Advanced Manufacturing of High-Purity 2-Aminobutyric Acid for Levetiracetam Production
Advanced Manufacturing of High-Purity 2-Aminobutyric Acid for Levetiracetam Production
The pharmaceutical landscape for antiepileptic drugs continues to evolve, with Levetiracetam standing out as a critical therapeutic agent due to its broad-spectrum efficacy and superior safety profile. At the heart of synthesizing this vital medication lies the production of its key precursor, 2-aminobutyric acid. Patent CN101838211A introduces a transformative processing method that addresses longstanding challenges in yield, purity, and cost-efficiency associated with traditional synthesis routes. By leveraging a modified Strecker synthesis starting from n-propanal, this innovation offers a robust pathway to achieve product content exceeding 99%, meeting the rigorous demands of international regulatory bodies. For R&D directors and procurement specialists alike, understanding this technological shift is paramount for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality.
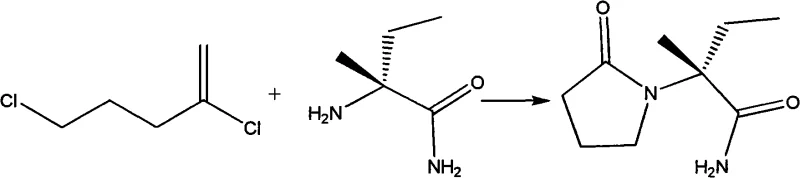
This patent details a comprehensive workflow that begins with the condensation of n-propanal, ammonium bicarbonate, and sodium cyanide to form an aminonitrile intermediate, often referred to as 'Haiyin' in the literature. Following this, a controlled low-pressure alkaline hydrolysis converts the intermediate into sodium 2-aminobutyrate. The process concludes with precise pH adjustment and recrystallization to isolate the final amino acid while effectively removing inorganic salt byproducts. This methodological rigor ensures that the resulting 2-aminobutyric acid is not only chemically pure but also free from the hazardous impurities that plague older manufacturing techniques, thereby streamlining the downstream synthesis of Levetiracetam.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-aminobutyric acid has been fraught with significant technical and economic hurdles that hinder efficient commercial scale-up of complex amino acids. One prevalent traditional route involves the bromination of butyric acid, where bromine substitutes the alpha-hydrogen atoms. This approach is inherently flawed due to the generation of corrosive hydrogen bromide gas, creating severe safety hazards and requiring expensive corrosion-resistant reactor infrastructure. Furthermore, the lack of selectivity in radical bromination often leads to poly-brominated byproducts, making the final purification extremely difficult and resulting in suboptimal purity levels that fail to meet modern pharmaceutical standards.
Another conventional pathway utilizes methionine as a starting material, employing desulfurization and demethylation reactions to yield the target amino acid. While chemically straightforward, this route is economically prohibitive for large-scale manufacturing due to the high cost of methionine feedstock. 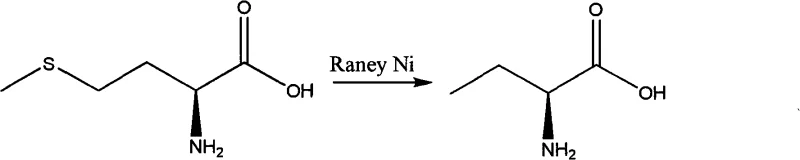 As illustrated in the reaction above, the reliance on precious metal catalysts like Raney Nickel adds another layer of complexity, necessitating rigorous metal removal steps to prevent contamination. These legacy methods collectively contribute to inflated production costs and extended lead times, creating supply chain vulnerabilities for manufacturers of high-purity intermediates.
As illustrated in the reaction above, the reliance on precious metal catalysts like Raney Nickel adds another layer of complexity, necessitating rigorous metal removal steps to prevent contamination. These legacy methods collectively contribute to inflated production costs and extended lead times, creating supply chain vulnerabilities for manufacturers of high-purity intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a Strecker-type synthesis that fundamentally reimagines the carbon skeleton construction of the molecule. By reacting n-propanal with ammonium bicarbonate and sodium cyanide, the process achieves high atom economy and selectivity under mild thermal conditions ranging from 40°C to 50°C. This eliminates the need for hazardous halogenation reagents and expensive sulfur-containing precursors. The subsequent hydrolysis step is performed under low-pressure alkaline conditions, which simplifies the engineering requirements and enhances operational safety compared to high-pressure acid hydrolysis methods.
The true brilliance of this novel approach lies in its downstream processing strategy, which integrates pH control and recrystallization to achieve exceptional purity. By adjusting the pH to the equivalence point using hydrochloric acid and subsequently employing ethanol for dissolution and filtration, the process effectively separates the organic amino acid from inorganic sodium chloride byproducts. This results in a final product with content exceeding 99%, directly addressing the impurity concerns of R&D teams. For procurement managers, this translates to cost reduction in API manufacturing, as the raw material costs are reported to be approximately half of those associated with the two traditional routes mentioned previously.
Mechanistic Insights into Strecker-Based Amino Acid Synthesis
The core of this innovative process relies on the classic yet optimized Strecker reaction mechanism, adapted here for industrial robustness. The reaction initiates with the nucleophilic attack of ammonia (derived from ammonium bicarbonate) on the carbonyl carbon of n-propanal, forming an imine intermediate. This imine is subsequently attacked by the cyanide ion from sodium cyanide to generate the alpha-aminonitrile. The patent specifies a precise molar ratio of n-propanal to sodium cyanide to ammonium bicarbonate of roughly 1:1~1.5:1.2~1.8, ensuring that the equilibrium is driven towards the formation of the nitrile while minimizing side reactions. Maintaining the temperature between 40°C and 50°C during this phase is critical to prevent the decomposition of unstable intermediates while ensuring complete conversion over a 22 to 28-hour period.
Following the formation of the aminonitrile, the mechanism shifts to alkaline hydrolysis, where the nitrile group is converted into a carboxylate. Under the specified conditions of 100°C to 130°C with a 30% alkali solution, the cyano group undergoes hydration to form an amide, which is further hydrolyzed to the carboxylic acid salt (sodium 2-aminobutyrate). The use of low pressure during this stage prevents the loss of volatile ammonia and ensures the integrity of the reaction vessel. Finally, the acidification step protonates the carboxylate to form the zwitterionic amino acid. The careful adjustment of pH to between 5.8 and 7.2 is crucial; this range corresponds to the isoelectric point of the amino acid, minimizing its solubility in the aqueous phase and facilitating precipitation, while keeping impurities in solution for removal during the final recrystallization.
How to Synthesize 2-Aminobutyric Acid Efficiently
Implementing this synthesis route requires strict adherence to the reaction parameters outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the reaction mixture in a stirred vessel, where temperature control is paramount during the exothermic addition of n-propanal. Operators must monitor the progression of the aminonitrile formation closely, as incomplete conversion at this stage can lead to carryover impurities that complicate the subsequent hydrolysis. Once the nitrile is formed, the transition to the hydrolysis phase involves the addition of concentrated alkali and a controlled ramp-up of temperature to facilitate the cleavage of the triple bond. The detailed standardized synthesis steps below provide a clear roadmap for laboratory and pilot-scale execution.
- React n-propanal with ammonium bicarbonate and sodium cyanide at 40-50°C to form the aminonitrile intermediate.
- Hydrolyze the intermediate under low-pressure alkaline conditions at 100-130°C to obtain sodium 2-aminobutyrate.
- Adjust pH to 5.8-7.2 using hydrochloric acid and an acid-binding agent, then recrystallize to remove sodium chloride byproducts.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the adoption of this patented process offers tangible strategic advantages beyond mere chemical elegance. The shift from expensive, specialty starting materials like methionine to commodity chemicals such as n-propanal and ammonium bicarbonate drastically simplifies the sourcing landscape. These raw materials are widely available in the global chemical market, reducing the risk of supply disruptions and price volatility that often plague niche precursor markets. Furthermore, the elimination of hazardous reagents like elemental bromine reduces the regulatory burden and insurance costs associated with handling dangerous goods, contributing to a more resilient and compliant supply chain.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the substitution of high-cost feedstocks with inexpensive bulk chemicals. By avoiding the use of methionine and eliminating the need for expensive metal catalysts like Raney Nickel, the direct material costs are significantly lowered. Additionally, the simplified purification protocol, which relies on standard recrystallization rather than complex chromatographic separations or distillation, reduces energy consumption and solvent usage. This holistic reduction in operational expenditure allows for substantial cost savings that can be passed down the value chain, enhancing the competitiveness of the final API.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is non-negotiable for pharmaceutical manufacturers. This process enhances reliability by utilizing a robust chemical pathway that is less sensitive to minor fluctuations in reaction conditions compared to radical bromination. The use of stable, non-volatile reagents and the avoidance of corrosive gases mean that production can be sustained over long campaigns without frequent maintenance shutdowns. This stability ensures a consistent flow of high-purity 2-aminobutyric acid, reducing lead time for high-purity intermediates and allowing downstream API manufacturers to maintain leaner inventory levels.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this method is exceptionally well-suited for commercial expansion. The reaction conditions operate at moderate temperatures and low pressures, meaning that existing standard stainless-steel reactors can be utilized without the need for exotic alloys or high-pressure autoclaves. The waste stream is primarily composed of benign inorganic salts and recoverable solvents like ethanol, simplifying wastewater treatment and aligning with green chemistry principles. This ease of scale-up facilitates the transition from kilogram-scale development to multi-ton commercial production with minimal capital investment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-aminobutyric acid using this novel methodology. These insights are derived directly from the technical specifications and comparative data provided in the patent documentation. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this intermediate into their existing manufacturing portfolios.
Q: How does this new process improve purity compared to bromination routes?
A: Unlike bromination routes which generate difficult-to-separate poly-brominated byproducts and corrosive HBr gas, this Strecker-based synthesis utilizes selective nucleophilic addition. The subsequent recrystallization steps effectively remove inorganic salts like sodium chloride, achieving purity levels exceeding 99% suitable for strict pharmaceutical standards.
Q: What are the primary cost drivers reduced in this manufacturing method?
A: The primary cost reduction comes from replacing expensive starting materials like methionine with commodity chemicals such as n-propanal and ammonium bicarbonate. Additionally, the elimination of complex purification steps required for removing heavy metal catalysts or halogenated impurities significantly lowers operational expenditures.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scalability. It operates under low-pressure alkaline conditions and utilizes standard unit operations like hydrolysis, filtration, and recrystallization, making it highly adaptable for small to medium-sized enterprises aiming for commercial tonnage production without specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminobutyric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of life-saving medications like Levetiracetam. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-aminobutyric acid meets the exacting standards required for pharmaceutical synthesis. Our facility is equipped to handle the specific unit operations required by this patent, from controlled aminonitrile formation to precise recrystallization.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project timelines and quality expectations. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing.
