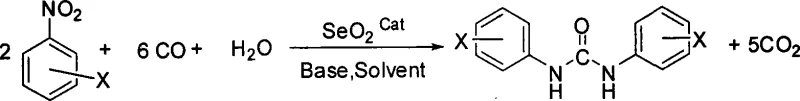
Advanced SeO2-Catalyzed Carbonylation for Commercial Scale-Up of Symmetrical Aryl Urea Intermediates
The chemical industry is currently witnessing a significant paradigm shift towards safer and more sustainable synthetic methodologies, particularly in the production of high-value intermediates. Patent CN101219972A introduces a groundbreaking approach for the synthesis of symmetrical aryl urea compounds, utilizing selenium dioxide (SeO2) as a highly efficient catalyst. This technology addresses critical pain points associated with traditional urea synthesis, specifically the reliance on hazardous phosgene gas or expensive transition metal complexes. By leveraging a reductive carbonylation strategy under normal pressure conditions, this method offers a robust pathway for generating high-purity pharmaceutical intermediates. The process operates within a moderate temperature range of 50 to 100°C, utilizing carbon monoxide and water as key reagents alongside nitrobenzene derivatives. For R&D directors and process chemists, this represents a viable alternative that balances reaction efficiency with stringent safety protocols, ensuring that the production of complex urea scaffolds can be achieved without compromising on environmental standards or operational safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of aromatic ureas has been dominated by the phosgenation of amines, a process fraught with severe safety hazards and logistical challenges. Phosgene is an extremely toxic gas that requires specialized containment infrastructure, and the reaction generates stoichiometric amounts of hydrochloric acid, leading to significant corrosion of production equipment and necessitating costly neutralization steps. Furthermore, alternative non-phosgene routes often rely on precious transition metal catalysts such as ruthenium, rhodium, or palladium complexes. While effective, these noble metals drive up raw material costs substantially and introduce the risk of heavy metal contamination in the final API, requiring rigorous and expensive purification steps to meet regulatory limits. Additionally, many existing carbonylation methods require high-pressure reactors to activate carbon monoxide effectively, increasing capital expenditure and operational complexity. These factors collectively create a bottleneck for procurement managers seeking cost-effective and scalable solutions for urea-based intermediates.
The Novel Approach
The methodology described in CN101219972A circumvents these traditional limitations by employing selenium dioxide as a non-precious metal catalyst capable of activating carbon monoxide under atmospheric pressure. This innovation eliminates the need for high-pressure autoclaves, thereby reducing equipment investment and simplifying the operational workflow. The use of nitrobenzene compounds as direct starting materials bypasses the need for a separate reduction step to generate anilines, streamlining the synthetic sequence into a single pot operation. The reaction system utilizes readily available industrial carbon monoxide tail gas, which may contain inert gases like nitrogen, further enhancing the economic feasibility by avoiding the need for ultra-high purity CO feeds. By operating at mild temperatures between 50 and 100°C, the process minimizes thermal degradation of sensitive functional groups, ensuring high selectivity and yield. This approach not only mitigates safety risks associated with phosgene but also drastically lowers the barrier to entry for manufacturers looking to produce symmetrical aryl ureas efficiently.
Mechanistic Insights into SeO2-Catalyzed Reductive Carbonylation
The core of this technological advancement lies in the unique ability of selenium dioxide to mediate the transfer of oxygen and facilitate the reduction of the nitro group in the presence of carbon monoxide and water. Mechanistically, the selenium species likely undergoes a redox cycle where it activates the nitro functionality, promoting its reduction to an intermediate amine or nitroso species in situ. Simultaneously, carbon monoxide acts as both a reducing agent and a carbonyl source, inserting into the nitrogen framework to form the urea linkage. The presence of water is critical, serving as a proton source and participating in the hydrolysis steps necessary for catalyst turnover. The base, whether inorganic like sodium acetate or organic like pyridine, plays a pivotal role in neutralizing acidic byproducts and maintaining the optimal pH for the catalytic cycle to proceed. This intricate interplay ensures that the reaction proceeds with high atom economy, converting the nitro substrate directly into the desired urea without isolating unstable intermediates.
Impurity control is another critical aspect where this mechanism excels, particularly for applications requiring high-purity pharmaceutical intermediates. The high selectivity observed in this system, with yields reaching up to 91% for unsubstituted diphenylurea, suggests that side reactions such as over-reduction to azo compounds or formation of asymmetric ureas are effectively suppressed. The specific choice of polar aprotic solvents like DMF or DMSO enhances the solubility of the polar transition states and stabilizes the catalytic species, further minimizing the formation of tarry byproducts. Moreover, the ability to tolerate a wide range of substituents, including electron-withdrawing groups like chloro and cyano, indicates that the electronic nature of the substrate does not significantly hinder the catalytic cycle. For quality control teams, this means a cleaner crude profile, which simplifies downstream purification and reduces the overall cost of goods sold by minimizing yield losses during recrystallization.
How to Synthesize Symmetrical Aryl Urea Efficiently
To implement this synthesis effectively, precise control over reaction parameters is essential to maximize yield and catalyst efficiency. The protocol involves charging a reactor with the nitrobenzene substrate, selenium dioxide catalyst, a suitable base, and a polar solvent, followed by the introduction of carbon monoxide at atmospheric pressure. The mixture is then heated to the optimal temperature range, typically around 90°C, for a duration of 5 to 8 hours to ensure complete conversion. Following the reaction, a unique workup procedure involves switching the gas atmosphere to oxygen or air, which induces the precipitation of the selenium catalyst, allowing for its physical separation via filtration. This streamlined workflow minimizes unit operations and facilitates the isolation of the target urea compound through standard distillation and recrystallization techniques.
- Charge a reactor with selenium dioxide catalyst, nitrobenzene derivative substrate, water, base (e.g., sodium acetate), and polar solvent like DMF.
- Maintain a continuous flow of carbon monoxide gas while heating the mixture to 50-100°C for 5 to 20 hours to facilitate reductive carbonylation.
- Cool the reaction to room temperature, switch gas flow to oxygen or air to precipitate the catalyst, then filter, distill solvent, and recrystallize the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial advantages that directly impact the bottom line and supply chain resilience for chemical manufacturers. The elimination of phosgene removes the need for specialized safety protocols and expensive containment systems, leading to significant reductions in capital expenditure and insurance costs. Furthermore, the substitution of expensive noble metal catalysts with inexpensive selenium dioxide drastically lowers the raw material cost per kilogram of product. The ability to use industrial grade carbon monoxide tail gas rather than high-purity cylinders provides additional flexibility in sourcing feedstocks, insulating the supply chain from volatility in specialty gas markets. These factors combine to create a manufacturing process that is not only economically superior but also more robust against supply disruptions.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of high-cost inputs with commoditized chemicals. By avoiding the use of precious metals like palladium or rhodium, the catalyst cost is reduced by orders of magnitude, directly improving the gross margin of the final intermediate. Additionally, the ambient pressure operation eliminates the energy costs associated with compressing gases and maintaining high-pressure reactors. The simplified workup procedure, which allows for catalyst precipitation via air oxidation, reduces the consumption of filtration aids and solvents compared to complex extraction workflows. These cumulative efficiencies result in a leaner cost structure that allows suppliers to offer more competitive pricing without sacrificing quality.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly enhanced by the use of widely available starting materials such as nitrobenzene derivatives and common solvents like DMF. Unlike processes dependent on scarce or geographically concentrated reagents, the inputs for this synthesis are produced globally at large scales, ensuring consistent availability. The robustness of the reaction conditions, which tolerate impurities in the carbon monoxide feed, further reduces the risk of batch failures due to feedstock quality variations. This reliability allows procurement managers to forecast production schedules with greater confidence and maintain lower safety stock levels, optimizing working capital utilization across the organization.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of high-pressure hazards and exothermic risks associated with phosgenation. The reaction can be easily adapted to standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. Environmentally, the process aligns with green chemistry principles by generating fewer hazardous wastes; the primary byproduct is carbon dioxide, and the selenium catalyst can potentially be recovered and recycled. This reduced environmental footprint simplifies regulatory compliance and waste disposal logistics, making it an attractive option for manufacturers operating under strict environmental regulations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its adoption for commercial production. The following questions address common inquiries regarding catalyst handling, substrate compatibility, and process safety. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Engaging with these technical details helps clarify the operational requirements and potential benefits for specific application scenarios.
Q: How does the SeO2 catalytic system compare to traditional phosgene methods?
A: Unlike phosgene methods which involve highly toxic reagents and corrosive HCl byproducts, the SeO2 catalytic system operates under normal pressure using CO and water, significantly enhancing operational safety and reducing equipment corrosion risks.
Q: What is the substrate scope for this urea synthesis method?
A: The method demonstrates high versatility, accommodating nitrobenzene derivatives with various substituents including electron-donating groups like alkyl and alkoxy, as well as electron-withdrawing groups such as halogens, nitro, cyano, and trifluoromethyl.
Q: Is the selenium dioxide catalyst recoverable in this process?
A: Yes, the process includes a specific step where switching the atmosphere from carbon monoxide to oxygen or air at the end of the reaction causes the catalyst to precipitate, allowing for easy filtration and potential recovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Symmetrical Aryl Urea Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting advanced synthetic technologies to maintain a competitive edge in the global market. Our team of expert process chemists has thoroughly evaluated the SeO2-catalyzed carbonylation route and is fully prepared to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of symmetrical aryl urea intermediate meets the highest industry standards for pharmaceutical applications.
We invite potential partners to collaborate with us to leverage this cost-effective and safe manufacturing technology for their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out to request specific COA data and route feasibility assessments to determine how this novel synthesis method can optimize your production costs and enhance your supply chain reliability. Let us help you secure a sustainable and economical source of high-quality urea intermediates for your next generation of products.
