Advanced Synthesis of C-13 and C-14 Modified Taxane Derivatives for Oncology Applications
The pharmaceutical industry continuously seeks next-generation oncology therapeutics that offer enhanced efficacy with reduced systemic toxicity, and patent CN107188895B presents a significant advancement in this domain by detailing the synthesis of novel paclitaxel analogs modified at the C-13 and C-14 positions. This intellectual property describes a sophisticated semi-synthetic methodology that transforms 1-dehydroxybaccatine VI into a series of structurally diverse taxane derivatives, specifically compounds A, B, C, and D, which retain the critical baccatin ring skeleton essential for microtubule stabilization. By strategically introducing different side chains and functional groups at the C-13 and C-14 loci, this technology enriches the library of available medicinal samples, providing crucial data for structure-activity relationship (SAR) studies aimed at optimizing therapeutic indices. The disclosed compounds demonstrate potent in vitro proliferation inhibitory effects against human A549 lung cancer cells, A2780 ovarian cancer cells, and HL-60 promyelocytic leukemia cells, suggesting a viable pathway for developing second-line chemotherapy agents. For research and development teams focusing on antineoplastic drug discovery, this patent offers a robust framework for generating high-purity intermediates that serve as valuable tools for biological screening and preclinical evaluation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for obtaining paclitaxel and its derivatives have historically relied heavily on the extraction from the bark of the Pacific yew tree, a process that is not only ecologically unsustainable but also suffers from extremely low natural abundance and complex isolation procedures. Furthermore, early semi-synthetic approaches often struggled with regioselectivity, particularly when attempting to modify the densely functionalized taxane core without affecting other sensitive hydroxyl groups, leading to complex mixtures that are difficult and costly to purify. The lack of precise control over the C-13 and C-14 positions in earlier synthetic routes frequently resulted in analogs with unpredictable pharmacokinetic profiles or diminished biological activity compared to the parent compound. Additionally, conventional protection and deprotection strategies often utilized harsh reagents or extreme conditions that could degrade the delicate oxetane ring or the ester linkages crucial for bioactivity. These limitations created a significant bottleneck in the supply chain for specialized taxane intermediates, driving up costs and extending lead times for pharmaceutical developers seeking novel candidates.
The Novel Approach
The methodology outlined in patent CN107188895B overcomes these historical challenges through a meticulously designed sequence of selective protection, oxidation, and coupling reactions that ensure high regiochemical fidelity. The process initiates with a gentle deacetylation using hydrazine hydrate, followed by the strategic installation of an isopropylidene group to protect the 9,10-diol system, thereby freeing up the C-13 and C-14 positions for targeted modification. 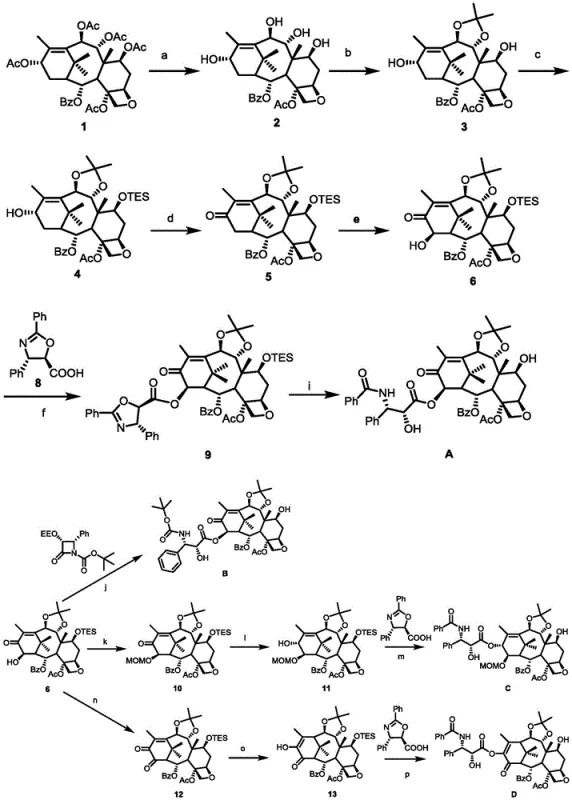 Subsequent steps employ triethylsilyl (TES) protection at C-7 and catalytic oxidation using tetrapropylammonium perruthenate (TPAP) to introduce carbonyl functionality with minimal over-oxidation. The introduction of side chains is achieved via efficient coupling reagents like dicyclohexylcarbodiimide (DCC) and 4-dimethylaminopyridine (DMAP) under mild conditions, preserving the stereochemical integrity of the molecule. This novel approach not only simplifies the purification workflow by reducing byproduct formation but also enables the scalable production of specific analogs like Compound C, which exhibits cytotoxicity comparable to natural paclitaxel, thereby validating the commercial viability of this synthetic route for reliable pharmaceutical intermediate supplier operations.
Subsequent steps employ triethylsilyl (TES) protection at C-7 and catalytic oxidation using tetrapropylammonium perruthenate (TPAP) to introduce carbonyl functionality with minimal over-oxidation. The introduction of side chains is achieved via efficient coupling reagents like dicyclohexylcarbodiimide (DCC) and 4-dimethylaminopyridine (DMAP) under mild conditions, preserving the stereochemical integrity of the molecule. This novel approach not only simplifies the purification workflow by reducing byproduct formation but also enables the scalable production of specific analogs like Compound C, which exhibits cytotoxicity comparable to natural paclitaxel, thereby validating the commercial viability of this synthetic route for reliable pharmaceutical intermediate supplier operations.
Mechanistic Insights into Regioselective Oxidation and Coupling
The core chemical innovation in this synthesis lies in the precise manipulation of the taxane oxidation state and the subsequent nucleophilic attack mechanisms employed during side-chain attachment. The oxidation of the C-13 hydroxyl group to a ketone is facilitated by the TPAP/NMO (N-methylmorpholine N-oxide) system, which operates via a catalytic cycle involving the ruthenium center cycling between +7 and +5 oxidation states to transfer oxygen atoms efficiently without requiring stoichiometric amounts of toxic heavy metal oxidants. This mechanistic pathway is critical for maintaining the stability of the adjacent C-14 position, allowing for subsequent differentiation where C-14 can be selectively hydroxylated or protected depending on the desired final analog. The coupling reaction at the C-13 or C-14 position utilizes an activated ester mechanism where DCC activates the carboxylic acid of the phenylisoserine derivative to form an O-acylisourea intermediate, which is then attacked by the taxane hydroxyl group. The presence of DMAP acts as a nucleophilic catalyst, accelerating the acylation rate and suppressing racemization of the chiral centers on the side chain, which is paramount for retaining the biological potency of the final drug substance. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters such as temperature and solvent polarity to maximize yield and minimize epimerization, ensuring the production of high-purity intermediates suitable for clinical applications.
Impurity control is another vital aspect of this mechanism, particularly regarding the removal of urea byproducts generated from DCC and residual ruthenium species from the oxidation step. The protocol specifies rigorous workup procedures involving aqueous washes and column chromatography to ensure that trace metal contaminants are reduced to levels compliant with ICH Q3D guidelines for elemental impurities. The use of montmorillonite K10 as a solid acid catalyst in the acetonide protection step further exemplifies a green chemistry approach, as it can be easily filtered off, reducing the burden on downstream waste treatment systems. By controlling the stoichiometry of reagents, such as maintaining a specific molar ratio of hydrazine hydrate to substrate during deacetylation, the process minimizes the formation of partially deacetylated intermediates that could complicate downstream purification. This level of mechanistic control translates directly into a cleaner crude product profile, reducing the number of crystallization cycles required and ultimately lowering the cost of goods sold for the final active pharmaceutical ingredient.
How to Synthesize C-13 and C-14 Modified Taxanes Efficiently
The synthesis of these complex taxane derivatives requires strict adherence to the optimized reaction conditions detailed in the patent to ensure reproducibility and high yield across multiple batches. The process begins with the dissolution of 1-dehydroxybaccatine VI in ethanol followed by the controlled addition of hydrazine hydrate, where maintaining the reaction temperature at room temperature for 15 to 18 hours is critical to achieving complete deacetylation without degrading the core structure. Following isolation of the tetra-deacetyl intermediate, the protection sequence involves dissolving the substrate in dichloromethane and adding 2,2-dimethoxypropane with a catalytic amount of Montmorillonite K10, a step that must be monitored via TLC to prevent over-reaction or hydrolysis of the newly formed acetonide. The subsequent oxidation and coupling steps demand anhydrous conditions and inert atmosphere protection to prevent moisture-induced side reactions, highlighting the need for specialized equipment and trained personnel in a GMP-compliant facility. Detailed standardized operating procedures for each transformation, including quenching protocols and solvent exchange methods, are essential for transferring this laboratory-scale chemistry to commercial production environments.
- Selective Deacetylation: React 1-dehydroxybaccatine VI with hydrazine hydrate in ethanol to remove acetyl groups at positions 7, 9, 10, and 13.
- Protection Strategy: Protect the 9,10-diol system using 2,2-dimethoxypropane and Montmorillonite K10, followed by silylation of the C-7 hydroxyl with TESCl.
- Oxidation and Coupling: Oxidize the C-13 position using TPAP/NMO, introduce C-14 functionality, and couple with phenylisoserine derivatives using DCC/DMAP.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic benefits primarily driven by the optimization of raw material utilization and the simplification of processing steps. The reliance on 1-dehydroxybaccatine VI as a starting material leverages an existing supply chain infrastructure, as this precursor is widely available from semi-synthetic paclitaxel production streams, thereby mitigating the risk of raw material shortages that often plague natural product extraction. The elimination of cryogenic conditions for the majority of the synthesis steps significantly reduces energy consumption and capital expenditure on specialized refrigeration equipment, contributing to a lower overall manufacturing footprint. Furthermore, the use of catalytic oxidation systems rather than stoichiometric heavy metal oxidants drastically reduces the volume of hazardous waste generated, simplifying environmental compliance and waste disposal logistics which are major cost drivers in pharmaceutical manufacturing.
- Cost Reduction in Manufacturing: The process achieves cost efficiency through the use of recoverable catalysts and common organic solvents like ethyl acetate and toluene, which are significantly cheaper than specialized fluorinated solvents often used in fine chemical synthesis. By avoiding the use of expensive transition metal complexes in stoichiometric quantities and utilizing solid-supported acids like Montmorillonite K10 that can be filtered and potentially regenerated, the direct material costs are substantially minimized. The high regioselectivity of the protection groups reduces the formation of isomeric impurities, which in turn decreases the loss of valuable intermediate during purification stages, leading to a higher overall mass balance and better yield per kilogram of starting material. These factors combine to create a more economically viable production model that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The synthetic pathway is designed with robustness in mind, utilizing reagents that are commercially available in bulk quantities from multiple global suppliers, thus preventing single-source bottlenecks. The reaction conditions are relatively mild, typically ranging from room temperature to moderate heating, which reduces the risk of thermal runaways and enhances operational safety, ensuring consistent batch-to-batch delivery schedules. The ability to produce multiple analogs (Compounds A, B, C, D) from a common advanced intermediate provides supply chain flexibility, allowing manufacturers to pivot production based on demand for specific derivatives without retooling the entire facility. This adaptability is crucial for maintaining continuity of supply in the face of changing clinical trial requirements or market demands for specific paclitaxel variants.
- Scalability and Environmental Compliance: The methodology is inherently scalable, as demonstrated by the successful execution of reactions on gram scales in the patent examples, with clear pathways for expansion to kilogram and tonne scales using standard stirred-tank reactors. The aqueous workup procedures and the absence of highly toxic reagents facilitate easier handling and containment, aligning with modern green chemistry principles and reducing the regulatory burden associated with hazardous chemical management. The process generates less saline waste compared to traditional acid-base extraction methods, lowering the load on wastewater treatment facilities and supporting corporate sustainability goals. This environmental compatibility not only reduces operational costs but also enhances the brand reputation of the manufacturer as a responsible partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these modified taxane compounds, based on the specific data and claims presented in the patent documentation. These insights are intended to clarify the feasibility of implementing this technology within existing manufacturing frameworks and to highlight the specific advantages it holds over legacy production methods.
Q: What is the primary advantage of modifying the C-13 and C-14 positions on the taxane skeleton?
A: Modifying these positions allows for the retention of the core anticancer ring skeleton while potentially reducing the toxic side effects associated with natural paclitaxel and enabling structure-activity relationship (SAR) studies.
Q: How does this synthesis method improve upon traditional extraction methods?
A: Unlike direct extraction from Taxus plants which is resource-intensive and low-yield, this semi-synthetic approach utilizes 1-dehydroxybaccatine VI to create diverse analogs with higher yields and better control over stereochemistry.
Q: Are the reaction conditions suitable for large-scale manufacturing?
A: Yes, the process utilizes standard organic solvents like dichloromethane and toluene, and many key steps proceed at room temperature or mild heating, facilitating scalability without extreme pressure or cryogenic requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paclitaxel Analog Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure necessary to bring these complex C-13 and C-14 modified paclitaxel compounds from patent to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision regardless of the project phase. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and quality of every batch, guaranteeing that our intermediates meet the exacting standards required for oncology drug development. Our commitment to quality assurance extends to full traceability of raw materials and comprehensive documentation packages that support regulatory filings and audits.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this optimized manufacturing process for your supply chain. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive innovation and efficiency in your pharmaceutical projects.
