Scaling High-Purity Dihalogenated Aromatic Ethers via Solvent-Free Phase Transfer Catalysis
The chemical industry is currently witnessing a paradigm shift towards greener, more efficient synthetic methodologies, particularly in the production of critical intermediates like dihalogenated aromatic ethers. Patent CN1271027C introduces a groundbreaking approach to synthesizing these compounds by utilizing phase transfer catalysis (PTC) in a completely aqueous environment, effectively eliminating the need for hazardous organic solvents. This innovation addresses long-standing challenges in the manufacturing of agrochemical and pharmaceutical intermediates, where traditional methods often suffer from low yields and complex purification requirements. By leveraging water as the sole solvent, this technology not only simplifies the downstream processing but also enables reaction conditions that were previously unattainable due to solvent limitations. For R&D directors and process engineers, this represents a significant opportunity to optimize existing production lines for dihalogenated aromatic ethers, ensuring higher throughput and reduced environmental footprint without compromising on product quality.
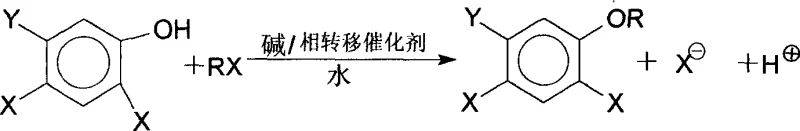
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dihalogenated aromatic ethers, such as 2,4-dichlorophenylisopropyl ether, has relied heavily on the use of organic solvents like toluene or cyclohexane to facilitate the phase transfer catalytic reaction. While effective to a degree, these conventional methods introduce significant inefficiencies into the manufacturing process. The presence of organic solvents creates azeotropic mixtures with water, which inherently limits the maximum reaction temperature to relatively low levels, typically around 65°C to 75°C. This thermal ceiling severely restricts reaction kinetics, resulting in prolonged reaction times often exceeding 10 hours and yielding suboptimal conversion rates, frequently hovering around 55%. Furthermore, the reliance on volatile organic compounds necessitates complex and energy-intensive recovery and rectification steps post-reaction. Incomplete removal of these solvents can lead to contamination in subsequent synthetic steps, requiring additional purification measures that drive up operational costs and extend production lead times significantly.
The Novel Approach
In stark contrast, the novel methodology described in patent CN1271027C revolutionizes this synthesis by completely abandoning organic solvents in favor of a water-only system. This bold departure from tradition unlocks the ability to heat the reaction mixture to substantially higher temperatures, specifically in the range of 90°C to 120°C, without the constraint of solvent boiling points or azeotrope formation. The elevated thermal energy dramatically accelerates the nucleophilic substitution between the dihalophenol and the brominated hydrocarbon, reducing reaction times to merely 4-6 hours while boosting yields to over 90%. The elimination of organic solvents also streamlines the workup procedure; there is no need for energy-heavy distillation columns to recover toluene or cyclohexane. Instead, the product can be isolated through simple liquid-liquid separation after washing, resulting in a final product with gas chromatography purity exceeding 95%. This streamlined approach not only enhances safety by removing flammable solvents but also aligns perfectly with modern green chemistry principles.
Mechanistic Insights into Phase Transfer Catalyzed Etherification
To fully appreciate the efficacy of this solvent-free system, one must understand the intricate mechanistic role played by the phase transfer catalyst. In this biphasic system, the inorganic phase consists of the aqueous sodium hydroxide solution which deprotonates the dihalophenol to form a substituted phenoxide anion. However, this anion is highly hydrophilic and remains trapped in the water layer, unable to react with the hydrophobic brominated hydrocarbon which forms the organic phase. The phase transfer catalyst, typically a quaternary ammonium or phosphonium salt, acts as a molecular shuttle. Its lipophilic cationic head associates with the phenoxide anion, forming an ion pair that is soluble in the organic phase. Once transported across the interface, the phenoxide anion acts as a potent nucleophile, attacking the electrophilic carbon of the brominated hydrocarbon to form the desired ether bond. After the substitution, the resulting halide anion pairs with the catalyst cation and is shuttled back to the aqueous phase to exchange for another phenoxide anion, completing the catalytic cycle.
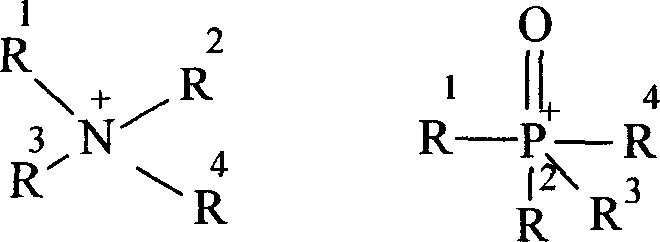
The choice of catalyst is critical for maximizing the efficiency of this interfacial transport. The patent highlights the effectiveness of quaternary ammonium salts such as cetyltrimethylammonium bromide and triethylbenzyl ammonium chloride, as well as phosphonium salts like tetrabutylphosphonium chloride. These structures feature long alkyl chains or aryl groups that provide the necessary lipophilicity to penetrate the organic phase effectively. Moreover, the stability of these catalysts under the harsh alkaline and high-temperature conditions (up to 120°C) ensures that they do not degrade during the reaction, allowing for sustained catalytic activity throughout the process. This robustness is essential for maintaining high turnover numbers and ensuring that the catalyst loading can be kept minimal, typically between 0.002 and 0.01 parts by weight relative to the phenol, further contributing to the economic viability of the process.
How to Synthesize Dihalogenated Aromatic Ethers Efficiently
Implementing this solvent-free protocol requires precise control over reaction parameters to maximize the benefits of the phase transfer mechanism. The process begins by charging the reactor with the dihalophenol substrate, a calculated portion of solid sodium hydroxide, the selected phase transfer catalyst, and water. It is crucial to note that the base is not added all at once; rather, a split-feed strategy is employed to manage the pH profile of the reaction mixture. The mixture is heated to initiate the dissolution and formation of the phenoxide species before the introduction of the alkylating agent. Detailed standard operating procedures regarding specific molar ratios, temperature ramping rates, and agitation speeds are critical for reproducibility on a commercial scale. For a comprehensive breakdown of the exact experimental conditions and step-by-step execution, please refer to the standardized synthesis guide below.
- Charge the reactor with dihalophenol, a portion of solid NaOH, phase transfer catalyst, and water, then heat to 90-120°C.
- Slowly add brominated hydrocarbon dropwise while maintaining temperature at 90-100°C and stir for 4-6 hours.
- Add the remaining NaOH, react for 1-2 hours, cool below 50°C, wash with NaOH solution, and separate the organic layer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this solvent-free PTC technology offers compelling economic and logistical advantages that extend far beyond simple yield improvements. The most immediate impact is seen in the drastic reduction of raw material costs associated with organic solvents. By eliminating the need for tons of toluene or cyclohexane per batch, manufacturers can significantly lower their direct material expenditure. Additionally, the removal of solvent recovery infrastructure reduces capital expenditure on distillation equipment and lowers the ongoing energy costs required to run these units. The simplified workflow also translates to shorter batch cycles, allowing facilities to increase their production capacity without expanding their physical footprint. This efficiency gain is vital for meeting tight delivery schedules in the fast-paced agrochemical and pharmaceutical sectors.
- Cost Reduction in Manufacturing: The elimination of organic solvents removes the entire cost center associated with solvent purchase, storage, and recovery. Traditional methods require significant energy input to distill and recycle solvents, whereas this aqueous method relies on simple phase separation. Furthermore, the higher reaction yields (>90%) mean that less raw material is wasted on side products or unreacted starting materials, directly improving the cost-of-goods-sold (COGS). The reduction in waste disposal costs is also substantial, as the aqueous waste stream is easier and cheaper to treat compared to mixed organic-aqueous waste streams containing halogenated solvents.
- Enhanced Supply Chain Reliability: Relying on water as the primary solvent mitigates supply chain risks associated with the volatility of petrochemical-derived solvents. Organic solvents are subject to price fluctuations based on crude oil markets and can face supply disruptions. Water, conversely, is universally available and cost-stable. Additionally, the simplified process flow reduces the number of unit operations required, decreasing the likelihood of mechanical failures or bottlenecks in the production line. This robustness ensures a more consistent and reliable supply of high-purity intermediates to downstream customers, safeguarding their own production schedules against upstream delays.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this technology inherently minimizes them. The absence of flammable organic solvents drastically reduces the fire hazard rating of the facility, potentially lowering insurance premiums and simplifying safety compliance protocols. From an environmental standpoint, the process generates significantly less volatile organic compound (VOC) emissions, making it easier to meet stringent environmental regulations. The high selectivity of the reaction also means fewer by-products are formed, simplifying the purification process and reducing the volume of hazardous chemical waste that requires specialized disposal, thereby supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is essential for stakeholders evaluating its adoption for commercial production. The following questions address common concerns regarding process safety, catalyst selection, and product quality assurance. These answers are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a reliable foundation for decision-making. Whether you are concerned about the stability of the catalyst at high temperatures or the purity profile of the final ether product, the following insights clarify how this technology performs under rigorous industrial conditions.
Q: What are the primary advantages of using water instead of organic solvents in this synthesis?
A: Using water eliminates the need for complex solvent recovery and rectification steps, significantly reducing energy consumption and environmental impact. Furthermore, the absence of organic solvents allows the reaction mixture to be heated to higher temperatures (above 90°C), which drastically accelerates reaction kinetics and improves overall yield compared to traditional methods limited by solvent boiling points.
Q: How does the two-step addition of NaOH improve the reaction outcome?
A: Adding NaOH in two stages prevents the alkalinity of the mixture from becoming too high during the initial reaction phase. This controlled basicity minimizes the hydrolysis of the dihalophenol substrate, which is a common side reaction that lowers efficiency. By optimizing the base concentration dynamically, the process achieves yields exceeding 90% while maintaining high product purity.
Q: Which phase transfer catalysts are most effective for this transformation?
A: The patent specifies that both quaternary ammonium salts and phosphonium salts are highly effective. Specific examples include cetyltrimethylammonium bromide, triethylbenzyl ammonium chloride, and tetrabutylphosphonium chloride. These catalysts facilitate the transport of the phenoxide anion from the aqueous phase into the organic phase where the nucleophilic substitution occurs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihalogenated Aromatic Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful implementation of advanced synthetic routes like the solvent-free PTC method requires more than just theoretical knowledge; it demands extensive practical experience in process engineering and scale-up. As a leading CDMO partner, we possess the technical expertise to translate laboratory-scale innovations into robust, commercial-scale manufacturing processes. Our facilities are equipped to handle diverse synthetic pathways, ranging from small-batch custom synthesis of 100 kgs to large-scale annual commercial production of 100 MT. We maintain stringent purity specifications through our rigorous QC labs, ensuring that every batch of dihalogenated aromatic ether meets the exacting standards required by the global agrochemical and pharmaceutical industries. Our commitment to quality and consistency makes us the ideal partner for companies seeking to secure their supply chain for these critical intermediates.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your specific supply chain requirements. Our technical team is ready to conduct a Customized Cost-Saving Analysis tailored to your current production volumes and quality needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to our deep reservoir of chemical knowledge and our state-of-the-art manufacturing capabilities. We encourage you to contact our technical procurement team today to request specific COA data for our dihalogenated aromatic ether portfolio and to discuss route feasibility assessments for your upcoming projects. Let us help you drive efficiency and sustainability in your chemical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
