Advanced Synthesis of Phosphatidylethanolamine Intermediates for Lipid Nanoparticle Applications
The landscape of lipid nanoparticle (LNP) technology and liposomal drug delivery systems relies heavily on the availability of high-purity synthetic phospholipids. Patent CN115433224A introduces a groundbreaking methodology for synthesizing di-fatty acyl phosphatidylethanolamine intermediates and their amino polyethylene glycol derivatives. This innovation addresses critical bottlenecks in the construction of phosphorus-oxygen bonds, which are fundamental to the structural integrity of these amphiphilic molecules. By utilizing a modified phosphitylating agent represented by Formula VI, the invention enables a mild, high-efficiency three-component condensation reaction. This approach not only enhances reaction selectivity but also simplifies the downstream purification processes, making it an ideal solution for the commercial scale-up of complex pharmaceutical intermediates. The resulting compounds exhibit exceptional purity profiles, essential for meeting the stringent regulatory requirements of injectable therapeutics.
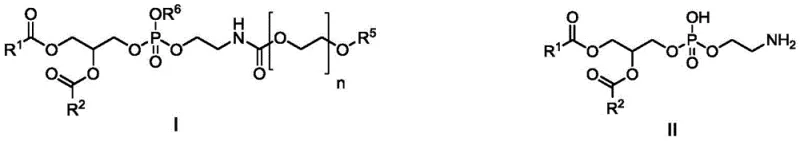
Traditional synthetic routes for phosphatidylethanolamine have long been plagued by inefficiencies and safety concerns. Conventional methods often employ pentavalent phosphonylating reagents, which struggle with low condensation yields when reacting with sterically hindered long-chain di-fatty acylglycerol esters. Alternatively, the direct use of trivalent reagents like phosphorus trichloride (PCl3) presents significant challenges; while reactive, PCl3 is prone to over-reaction, leading to the formation of symmetrical by-products where two alcohol fragments attach to the phosphorus atom, drastically reducing overall yield. Furthermore, existing literature frequently relies on amine protecting groups like Cbz (benzyloxycarbonyl), which necessitate removal via palladium-carbon catalyzed hydrogenation. This step introduces substantial costs related to expensive catalysts, specialized high-pressure equipment, and rigorous safety protocols for handling hydrogen gas. These limitations create a pressing need for a more robust and economically viable synthetic strategy.
The novel approach detailed in the patent circumvents these historical obstacles through the strategic design of the phosphitylating reagent and the selection of compatible protecting groups. By employing a compound of Formula VI, where the phosphorus center is modified with specific aryl and leaving groups, the reaction achieves superior chemoselectivity. This modification prevents the unwanted double-condensation side reactions common with simple PCl3. Additionally, the method favors the use of Boc (tert-butyloxycarbonyl) or similar acid-labile protecting groups for the amine functionality. This choice is pivotal as it allows for deprotection under mild acidic conditions, completely bypassing the need for heavy metal hydrogenation. The result is a streamlined process that offers cost reduction in lipid nanoparticle manufacturing by eliminating expensive catalytic steps and improving the stability of intermediates throughout the synthesis.
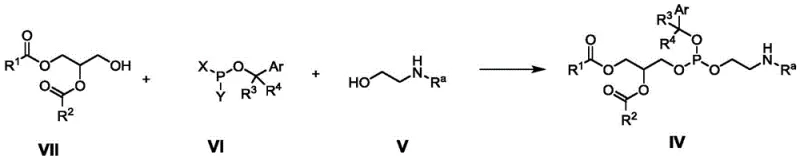
Mechanistically, the synthesis hinges on a precise three-component condensation involving a di-fatty acyl glycerol alcohol ester (Formula VII), the modified phosphitylating agent (Formula VI), and a protected ethanolamine (Formula V). The reaction is typically catalyzed by nitrogenous reagents such as tetrazole or phenyltetrazole, which activate the phosphorus center for nucleophilic attack by the alcohol and amine components. This activation ensures that the phosphite triester intermediate (Formula IV) is formed with high regioselectivity. Following condensation, a controlled oxidation step using agents like m-chloroperoxybenzoic acid (mCPBA) converts the trivalent phosphorus into the stable pentavalent phosphate state (Formula III). This two-stage process—condensation followed by oxidation—allows for better control over impurity profiles compared to direct phosphorylation. The final deprotection step utilizes acids like trifluoroacetic acid to cleave both the amino and hydroxyl protecting groups simultaneously or sequentially, yielding the target phosphatidylethanolamine (Formula II) with minimal degradation of the sensitive fatty acid chains.
Impurity control is rigorously maintained through the stability of the intermediates. The use of bulky aryl groups on the phosphitylating reagent provides steric shielding that prevents hydrolysis during the workup phase. Furthermore, the choice of solvent systems, such as dichloromethane or tetrahydrofuran, combined with low-temperature controls during reagent addition, minimizes thermal degradation. The purification strategy often involves simple column chromatography or slurry washing, which effectively removes phosphorous-containing by-products and unreacted starting materials. This level of control is critical for producing high-purity phosphatidylethanolamine suitable for parenteral applications, where trace metal contaminants or organic impurities can trigger immunogenic responses.
How to Synthesize Phosphatidylethanolamine Intermediates Efficiently
The synthesis protocol outlined in the patent provides a reproducible framework for generating these critical lipid components. The process begins with the preparation of the di-fatty acyl glycerol backbone, followed by the key phosphitylation step. Operators must ensure strict anhydrous conditions to prevent premature hydrolysis of the phosphitylating agent. The reaction temperature is carefully managed, typically starting at low temperatures (0°C to -30°C) during reagent addition and warming to room temperature to drive completion. Monitoring via TLC or LC-MS is essential to confirm the consumption of starting materials before proceeding to the oxidation phase. The detailed standardized synthesis steps see the guide below.
- Perform a three-component condensation reaction between a di-fatty acyl glycerol ester, a modified phosphitylating agent (Formula VI), and a protected ethanolamine under nitrogenous catalysis.
- Oxidize the resulting phosphite triester intermediate using mild oxidizing agents such as m-chloroperoxybenzoic acid to form the phosphate bond.
- Execute acidic deprotection to remove amino and hydroxyl protecting groups simultaneously, yielding the final phosphatidylethanolamine or its PEGylated derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the deprotection workflow. By eliminating the reliance on palladium-carbon hydrogenation, facilities can avoid the capital expenditure associated with high-pressure hydrogenation reactors and the recurring costs of precious metal catalysts. This shift not only reduces the direct cost of goods sold but also mitigates supply chain risks associated with the volatility of precious metal markets. Additionally, the mild reaction conditions reduce energy consumption and lower the safety burden on manufacturing sites, facilitating smoother regulatory audits and faster time-to-market for new lipid formulations.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and hydrogenation equipment significantly lowers operational expenditures. The use of readily available reagents like tetrazole and mCPBA, combined with simplified purification steps, reduces the overall material cost per kilogram. Furthermore, the higher selectivity of the new phosphitylating agents minimizes waste generation, leading to improved atom economy and reduced disposal costs for hazardous by-products.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including various fatty acids and protected ethanolamines, are commodity chemicals with robust global supply chains. Unlike specialized phosphonylating reagents that may have single-source suppliers, the modified reagents described can be synthesized in-house or sourced from multiple vendors. This diversification ensures reducing lead time for high-purity phosphatidylethanolamine intermediates and protects against supply disruptions caused by geopolitical or logistical factors.
- Scalability and Environmental Compliance: The process operates under mild conditions without the need for extreme temperatures or pressures, making it inherently safer and easier to scale from pilot batches to multi-ton production. The avoidance of toxic reagents like POCl3 and the reduction of heavy metal usage align with green chemistry principles, simplifying environmental compliance and waste treatment. This sustainability profile is increasingly important for pharmaceutical companies aiming to meet corporate ESG (Environmental, Social, and Governance) goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, safety profiles, and compatibility with downstream PEGylation processes.
Q: Why is the novel phosphitylating agent superior to traditional PCl3 methods?
A: Traditional PCl3 methods often suffer from over-reaction where two identical alcohol fragments link to the phosphorus, reducing yield. The novel modified reagents (Formula VI) offer better steric control and stability, preventing side reactions and ensuring high selectivity for the desired ethanolamine linkage.
Q: How does this method improve cost efficiency compared to Cbz-protection routes?
A: Conventional routes using Cbz protection require palladium-carbon hydrogenation for removal, which involves expensive catalysts, specialized hydrogenation equipment, and safety risks. The new method utilizes Boc or similar groups that can be removed under mild acidic conditions, eliminating the need for heavy metal catalysts and significantly lowering operational costs.
Q: Is this synthesis route suitable for large-scale manufacturing of LNPs?
A: Yes, the process features mild reaction conditions (often room temperature to 60°C) and avoids highly toxic or unstable reagents. The high selectivity and ease of purification via column chromatography or slurry washing make it highly scalable for commercial production of lipid nanoparticle components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphatidylethanolamine Intermediate Supplier
As the demand for lipid-based drug delivery systems continues to surge, securing a dependable source of high-quality intermediates is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the one described in CN115433224A to deliver superior products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest standards required for clinical and commercial applications.
We invite you to collaborate with us to optimize your lipid nanoparticle supply chain. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific formulation requirements. Contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your development timeline with reliable, cost-effective solutions.
