Advanced Synthesis of Trans-Alpha-Benzoin Oxime for High-Efficiency Metallurgical Extraction
The chemical industry is witnessing a significant paradigm shift towards greener synthetic methodologies, particularly in the production of specialized chelating agents used in hydrometallurgy. Patent CN102702023A introduces a robust and environmentally benign synthesis route for trans-alpha-benzoin oxime, a critical reagent in the extraction and separation of valuable metals like copper, molybdenum, and tungsten. This innovation addresses the longstanding challenges associated with traditional manufacturing processes, which have historically relied on hazardous reagents and harsh reaction conditions. By leveraging imidazolium salt ionic liquids as organocatalysts, this method not only enhances the safety profile of the production line but also improves the selectivity towards the biologically and chemically active trans-isomer. For R&D directors and procurement specialists in the mining chemical sector, this technology represents a viable pathway to securing high-purity intermediates while adhering to increasingly stringent global environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-benzoin derivatives, which serve as the precursors to alpha-benzoin oxime, has been plagued by significant safety and operational drawbacks. The classical bimolecular reduction of esters requires the use of extremely reactive metals such as sodium or alkyllithium reagents, necessitating absolutely anhydrous solvent conditions that are difficult and costly to maintain on an industrial scale. Furthermore, the alternative benzoin condensation reaction traditionally employs sodium cyanide or potassium cyanide as catalysts, substances that pose severe toxicity risks to personnel and create complex waste disposal challenges for facility managers. These conventional routes often suffer from poor selectivity, leading to mixtures of isomers that require energy-intensive separation processes, ultimately driving up the cost of goods sold and compromising the reliability of the supply chain for downstream metallurgical applications.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes imidazolium salt ionic liquids to catalyze the benzoin condensation under mild alkaline conditions, effectively eliminating the need for toxic cyanides or pyrophoric metals. This novel catalytic system operates efficiently at temperatures ranging from room temperature to 70°C, significantly reducing energy consumption and thermal stress on the equipment. The use of these ionic liquids facilitates a smoother reaction profile with improved yields, while the subsequent oximation step is optimized to favor the formation of the desired product. By integrating this green chemistry approach, manufacturers can achieve a substantial reduction in hazardous waste generation and simplify the overall process flow, making it an attractive option for cost reduction in mining chemical manufacturing without sacrificing product quality.
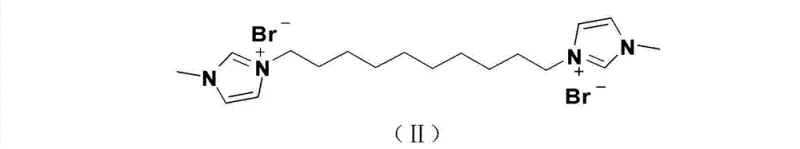
Mechanistic Insights into Imidazolium Salt-Catalyzed Benzoin Condensation
The core of this synthetic breakthrough lies in the unique mechanistic pathway enabled by the imidazolium salt catalyst, which functions through an N-heterocyclic carbene (NHC) intermediate. Under basic conditions, the imidazolium salt generates a nucleophilic carbene species that attacks the carbonyl carbon of the benzaldehyde substrate, inducing an umpolung or polarity inversion of the carbonyl group. This activation allows for the nucleophilic addition of a second benzaldehyde molecule, forming the carbon-carbon bond essential for the benzoin skeleton. This organocatalytic cycle is highly efficient and avoids the heavy metal contamination often associated with transition metal catalysts, ensuring that the final product meets the rigorous purity specifications required for analytical and extractive applications in the mining industry.
Following the formation of the alpha-benzoin intermediate, the subsequent oximation reaction must be carefully controlled to maximize the ratio of the trans-isomer, as only this stereoisomer exhibits the necessary chelating properties for metal extraction. The patent highlights that reaction temperature and purification techniques play pivotal roles in isomer control; while the reaction may produce a mixture, the implementation of gradient column chromatography allows for the precise separation of the trans-alpha-benzoin oxime from its cis-counterpart. This level of purification is critical because the presence of the inactive cis-isomer can dilute the efficacy of the final chelating agent, thereby reducing its value in high-stakes mineral processing operations where recovery rates are paramount.
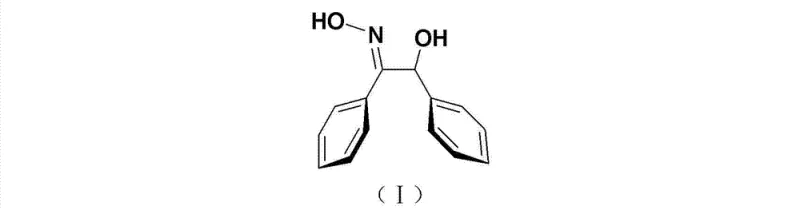
How to Synthesize Trans-Alpha-Benzoin Oxime Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step framework for producing high-purity trans-alpha-benzoin oxime suitable for commercial deployment. The process begins with the condensation of benzaldehyde in the presence of the ionic liquid catalyst and a mild base, followed by a straightforward workup to isolate the alpha-benzoin intermediate. The detailed standardized synthesis steps below describe the precise molar ratios, solvent choices, and purification techniques required to replicate this success in a pilot or production plant setting, ensuring consistent quality and yield across batches.
- Perform benzoin condensation of benzaldehyde using an imidazolium salt ionic liquid catalyst and base in organic solvent at mild temperatures.
- Isolate the intermediate alpha-benzoin through aqueous workup and recrystallization to ensure high purity before the next step.
- React the purified alpha-benzoin with hydroxylamine hydrochloride under basic conditions, followed by gradient column chromatography to isolate the trans-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ionic liquid-catalyzed synthesis offers compelling strategic advantages beyond mere technical feasibility. The shift away from regulated and hazardous substances like cyanides simplifies the regulatory compliance landscape, reducing the administrative burden and insurance costs associated with handling toxic materials. Furthermore, the reliance on readily available starting materials such as benzaldehyde and common organic solvents ensures a stable and resilient supply chain, mitigating the risks of raw material shortages that can disrupt production schedules. This robustness is essential for maintaining continuous operations in the volatile market of specialty mining chemicals.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous catalysts like sodium cyanide directly lowers the raw material costs and the associated expenses for safety infrastructure and waste treatment. By operating under milder conditions, the process also reduces energy consumption for heating and cooling, contributing to a lower overall carbon footprint and operational expenditure. Additionally, the simplified purification workflow minimizes product loss during isolation, enhancing the overall mass balance and economic efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: The use of stable ionic liquid catalysts and common reagents reduces dependency on specialized chemical suppliers who may face production bottlenecks. This flexibility allows for more agile sourcing strategies and shorter lead times for high-purity mining chemical intermediates, ensuring that downstream metallurgical plants receive their critical reagents without delay. The robustness of the reaction conditions also means that production is less susceptible to minor fluctuations in environmental parameters, guaranteeing consistent output.
- Scalability and Environmental Compliance: The green nature of this synthesis aligns perfectly with modern environmental, health, and safety (EHS) standards, facilitating easier permitting for capacity expansion. The process generates less hazardous waste, simplifying effluent treatment and reducing the environmental liability of the manufacturing site. This scalability ensures that the technology can be seamlessly transitioned from laboratory benchtop to multi-ton commercial production to meet growing global demand for efficient metal extraction agents.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of trans-alpha-benzoin oxime, based on the detailed specifications provided in the patent literature. These insights are designed to clarify the operational benefits and technical nuances for stakeholders evaluating this technology for integration into their supply chains.
Q: Why is the trans-isomer of alpha-benzoin oxime preferred for metallurgical applications?
A: The trans-isomer possesses the specific stereochemical configuration required to effectively chelate metal ions such as copper, molybdenum, and tungsten, whereas the cis-isomer lacks the necessary spatial arrangement for stable complex formation.
Q: How does the ionic liquid catalyst improve safety compared to traditional methods?
A: Traditional benzoin condensation often relies on highly toxic cyanide salts or dangerous alkali metals; the imidazolium ionic liquid catalyst operates under mild, non-toxic conditions, significantly reducing environmental and operator hazards.
Q: What purification method ensures the highest isomeric purity?
A: While recrystallization is used for the intermediate, the final trans-alpha-benzoin oxime is best purified using gradient column chromatography, which effectively separates the closely related cis and trans isomers that are difficult to distinguish by simple crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-Alpha-Benzoin Oxime Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance chelating agents play in the efficiency of modern hydrometallurgy and mineral processing. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of this vital intermediate. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of trans-alpha-benzoin oxime meets the exacting standards required for effective metal ion separation and analysis.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this greener manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and sustainability in the competitive global market.
