Advanced Synthesis of Epoxy Skeleton Nitrile Compounds for Pharmaceutical Intermediates
Advanced Synthesis of Epoxy Skeleton Nitrile Compounds for Pharmaceutical Intermediates
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of the pharmaceutical and fine chemical industries, particularly regarding the efficient production of heterocyclic building blocks. A significant breakthrough in this domain is detailed in patent CN108250164B, which discloses a novel class of nitrile compounds containing an epoxy skeleton and a robust synthetic methodology for their preparation. This technology addresses the critical need for reliable access to functionalized aryloxiranes, which serve as versatile precursors for biologically active molecules. By leveraging a creative optimization of reaction parameters, including specific palladium catalysts and oxidizing agents, this invention provides a brand-new synthesis route that offers superior control over product purity and yield compared to legacy methods. For R&D teams seeking to streamline their pipeline for furan-based drug candidates, this methodology represents a substantial advancement in process chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of furan compounds and their precursors has relied on complex multi-step sequences that often suffer from poor atom economy and harsh reaction conditions. Prior art, such as the methods disclosed in CN101148442A and CN105198841A, typically involves intricate cyclization strategies or the use of unstable intermediates that complicate purification and reduce overall throughput. Traditional approaches frequently require stoichiometric amounts of heavy metal reagents or extreme temperatures that degrade sensitive functional groups, leading to broad impurity profiles that are difficult to manage during scale-up. Furthermore, many existing routes lack modularity, making it challenging to introduce diverse substituents on the aromatic ring without redesigning the entire synthetic pathway. These limitations create significant bottlenecks for procurement managers who struggle with the high cost of goods and supply chain volatility associated with specialized, low-yield intermediates.
The Novel Approach
In stark contrast, the methodology described in the present invention introduces a highly efficient two-step strategy that transforms simple, commercially available starting materials into valuable epoxy skeleton nitrile compounds. The core innovation lies in the sequential application of a palladium-catalyzed oxidative coupling followed by a selective epoxidation. As illustrated in the reaction scheme below, the process begins with the coupling of an acrylonitrile derivative and an aryl boronic acid, proceeding through a cinnamonitrile intermediate before final cyclization to the epoxide. This route eliminates the need for pre-functionalized halides often required in cross-coupling, utilizing instead the more stable and diverse boronic acid partners. The result is a streamlined workflow that minimizes waste generation and maximizes the structural diversity accessible to medicinal chemists, thereby offering a distinct competitive advantage in the manufacturing of complex pharmaceutical intermediates.
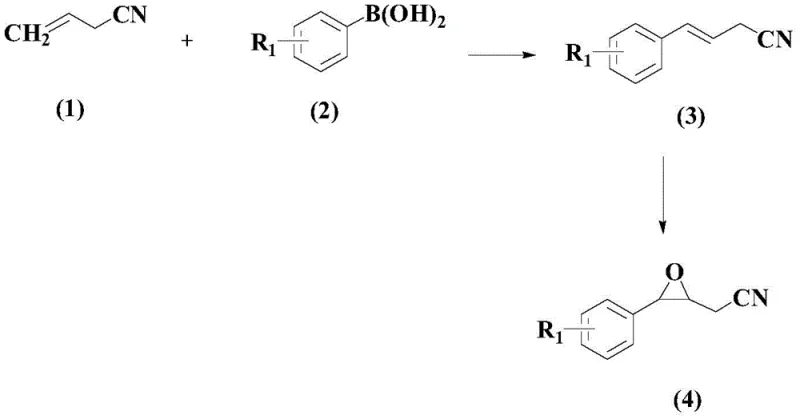
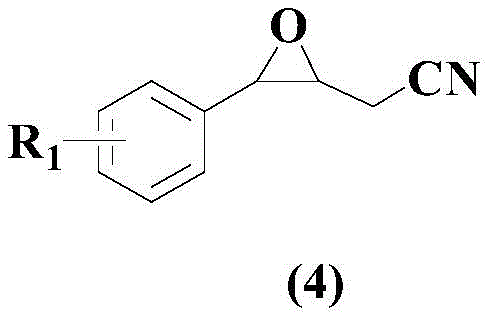
The structural integrity of the final product is paramount for downstream applications, and the novel compound represented by Formula (4) exhibits exceptional stability and reactivity profiles. This specific architecture, featuring a nitrile group adjacent to an oxirane ring on an aromatic scaffold, serves as a privileged motif for further derivatization. The ability to precisely tune the electronic properties of the aromatic ring through the selection of different boronic acid inputs allows for the rapid generation of analog libraries. For supply chain leaders, this modularity translates to a flexible manufacturing platform where a single set of optimized reaction conditions can accommodate a wide range of substrates, reducing the need for extensive process re-validation for each new variant. Consequently, this approach not only enhances the speed of discovery but also solidifies the reliability of the supply chain for high-purity specialty chemicals.
Mechanistic Insights into Pd-Catalyzed Oxidative Coupling and Epoxidation
The success of this synthetic route hinges on the meticulous selection of catalytic systems and reaction conditions that drive the transformation with high fidelity. In the first step (S1), the reaction proceeds via a palladium-catalyzed oxidative Heck-type coupling mechanism. The use of palladium acetate (Pd(OAc)2) in conjunction with 2,2'-bipyridine as a ligand creates a highly active catalytic species capable of activating the C-H bond of the acrylonitrile derivative. Crucially, the presence of silver acetate as a stoichiometric oxidant facilitates the regeneration of the active Pd(II) species, while trifluoroacetic acid (TFA) plays a dual role in protonating intermediates and maintaining the acidity required for the catalytic cycle. Experimental data indicates that deviating from this specific combination, such as substituting Pd(OAc)2 with PdCl2 or replacing TFA with acetic acid, results in a dramatic decrease in yield, underscoring the non-obvious nature of this optimized system. The reaction is conducted in tetrahydrofuran (THF) at temperatures between 70°C and 100°C, ensuring complete conversion while minimizing side reactions.
Following the isolation of the cinnamonitrile intermediate, the second step (S2) involves a stereoselective epoxidation to install the oxygen heterocycle. This transformation is achieved using meta-chloroperoxybenzoic acid (m-CPBA) in dichloromethane at mild temperatures ranging from 20°C to 40°C. The choice of m-CPBA is critical, as comparative studies show that other oxidants like tert-butyl hydroperoxide (TBHP) or potassium persulfate fail to deliver comparable yields or selectivity. The mild conditions of this step preserve the integrity of the nitrile group and the aromatic system, preventing decomposition or polymerization. This mechanistic precision ensures that the final epoxy skeleton nitrile compound is obtained with high chemical purity, reducing the burden on downstream purification processes. For technical teams, understanding these mechanistic nuances is essential for troubleshooting and optimizing the process for commercial scale-up, ensuring consistent quality across large batches.
How to Synthesize Epoxy Skeleton Nitrile Compounds Efficiently
The practical implementation of this synthesis requires strict adherence to the optimized parameters identified during the development phase to ensure reproducibility and safety. The process is designed to be operationally simple, utilizing standard laboratory equipment and readily available reagents, which facilitates a smooth transition from bench-scale experimentation to pilot plant production. Operators should focus on maintaining the precise molar ratios of the catalyst, ligand, and oxidant, as slight deviations can impact the reaction kinetics and final yield. The workup procedures involve standard aqueous extractions and flash column chromatography, techniques that are well-established in industrial settings. Below is a summary of the standardized protocol derived from the patent examples, which serves as a guideline for producing these high-value intermediates.
- Perform a palladium-catalyzed coupling reaction between an acrylonitrile derivative and an aryl boronic acid using Pd(OAc)2, silver acetate, and trifluoroacetic acid in THF at 70-100°C.
- Isolate the intermediate cinnamonitrile derivative via standard aqueous workup and flash column chromatography.
- Conduct an epoxidation reaction on the isolated intermediate using m-CPBA in dichloromethane at 20-40°C to yield the final epoxy skeleton nitrile compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers transformative benefits for organizations focused on cost efficiency and supply chain resilience. By shifting away from legacy methods that rely on scarce or expensive reagents, manufacturers can achieve significant reductions in raw material costs and processing time. The use of commodity chemicals such as acrylonitrile derivatives and aryl boronic acids ensures a stable supply base, mitigating the risks associated with sourcing specialized precursors. Furthermore, the high yields reported in the patent examples demonstrate a robust process that minimizes material loss, directly contributing to improved margin structures. For procurement managers, this translates to a more predictable cost model and the ability to negotiate better terms with suppliers due to the increased flexibility in raw material selection.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences and the use of a highly efficient catalytic system drastically simplify the production workflow. By avoiding the need for expensive transition metal catalysts that require rigorous removal steps, the process reduces both material costs and waste disposal expenses. The high atom economy of the coupling reaction ensures that a greater proportion of the starting materials are converted into the desired product, lowering the effective cost per kilogram. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling, further enhancing the overall economic viability of the manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as substituted boronic acids and acrylonitriles ensures a continuous and stable supply chain. Unlike proprietary intermediates that may be subject to single-source bottlenecks, the reagents used in this process are produced by multiple global suppliers, reducing the risk of disruption. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, allowing for greater flexibility in vendor qualification. This reliability is crucial for maintaining uninterrupted production schedules and meeting the demanding delivery timelines of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing solvents like THF and dichloromethane that are standard in industrial organic synthesis and can be efficiently recovered and recycled. The absence of hazardous high-pressure steps or extremely low-temperature requirements simplifies the engineering controls needed for large-scale production. Moreover, the high selectivity of the reaction minimizes the formation of by-products, reducing the load on wastewater treatment facilities and aligning with increasingly stringent environmental regulations. This commitment to green chemistry principles not only lowers compliance costs but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these epoxy skeleton nitrile compounds. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and advantages of this technology. Understanding these details is essential for stakeholders evaluating the potential integration of this route into their existing manufacturing portfolios.
Q: What are the critical catalyst conditions for the initial coupling step?
A: The process requires palladium acetate (Pd(OAc)2) as the optimal catalyst, paired with 2,2'-bipyridine as a ligand and silver acetate as the oxidant in a THF solvent system to achieve yields exceeding 80%.
Q: How does this method improve upon traditional furan synthesis routes?
A: Unlike conventional multi-step pathways, this novel approach utilizes a streamlined two-step sequence starting from readily available acrylonitriles and boronic acids, significantly simplifying the supply chain for downstream furan production.
Q: Is the epoxidation step scalable for industrial production?
A: Yes, the epoxidation utilizes m-CPBA in dichloromethane under mild temperatures (20-40°C), avoiding hazardous high-pressure conditions and allowing for straightforward scale-up with standard safety protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epoxy Skeleton Nitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates that accelerate drug discovery and development. Our team of expert chemists has extensively evaluated the technology described in CN108250164B and confirmed its potential for delivering superior results in the synthesis of complex pharmaceutical building blocks. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet stringent purity specifications. Our state-of-the-art rigorous QC labs are equipped to verify the identity and purity of every batch, guaranteeing consistency and reliability for your most demanding projects.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical intermediates can drive value and efficiency in your supply chain.
