Revolutionizing Bipyridine Derivative Synthesis: A Commercial Scale-Up Perspective on Asymmetric Functionalization
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and selective synthetic routes for complex pharmaceutical intermediates. A significant breakthrough in this domain is documented in patent CN108484493B, which details a novel bifunctional group conversion method for 3,3'-dinitro-2,2'-bipyridine derivatives. This technology represents a paradigm shift from conventional symmetric functionalization strategies, enabling the direct transformation of dinitro precursors into valuable 3-amino-3'-hydroxy-2,2'-bipyridine derivatives. For R&D directors and procurement specialists alike, this innovation addresses critical bottlenecks in the synthesis of nitrogen-containing ligands and bioactive scaffolds. By leveraging a copper-mediated mechanism under aerobic conditions, the process eliminates the need for expensive noble metal catalysts or harsh reducing environments, thereby offering a robust pathway for the production of high-purity fine chemicals. The ability to access asymmetric substitution patterns on the bipyridine core opens new avenues for drug discovery and material science applications, making this patent a cornerstone for modern supply chain optimization in the specialty chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical modification of 3,3'-dinitro-2,2'-bipyridine has been plagued by a lack of selectivity, forcing chemists into multi-step sequences or accepting mixtures of symmetric products. Traditional reduction protocols, such as those utilizing tin(II) chloride in hydrochloric acid, invariably reduce both nitro groups simultaneously to yield the corresponding diamino derivatives, as illustrated in the upper reaction pathway of the provided schematic. Alternatively, other reductive conditions employing sodium sulfide tend to promote cyclization or azo-coupling reactions, leading to structurally distinct byproducts like phenazine derivatives rather than the desired amino-hydroxy motif. These conventional pathways not only limit the structural diversity accessible to medicinal chemists but also introduce significant purification challenges due to the formation of closely related impurities. Furthermore, the reliance on stoichiometric amounts of heavy metal reductants or strongly acidic conditions generates substantial waste streams, complicating environmental compliance and increasing the overall cost of goods sold. The inability to differentiate between the two nitro groups in a single operational step has long been a persistent obstacle in the efficient manufacturing of asymmetric bipyridine ligands.
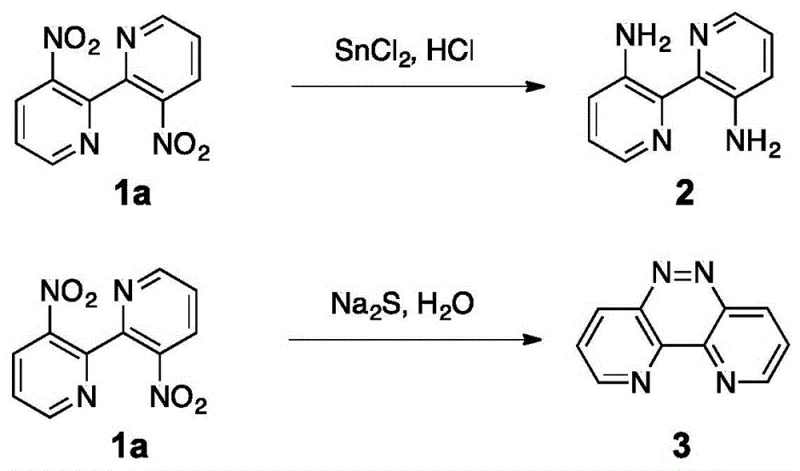
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN108484493B introduces a highly selective bifunctional conversion strategy that breaks the symmetry of the starting material with remarkable precision. By employing a specific copper reagent in a polar aprotic solvent under air atmosphere, the process facilitates the simultaneous reduction of one nitro group to an amine and the substitution of the other to a hydroxyl group. This divergent reactivity is achieved without the need for protecting groups or sequential addition of reagents, effectively collapsing what would traditionally be a multi-step synthesis into a single pot operation. The reaction conditions are notably mild yet effective, operating at temperatures between 100°C and 150°C, which ensures thermal stability of the sensitive bipyridine core while driving the conversion to completion. This novel approach not only streamlines the synthetic workflow but also drastically improves the atom economy of the process by minimizing the formation of symmetric byproducts. For industrial partners, this translates to a simplified manufacturing protocol that reduces operator intervention and enhances batch-to-batch consistency, crucial factors for maintaining supply chain reliability in the production of critical pharmaceutical intermediates.
Mechanistic Insights into Copper-Catalyzed Bifunctional Conversion
The underlying mechanism of this transformation involves a sophisticated interplay between the copper species and the nitro functionalities, mediated by the solvent and atmospheric oxygen. The copper reagent, likely acting as a Lewis acid and a redox mediator, coordinates with the nitrogen atoms of the bipyridine system, activating one nitro group for nucleophilic attack by solvent-derived hydroxide equivalents while simultaneously facilitating the electron transfer required for the reduction of the second nitro group to an amine. This dual activation mode is critical for achieving the observed regioselectivity, ensuring that the two nitro groups undergo distinct chemical fates despite their structural equivalence in the starting material. The presence of air is not merely incidental but plays a vital role in regenerating the active catalytic species and preventing the over-reduction of the intermediate species to the diamino compound. Understanding this mechanistic nuance is essential for R&D teams aiming to optimize the process further, as slight variations in oxygen availability or copper oxidation state could influence the ratio of amino to hydroxy products. The robustness of this mechanism across various substituents on the bipyridine ring suggests a broad substrate scope, allowing for the synthesis of diverse derivatives without compromising the integrity of the core transformation.
From an impurity control perspective, the specificity of this copper-mediated pathway offers significant advantages over non-catalytic reduction methods. The reaction profile is characterized by the formation of a single major product, with minimal generation of the symmetric diamino or azo-linked impurities that typically plague conventional syntheses. This high level of chemoselectivity simplifies the downstream purification process, often allowing for isolation of the target compound through standard extraction and chromatography techniques without the need for complex recrystallization sequences. For quality assurance teams, this means a cleaner crude profile and a higher likelihood of meeting stringent purity specifications required for GMP manufacturing. The ability to suppress side reactions also enhances the overall yield of the process, maximizing the utility of the starting materials and reducing the volume of waste solvents generated per kilogram of product. Such control over the impurity profile is a key determinant in the regulatory acceptance of new synthetic routes for active pharmaceutical ingredients and their advanced intermediates.
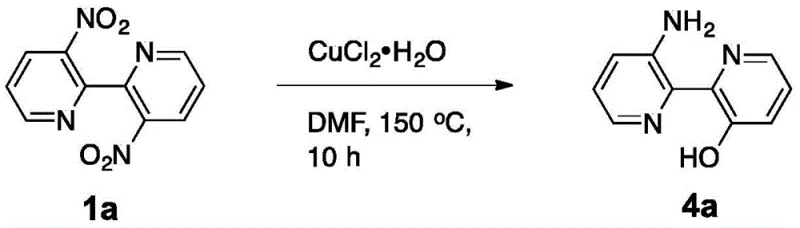
How to Synthesize 3-Amino-3-Hydroxy-2,2-Bipyridine Efficiently
The practical implementation of this synthesis involves a straightforward procedure that balances reaction efficiency with operational simplicity, making it highly suitable for translation from laboratory to pilot plant scales. The process begins with the suspension of the 3,3'-dinitro-2,2'-bipyridine derivative in a high-boiling polar solvent such as N,N-Dimethylformamide (DMF), followed by the addition of a hydrated copper salt like copper(II) chloride dihydrate. The reaction mixture is then heated under an open air atmosphere, eliminating the need for costly inert gas purging systems, and maintained at elevated temperatures for a duration sufficient to drive the bifunctional conversion to completion. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining 3,3-dinitro-2,2-bipyridine derivative with a copper reagent such as copper chloride dihydrate in a polar aprotic solvent like DMF.
- Heat the reaction mixture under air atmosphere at temperatures between 100°C and 150°C for approximately 10 hours to facilitate bifunctional conversion.
- Perform post-reaction workup involving aqueous extraction, solvent removal, and column chromatography to isolate the pure 3-amino-3-hydroxy product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers compelling economic and logistical benefits that extend beyond mere chemical elegance. The elimination of complex multi-step sequences directly correlates to a reduction in manufacturing cycle time, allowing for faster turnaround on custom synthesis orders and improved responsiveness to market demand fluctuations. Furthermore, the use of commodity-grade copper reagents instead of precious metal catalysts significantly lowers the raw material cost base, providing a buffer against volatility in the pricing of specialized chemical inputs. The operational simplicity of running the reaction under air conditions also reduces the capital expenditure required for specialized reactor infrastructure, making it easier for contract manufacturing organizations to adopt the technology without major retrofitting. These factors combine to create a more resilient supply chain capable of delivering high-value intermediates at a competitive price point, ensuring continuity of supply for downstream pharmaceutical production.
- Cost Reduction in Manufacturing: The economic advantage of this process is primarily driven by the substitution of expensive noble metal catalysts with abundant and inexpensive copper salts, which drastically reduces the direct material cost per kilogram of product. Additionally, the convergence of two functional group transformations into a single reaction step eliminates the need for intermediate isolation and purification, thereby saving on solvent consumption, labor hours, and energy usage associated with multiple unit operations. The simplified workup procedure, which relies on standard extraction and chromatography, further minimizes waste disposal costs and reduces the burden on environmental health and safety departments. Collectively, these efficiencies result in a substantially lower cost of goods sold, enabling more aggressive pricing strategies in the competitive fine chemical market without sacrificing margin.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the reliance on readily available starting materials and reagents mitigates the risk of supply disruptions that often accompany specialized or proprietary catalysts. The robustness of the reaction under aerobic conditions means that production is less susceptible to delays caused by inert gas supply issues or equipment failures related to pressure systems. This operational resilience ensures a more predictable production schedule, allowing supply chain planners to maintain lower safety stock levels while still meeting delivery commitments. The scalability of the process from gram to multi-kilogram batches further supports a flexible manufacturing model that can adapt quickly to changes in order volume, ensuring that customers receive their materials on time regardless of market fluctuations.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly smaller than traditional methods due to the avoidance of stoichiometric heavy metal waste and the reduction in solvent intensity through step economy. The use of copper, which is easier to recover and recycle than many other transition metals, aligns with increasingly stringent global regulations regarding heavy metal discharge in pharmaceutical manufacturing. Moreover, the high selectivity of the reaction reduces the generation of hazardous byproducts, simplifying the treatment of effluent streams and lowering the cost of environmental compliance. This green chemistry profile not only enhances the corporate sustainability credentials of the manufacturer but also future-proofs the supply chain against tightening environmental legislation, ensuring long-term viability of the production route.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this bifunctional conversion technology, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, ensuring accuracy and relevance for decision-makers. Understanding these nuances is critical for assessing the feasibility of integrating this route into existing manufacturing portfolios.
Q: What distinguishes this copper-catalyzed method from traditional reduction techniques?
A: Unlike traditional methods that reduce both nitro groups to amines or form azo compounds, this novel approach achieves asymmetric functionalization, yielding one amino and one hydroxy group simultaneously in a single step.
Q: Is inert gas protection required for this synthesis?
A: No, the process operates effectively under air conditions, which significantly simplifies the operational requirements and reduces equipment costs compared to inert atmosphere reactions.
Q: What are the scalability prospects for this bipyridine derivative synthesis?
A: The method utilizes cheap and readily available raw materials with a simple workup procedure, demonstrating strong potential for commercial scale-up from gram to multi-kilogram production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Amino-3-Hydroxy-2,2-Bipyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of robust and scalable synthetic routes for complex heterocyclic intermediates like 3-amino-3-hydroxy-2,2'-bipyridine derivatives. Our CDMO team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and assay. Our infrastructure is designed to handle the specific solvent and thermal requirements of this copper-catalyzed process, guaranteeing consistent quality and supply continuity for our global partners.
We invite pharmaceutical and agrochemical companies to leverage our expertise to optimize their supply chains and reduce development timelines. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that evaluates the specific economic impact of switching to this novel synthesis route for your projects. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your unique molecular requirements. Let us help you transform this innovative chemistry into a commercial reality, driving efficiency and value across your entire product lifecycle.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
