Industrial Scale Synthesis of 2-Chloro-5-Cyanopyrimidine via Cyanoacetic Acid Route
The pharmaceutical industry is constantly seeking robust, scalable, and cost-effective pathways for critical oncology intermediates. Patent CN110845426A, published in early 2020, introduces a transformative preparation method for 2-chloro-5-cyanopyrimidine (CAS: 1753-50-0), a pivotal building block in the synthesis of anticancer agents. This technology represents a significant departure from traditional synthetic strategies by utilizing cyanoacetic acid as a foundational raw material. The disclosed route leverages a sequence involving N,N-dimethylformamide dimethyl acetal (DMF-DMA) condensation, followed by Vilsmeier reaction, addition, ring closure, and final chlorination. For R&D directors and procurement specialists, this patent offers a compelling solution to the chronic issues of high raw material costs and complex purification associated with legacy methods. By shifting the synthetic entry point to commodity chemicals, the process effectively bypasses the economic and environmental bottlenecks of transition metal catalysis and hazardous gas handling.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 2-chloro-5-cyanopyrimidine was plagued by three distinct but equally problematic methodologies, each presenting severe barriers to efficient industrial production. The first conventional route, often cited in earlier literature, relies on heavy metal chemistry that is increasingly untenable in modern GMP environments. As illustrated below, this pathway typically involves the use of copper cyanide and subsequent diazotization steps.
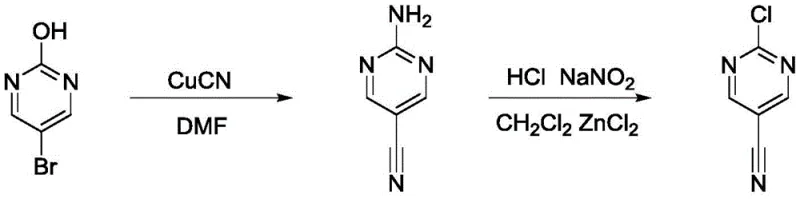
This approach suffers from inherently low reaction yields and generates substantial heavy metal waste, necessitating expensive and rigorous purification protocols to meet residual metal specifications for pharmaceutical use. Furthermore, the second historical method extends the synthetic timeline unnecessarily, introducing significant safety risks through the utilization of phosgene or its highly toxic equivalents. The complexity of this multi-step sequence, depicted in the following scheme, results in a diminished overall yield and complicates supply chain management due to the handling of hazardous gases.
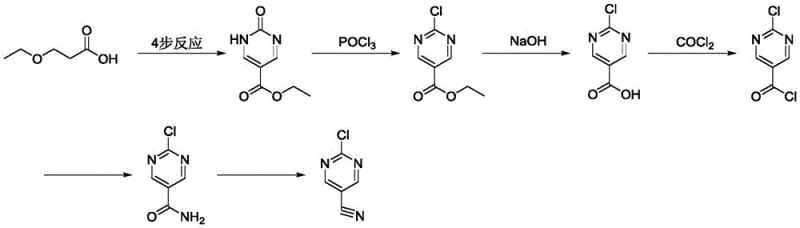
Finally, a more recent approach attempted to streamline the process using palladium-catalyzed carbonylation. While chemically elegant, this method is economically fragile due to its dependence on precious metal catalysts. The volatility of palladium prices directly impacts the cost of goods sold (COGS), and the requirement for specialized ligands adds another layer of expense. Additionally, the removal of trace palladium from the final API intermediate remains a persistent technical challenge, often requiring scavengers that further erode profit margins.
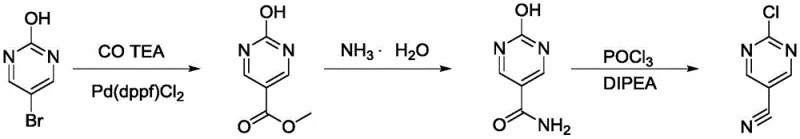
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in CN110845426A reconstructs the pyrimidine core from simple, abundant precursors. By initiating the synthesis with cyanoacetic acid, the process immediately sidesteps the need for expensive halogenated pyrimidine starting materials or precious metal catalysts. The strategy employs a clever cascade where cyanoacetic acid is first condensed with DMF-DMA to form 3-(dimethylamino)acrylonitrile. This intermediate then undergoes a Vilsmeier-Haack type formylation using oxalyl chloride, setting the stage for ring closure. The subsequent cyclization with O-methoxyisourea sulfate efficiently constructs the pyrimidine nucleus, which is finally chlorinated using phosphorus pentachloride. This linear progression is not only chemically robust but also operationally simple, eliminating the need for column chromatography at every stage and relying instead on standard crystallization and extraction techniques.
Mechanistic Insights into the Cyanoacetic Acid Cascade
The mechanistic elegance of this synthesis lies in its ability to build complexity from simplicity without generating intractable impurity profiles. The initial condensation of cyanoacetic acid with DMF-DMA proceeds via a nucleophilic attack of the enol form of the acid on the electrophilic carbon of the acetal, followed by elimination of methanol and dimethylamine to yield the electron-rich enamine intermediate, 3-(dimethylamino)acrylonitrile. This species is crucial as it activates the alpha-position for the subsequent Vilsmeier reaction. When treated with the Vilsmeier reagent generated in situ from DMF and oxalyl chloride, the enamine undergoes electrophilic substitution. The use of oxalyl chloride here is particularly advantageous as it acts as a mild dehydrating agent and chlorinating source, facilitating the formation of the reactive iminium salt which is essential for the next cyclization step.
The ring-closing step involves the reaction of the Vilsmeier intermediate with O-methoxyisourea sulfate in the presence of pyridine. Pyridine serves a dual role here: it acts as a solvent and as a base to neutralize the acid byproducts, driving the equilibrium towards the formation of the pyrimidine ring. The nucleophilic nitrogen of the isourea attacks the electrophilic center of the Vilsmeier adduct, followed by intramolecular cyclization and elimination of dimethylamine to form 2-methoxy-4-cyanopyrimidine. The final transformation involves the conversion of the methoxy group to a chloro group using phosphorus pentachloride (PCl5). This chlorination is highly selective for the 2-position of the pyrimidine ring due to the activation by the adjacent nitrogen atoms. The mechanism ensures that the nitrile group at the 5-position remains intact, preserving the functionality required for downstream coupling reactions in API synthesis. This precise control over regioselectivity minimizes the formation of isomeric impurities, thereby simplifying the purification workflow.
How to Synthesize 2-Chloro-5-Cyanopyrimidine Efficiently
The execution of this synthesis requires careful control of temperature and stoichiometry to maximize yield and minimize side reactions. The process is divided into four distinct operational stages, beginning with the condensation in dioxane and concluding with the chlorination in toluene. Each step has been optimized to allow for straightforward work-up procedures such as aqueous washing and solvent swapping, avoiding the need for silica gel chromatography. For a detailed breakdown of the specific reaction conditions, molar ratios, and isolation techniques, please refer to the standardized protocol below.
- Condensation of cyanoacetic acid with DMF-DMA in dioxane to form 3-(dimethylamino)acrylonitrile.
- Vilsmeier-Haack formylation using DMF and oxalyl chloride to generate the reactive intermediate.
- Cyclization with O-methoxyisourea sulfate in acetonitrile/pyridine to form the pyrimidine ring.
- Chlorination using phosphorus pentachloride (PCl5) to yield the final 2-chloro-5-cyanopyrimidine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the shift to this cyanoacetic acid-based route offers profound advantages for supply chain stability and cost management. The primary driver of value is the substitution of high-cost, volatile raw materials with commodity chemicals. Cyanoacetic acid, DMF-DMA, and phosphorus pentachloride are widely available bulk chemicals with stable pricing structures, unlike palladium catalysts or specialized brominated heterocycles. This fundamental change in the bill of materials (BOM) decouples the production cost from the fluctuations of the precious metals market. Furthermore, the elimination of column chromatography is a massive operational win. Chromatography is a major bottleneck in fine chemical manufacturing, consuming vast amounts of solvents and silica while limiting batch sizes. By replacing this with crystallization and pulping, the process becomes inherently more scalable and environmentally friendly.
- Cost Reduction in Manufacturing: The most immediate impact is seen in the raw material costs. By removing the dependency on palladium catalysts and expensive brominated starting materials, the direct material cost is drastically reduced. Additionally, the avoidance of column chromatography significantly lowers the consumption of solvents and stationary phases, which are often hidden cost drivers in intermediate synthesis. The simplified post-treatment also reduces labor hours and equipment occupancy time, leading to a lower overall conversion cost per kilogram. This efficiency allows for a more competitive pricing structure for the final API, enhancing the market viability of the end drug product.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available reagents. Sourcing cyanoacetic acid and common solvents like dioxane and toluene is far less risky than sourcing specialized catalysts or toxic gases like phosgene. This reduces the lead time for raw material procurement and minimizes the risk of production stoppages due to supply shortages. Moreover, the robustness of the reaction conditions—operating at moderate temperatures and pressures—means that the process can be easily transferred between different manufacturing sites or CDMO partners without requiring specialized infrastructure, ensuring continuity of supply.
- Scalability and Environmental Compliance: The environmental footprint of this process is significantly smaller than that of the prior art. The absence of heavy metals like copper and palladium simplifies wastewater treatment and reduces the burden of hazardous waste disposal. The elimination of phosgene removes a major safety hazard, lowering insurance premiums and regulatory compliance costs. The process is designed for scalability, with reaction steps that exotherm safely and can be managed in standard glass-lined or stainless steel reactors. This makes the technology ideal for commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to ramp up production volumes rapidly to meet market demand without compromising on safety or quality standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this method is preferred for industrial applications.
Q: Why is the cyanoacetic acid route superior to palladium-catalyzed methods?
A: The cyanoacetic acid route eliminates the need for expensive palladium catalysts, which are subject to volatile market pricing and require complex removal processes to meet heavy metal specifications in pharmaceuticals.
Q: Does this process involve hazardous reagents like phosgene?
A: No, unlike previous methods that utilized toxic phosgene or its equivalents, this novel pathway relies on safer reagents like oxalyl chloride and phosphorus pentachloride under controlled conditions, significantly improving operational safety.
Q: Is column chromatography required for purification?
A: No, a key advantage of this method is that none of the reaction steps require column chromatography. Purification is achieved through standard work-up procedures like washing, crystallization, and pulping, making it highly suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-5-Cyanopyrimidine Supplier
The technological advancements outlined in CN110845426A underscore the potential for high-efficiency manufacturing of critical oncology intermediates. At NINGBO INNO PHARMCHEM, we specialize in translating such innovative patent technologies into commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific reagents and conditions required for this cyanoacetic acid route, ensuring that your supply of 2-chloro-5-cyanopyrimidine is consistent and reliable. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical grade intermediates.
We invite you to leverage our technical expertise to optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this novel synthesis can drive value for your organization. Let us collaborate to secure a sustainable and cost-effective supply of this vital building block for your cancer therapy pipeline.
