Advanced Recovery Technology for Ursodeoxycholic Acid and Chenodeoxycholic Acid from Waste Streams
The pharmaceutical industry constantly faces the dual challenge of optimizing synthesis yields for critical active pharmaceutical ingredients while managing the environmental impact of chemical waste. Patent CN102766185A introduces a groundbreaking methodology specifically designed to address the inefficiencies in the production of bile acid derivatives. This technology focuses on the recovery of ursodesoxycholic acid (UDCA) and chenodeoxycholic acid (CDCA) from waste mother liquors generated during the purification of UDCA. Traditionally, these waste streams are treated as hazardous byproducts, yet they contain substantial quantities of valuable isomers that are chemically distinct and therapeutically significant. The structural relationship between these two compounds is fundamental to understanding the separation challenge, as they differ only in the stereochemistry at the 7-position hydroxyl group.
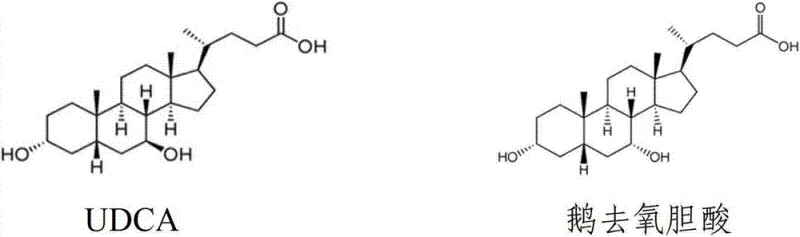
By implementing this recovery strategy, manufacturers can drastically improve the overall atom economy of their synthesis campaigns. The patent outlines a robust chemical process that leverages the subtle physicochemical differences between these isomers to achieve high-purity separation. For a reliable pharmaceutical intermediates supplier, adopting such circular economy principles is not merely an environmental gesture but a strategic imperative for cost reduction in pharmaceutical manufacturing. The ability to reclaim high-value starting materials or co-products from waste streams directly impacts the bottom line by reducing the net consumption of raw materials and minimizing waste disposal fees associated with regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the conventional synthesis of ursodesoxycholic acid, the final purification steps often generate significant volumes of mother liquor that are rich in unreacted starting materials and isomeric byproducts. Historically, the industry standard has been to treat this liquor as waste, subjecting it to incineration or chemical neutralization before discharge. This approach represents a massive loss of potential value, as the waste liquor typically contains a complex mixture of ursodesoxycholic acid and chenodeoxycholic acid that requires minimal processing to become saleable product. Furthermore, the disposal of organic-rich aqueous waste imposes a heavy burden on wastewater treatment facilities and increases the carbon footprint of the manufacturing site. The lack of a dedicated recovery protocol means that yield losses accumulate silently, eroding profit margins without immediate visibility to the procurement team.
The Novel Approach
The novel approach detailed in the patent circumvents these issues by introducing a selective precipitation technique based on ammonium salt formation. Instead of discarding the waste, the process first dissolves the mother liquor in an inorganic base to solubilize the acidic components, followed by filtration to remove insoluble impurities and color bodies. Subsequent acidification precipitates a mixed solid containing both target acids. The breakthrough lies in the next step: dissolving this mixed solid in an organic solvent and treating it with a specific organic amine. This reaction creates ammonium salts with distinct solubility profiles. Upon controlled cooling, the ursodeoxycholic acid ammonium salt crystallizes out selectively, leaving the chenodeoxycholic acid ammonium salt in the supernatant. This elegant separation avoids the need for expensive preparative chromatography at the bulk stage, offering a streamlined path to high-purity isolation.
Mechanistic Insights into Selective Ammonium Salt Precipitation
The core of this technology relies on the precise manipulation of solubility equilibria through salt formation. When an organic amine reacts with the carboxylic acid group of the bile acid, it forms an ionic ammonium carboxylate salt. The crystal lattice energy and solvation properties of these salts are heavily influenced by the stereochemistry of the steroid backbone. In the specific solvent systems described, such as sec-butyl alcohol or ethyl acetate, the 7-beta-hydroxy configuration of the UDCA ammonium salt promotes tighter packing or lower solubility at reduced temperatures compared to the 7-alpha-hydroxy configuration of the CDCA salt. This differential solubility is the driving force for the separation.
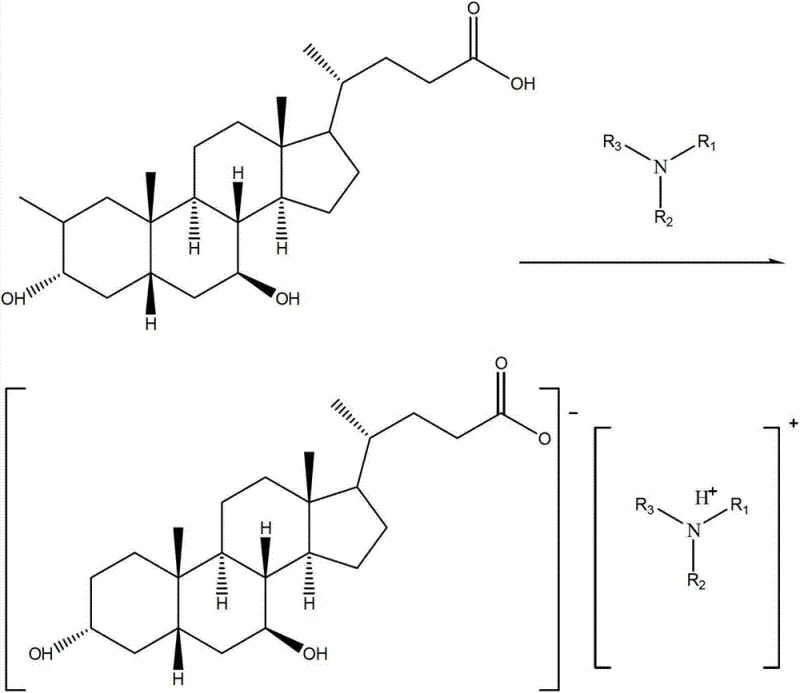
Furthermore, the process incorporates a rigorous impurity control mechanism through the initial alkaline dissolution and membrane filtration steps. By utilizing ultrafiltration or nanofiltration membranes with specific molecular weight cut-offs, high molecular weight polymeric impurities and colored degradation products are effectively removed before the critical separation step. This ensures that the subsequent crystallization of the ammonium salt occurs from a clean matrix, preventing occlusion of impurities within the crystal lattice. The final acidification step regenerates the free acid form, yielding a product that meets stringent purity specifications suitable for further pharmaceutical processing or direct use as a high-purity pharmaceutical intermediate.
How to Synthesize Ursodeoxycholic Acid Efficiently
The synthesis protocol described offers a practical roadmap for converting waste streams into valuable assets. The process begins with the alkaline dissolution of the waste mother liquor, followed by acidification to isolate the total cholic acid fraction. This crude mixture is then subjected to the selective ammonium salt precipitation in an organic medium. The key to success lies in the careful selection of the organic amine and the control of the cooling rate to maximize the yield of the precipitated UDCA salt. Detailed standardized synthetic steps see the guide below.
- Dissolve the waste mother liquor in an inorganic base solution, filter to remove impurities, and then acidify with mineral acid to obtain a mixed precipitate of ursodesoxycholic acid and chenodeoxycholic acid.
- Dissolve the mixed precipitate in an organic solvent, add a specific organic amine, and reflux followed by cooling to selectively precipitate ursodeoxycholic acid ammonium salt while keeping chenodeoxycholic acid ammonium salt in solution.
- Separate the solids and filtrate, then acidify each fraction independently to recover high-purity ursodesoxycholic acid and chenodeoxycholic acid respectively.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the implementation of this recovery technology translates into tangible strategic benefits beyond simple yield improvement. The ability to source high-quality bile acids from internal waste streams reduces dependency on external raw material suppliers, thereby insulating the production schedule from market volatility and supply disruptions. This vertical integration of material flow enhances the overall resilience of the supply chain, ensuring consistent availability of critical intermediates for downstream drug formulation.
- Cost Reduction in Manufacturing: The most immediate impact is the drastic reduction in raw material costs. By recovering ursodesoxycholic acid and chenodeoxycholic acid that would otherwise be lost, the effective cost per kilogram of the final product decreases significantly. Additionally, the reagents used in this process, such as sodium hydroxide, hydrochloric acid, and common organic amines, are commodity chemicals with stable pricing and wide availability. Eliminating the need for expensive transition metal catalysts or specialized chiral resolving agents further drives down the operational expenditure, resulting in substantial cost savings without compromising product quality.
- Enhanced Supply Chain Reliability: Relying on waste recovery creates a secondary internal source of supply that complements primary synthesis routes. This diversification mitigates the risk of shortages caused by upstream bottlenecks or geopolitical factors affecting raw material imports. Since the feedstock is generated onsite during the normal course of production, the lead time for obtaining these recovered intermediates is virtually eliminated. This reliability allows for more accurate production planning and inventory management, reducing the need for safety stock and freeing up working capital.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as filtration, crystallization, and distillation that are standard in fine chemical manufacturing. The mild reaction conditions, typically ranging from ambient temperature to moderate heating, reduce energy consumption and equipment wear. From an environmental perspective, converting hazardous waste into non-hazardous, high-value products significantly lowers the volume of waste requiring treatment. This alignment with green chemistry principles simplifies regulatory compliance and enhances the corporate sustainability profile, which is increasingly important for maintaining relationships with global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recovery technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of adoption in your own manufacturing facilities.
Q: What is the primary advantage of this recovery method over traditional disposal?
A: The primary advantage is the transformation of hazardous waste into high-value pharmaceutical intermediates. Traditional methods often discard waste mother liquor containing significant amounts of active ingredients, leading to environmental pollution and economic loss. This patented process recovers both ursodesoxycholic acid and chenodeoxycholic acid with high efficiency, turning a cost center into a revenue stream while reducing environmental compliance burdens.
Q: How does the process achieve separation of the two isomers?
A: The separation relies on the differential solubility of their respective ammonium salts in specific organic solvents. By carefully selecting the organic amine and solvent system, ursodeoxycholic acid ammonium salt is induced to crystallize out of the solution upon cooling, whereas the chenodeoxycholic acid ammonium salt remains dissolved. This physical property difference allows for a clean separation without requiring complex chromatographic steps at the initial stage.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for industrial scalability. It utilizes common, low-cost reagents such as sodium hydroxide, hydrochloric acid, and standard organic solvents like acetone or ethyl acetate. The reaction conditions are mild, typically operating between 15°C and 70°C, which minimizes energy consumption and equipment stress, making it highly feasible for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ursodeoxycholic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex recovery processes like the one described in CN102766185A can be seamlessly integrated into your supply chain. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and potency of every batch.
We invite you to explore how our advanced recovery capabilities can optimize your production economics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific waste stream profile. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can become your long-term partner in sustainable chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
