Advanced Synthesis of Non-Coplanar Benzimidazole Diamines for High-Performance Polymers
Advanced Synthesis of Non-Coplanar Benzimidazole Diamines for High-Performance Polymers
The development of advanced polymeric materials often hinges on the precise molecular architecture of their monomeric building blocks. In the realm of high-performance polymers such as polyimides and polyamides, the rigidity and planarity of traditional aromatic diamines frequently lead to materials with excellent thermal stability but poor optical properties and limited solubility. Addressing this critical bottleneck, patent CN112608279A introduces a groundbreaking class of non-coplanar benzimidazole diamines. This technology leverages steric hindrance to twist the molecular backbone, effectively disrupting conjugation and unlocking superior optical transparency and processability. As a leading entity in fine chemical synthesis, we recognize this patent as a pivotal advancement for the electronic chemical manufacturing sector, offering a robust pathway to next-generation optoelectronic materials.
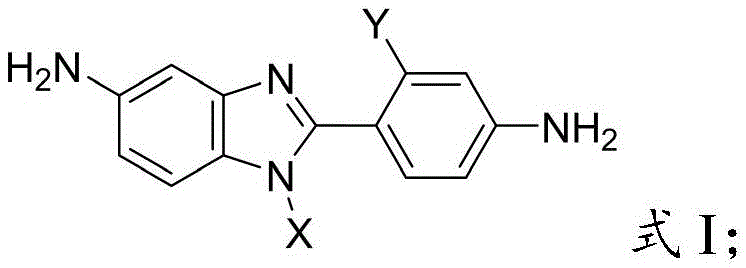
The core innovation lies in the structural design depicted in Formula I, where specific side groups are strategically introduced on both the benzimidazole ring and the pendant phenyl ring. This dual-substitution strategy is not merely a cosmetic change; it is a fundamental engineering of molecular geometry. By forcing the benzimidazole and phenyl rings out of a single plane, the synthesis creates a 'twisted' conformation. This non-coplanar geometry reduces the intermolecular charge transfer complexes that typically cause coloration in polyimides, thereby yielding polymers that are lighter in color and more transparent. For R&D directors seeking to enhance the performance of display substrates or flexible electronics, this structural motif offers a compelling solution to the age-old trade-off between thermal stability and optical clarity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzimidazole-based monomers has relied on straightforward condensation reactions that often result in highly planar, rigid structures. While these conventional monomers impart exceptional mechanical strength and thermal resistance to the resulting polymers, they suffer from significant drawbacks in modern applications. The extensive pi-conjugation in planar systems leads to strong intermolecular interactions, which drastically reduce solubility in common organic solvents. This poor solubility complicates processing, often requiring hazardous high-boiling solvents or extreme temperatures for film casting. Furthermore, the strong charge-transfer interactions inherent in these flat molecules result in deep yellow or brown coloration, rendering them unsuitable for applications requiring high optical transmittance, such as flexible display cover windows or transparent conductive films.
The Novel Approach
The methodology outlined in the patent data presents a sophisticated departure from these traditional limitations by incorporating bulky substituents directly into the monomer scaffold. As illustrated by the variable groups X and Y, the synthesis allows for the introduction of diverse alkyl, aryl, or halogenated groups. 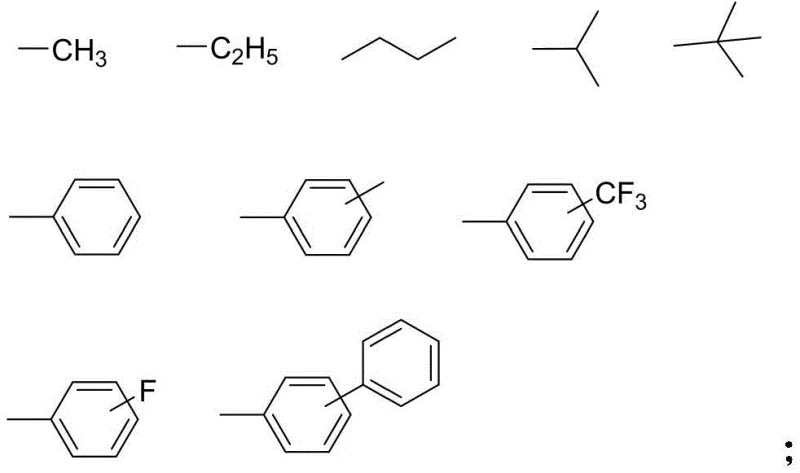
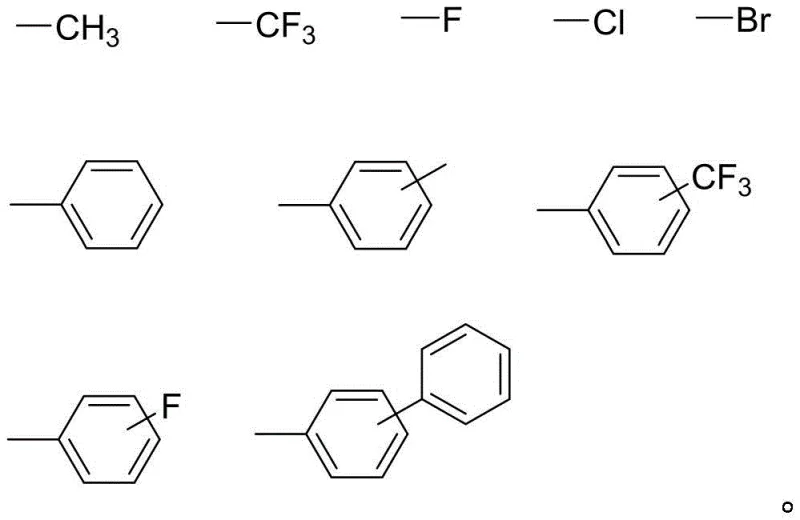 This structural diversity enables fine-tuning of the dihedral angle between the rings. The novel approach ensures that the resulting diamine monomers possess a distorted, three-dimensional shape. This distortion effectively shields the conjugated system, minimizing color generation while maintaining the inherent thermal stability of the benzimidazole heterocycle. Consequently, polymers derived from these monomers exhibit a rare combination of high glass transition temperatures, excellent solubility for solution processing, and superior optical properties, meeting the rigorous demands of the semiconductor and display industries.
This structural diversity enables fine-tuning of the dihedral angle between the rings. The novel approach ensures that the resulting diamine monomers possess a distorted, three-dimensional shape. This distortion effectively shields the conjugated system, minimizing color generation while maintaining the inherent thermal stability of the benzimidazole heterocycle. Consequently, polymers derived from these monomers exhibit a rare combination of high glass transition temperatures, excellent solubility for solution processing, and superior optical properties, meeting the rigorous demands of the semiconductor and display industries.
Mechanistic Insights into Condensation and Cyclization Chemistry
The synthesis pathway described in the patent is a masterclass in efficient organic transformation, proceeding through a logical sequence of condensation, cyclization, and reduction. The initial step involves the acylation of a nitro-substituted phenylenediamine with a substituted benzoyl chloride. This condensation reaction is meticulously controlled at low temperatures, typically between 0-25°C, using acid-binding agents like triethylamine or pyridine to scavenge the generated hydrochloric acid. This mild condition is crucial for preventing side reactions and ensuring high selectivity. Following the formation of the amide intermediate, the process moves to the ring-closing step. This is the thermodynamic driver of the benzimidazole formation, requiring elevated temperatures of 180-220°C in high-boiling polar aprotic solvents such as N-methyl pyrrolidone (NMP) or sulfolane. The use of acidic catalysts like p-toluenesulfonic acid facilitates the dehydration cyclization, closing the imidazole ring with high efficiency.
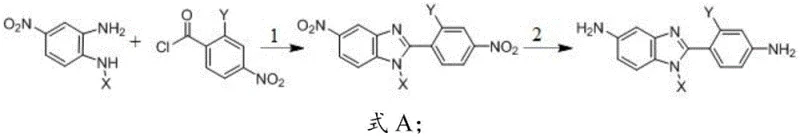
The final and perhaps most critical step for achieving the diamine functionality is the catalytic hydrogenation. The patent specifies the use of robust heterogeneous catalysts such as palladium on carbon (Pd/C), platinum on carbon (Pt/C), or active nickel. Conducted under hydrogen pressures of 0.5-3 MPa and temperatures of 40-100°C, this step simultaneously reduces the nitro groups on both the benzimidazole core and the pendant phenyl ring to primary amines. This chemoselective reduction is vital; it must convert the nitro groups without hydrogenating the aromatic rings or cleaving the newly formed benzimidazole structure. The mechanistic precision here ensures that the final product retains the intended non-coplanar geometry established by the substituents X and Y. Impurity control is inherently managed through this sequence, as the harsh cyclization conditions tend to degrade unstable byproducts, and the final recrystallization from N-methylformamide and water further purifies the diamine to stringent specifications required for polymerization.
How to Synthesize Non-Coplanar Benzimidazole Diamine Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and purity. The process is designed to be telescoped where possible, minimizing isolation steps and solvent usage. The initial condensation can be monitored via TLC to ensure complete conversion before proceeding to the high-temperature cyclization. The crude cyclized product often does not require extensive purification before the final hydrogenation, streamlining the workflow. However, the final recrystallization is non-negotiable for achieving the high purity necessary for electronic-grade applications. The detailed operational parameters, including specific molar ratios and solvent volumes, are critical for reproducibility on a commercial scale.
- Perform condensation reaction between nitro-substituted diamine and benzoyl chloride derivatives using an acid-binding agent at 0-25°C.
- Execute ring-closing reaction on the crude condensation product using an acidic catalyst at elevated temperatures (180-220°C).
- Conduct catalytic hydrogenation of the nitro-intermediate under pressure (0.5-3 MPa) using Pd/C or similar catalysts to yield the final diamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers tangible strategic benefits beyond mere technical performance. The reliance on commodity chemicals and standard reaction types significantly de-risks the supply chain. Unlike processes requiring exotic transition metal catalysts or cryogenic conditions, this method utilizes widely available reagents and equipment found in standard multipurpose chemical plants. This accessibility translates directly into cost stability and supply continuity, essential factors for long-term project planning in the volatile fine chemical market.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive starting materials and the elimination of complex purification steps between the condensation and cyclization phases. By avoiding the need for expensive chromatographic separations and utilizing simple filtration and recrystallization for purification, the overall cost of goods sold is significantly reduced. Furthermore, the high yields reported in the examples, often exceeding 90% in the final hydrogenation step, minimize raw material waste and maximize throughput per batch, driving down the unit cost for high-purity electronic chemical manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent production schedules. The reaction conditions, particularly the hydrogenation step, are well-understood and easily scalable from laboratory autoclaves to industrial reactors. This scalability means that suppliers can rapidly ramp up production to meet surging demand without lengthy process re-validation. Additionally, the stability of the intermediates allows for potential stockpiling, providing a buffer against supply chain disruptions and ensuring reliable delivery of critical polyimide monomers to downstream polymer manufacturers.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process is favorable. The solvents used, such as dichloromethane and alcohols, are standard and have established recovery and recycling protocols. The catalytic hydrogenation generates water as the primary byproduct, avoiding the heavy metal waste streams associated with stoichiometric reductions. This cleaner profile simplifies waste treatment and aligns with increasingly stringent global environmental regulations, reducing the compliance burden and associated costs for manufacturing facilities.
Frequently Asked Questions (FAQ)
Understanding the nuances of this technology is key to leveraging its full potential. The following questions address common inquiries regarding the practical application and scalability of this synthesis. These insights are derived directly from the technical disclosures and experimental data provided in the patent literature, ensuring accuracy and relevance for technical decision-makers evaluating this material for their supply chains.
Q: How does the non-coplanar structure improve polymer performance?
A: The introduction of bulky side groups (X and Y) forces the benzimidazole and phenyl rings to twist, breaking conjugation. This significantly enhances optical transparency and solubility compared to traditional planar monomers.
Q: What are the critical reaction conditions for the cyclization step?
A: The ring-closing reaction requires high thermal energy, typically between 180-220°C, in high-boiling solvents like NMP or sulfolane, catalyzed by strong acids such as p-toluenesulfonic acid.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the process utilizes cheap, commercially available raw materials and standard unit operations like hydrogenation and recrystallization, making it highly scalable with high yields exceeding 90% in the final step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Non-Coplanar Benzimidazole Diamine Supplier
The synthesis of non-coplanar benzimidazole diamines represents a significant leap forward in materials science, bridging the gap between thermal performance and optical clarity. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to bring this innovative chemistry from the patent lab to your production line. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale samples to tonnage supply is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch meets the exacting standards required for high-performance polyimide applications.
We invite you to explore how this advanced monomer technology can enhance your product portfolio. Whether you are developing flexible displays, high-temperature adhesives, or advanced composite materials, our custom synthesis capabilities can be tailored to your specific needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you integrate these high-value intermediates into your manufacturing processes efficiently and economically.
