures make this process highly amenable to scale-up from laboratory to industrial production. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, reducing the burden of compliance and permitting. The ability to run reactions at moderate temperatures and pressures lowers energy consumption, further enhancing the sustainability profile of the manufacturing process. This scalability ensures that the method can support growing market demand for Cabazitaxel without the need for disproportionate capital investment in specialized infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthetic methodology. The answers are derived directly from the patent specifications and are intended to clarify the operational advantages and chemical principles underlying the process. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: What is the starting material for this Cabazitaxel synthesis method?
A: The synthesis begins with 10-deacetylbaccatin III (10-DAB III), a naturally derived taxane precursor extracted from Taxus cuspidata, which serves as the core scaffold for the reaction sequence.
Q: How does this method improve upon previous synthetic routes like US5889043?
A: Unlike prior art that introduces the expensive side chain early or uses harsh methylation conditions, this method employs a strategic protection sequence that allows for mild methylation and late-stage coupling, significantly reducing material waste and improving overall yield.
Q: Why is the order of hydroxyl protection critical in this process?
A: The 7 and 10 hydroxyl groups are more reactive than the 13 hydroxyl. By protecting 7 and 10 first, then 13, and subsequently deprotecting 7 and 10 for methylation, the process achieves high regioselectivity and minimizes byproduct formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cabazitaxel Supplier
The technical potential of this synthetic route is immense, offering a pathway to more affordable and accessible prostate cancer treatments. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such complex chemistries to life. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs ensuring that every batch meets the highest international standards. We understand the critical nature of oncology APIs and are dedicated to providing a supply partner that prioritizes consistency, compliance, and technical excellence.
We invite you to explore how our expertise can optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Let us collaborate to drive efficiency and innovation in the production of life-saving medicines.
