Advanced Enzymatic Reduction Technology for Scalable Production of Chiral Pharmaceutical Intermediates
Advanced Enzymatic Reduction Technology for Scalable Production of Chiral Pharmaceutical Intermediates
The global demand for high-purity chiral building blocks continues to surge, driven by the relentless expansion of the statin drug market and complex active pharmaceutical ingredient (API) synthesis. In this context, Patent CN114908129B introduces a groundbreaking biocatalytic methodology for the preparation of (R)-4-chloro-3-hydroxybutyric acid ethyl ester, a critical precursor for various therapeutic agents including L-carnitine and macrolactim derivatives. This technology leverages a specifically engineered NADP-dependent isopropanol dehydrogenase mutant to achieve asymmetric reduction with exceptional stereocontrol. By shifting away from traditional transition metal catalysis, this innovation addresses long-standing challenges in impurity profiles and process safety, offering a robust alternative for reliable pharmaceutical intermediate supplier networks seeking sustainable manufacturing solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of (R)-4-chloro-3-hydroxybutyric acid ethyl ester has relied heavily on chemical hydrogenation utilizing chiral ruthenium complexes under rigorous high-pressure conditions. While capable of achieving moderate enantiomeric excess, these conventional routes suffer from significant economic and operational drawbacks that hinder cost reduction in API manufacturing. The reliance on precious metal catalysts not only inflates raw material costs but also introduces severe contamination risks, necessitating complex and expensive downstream purification steps to meet stringent heavy metal limits. Furthermore, the requirement for high-pressure hydrogenation reactors imposes substantial capital expenditure (CAPEX) and creates inherent safety hazards, limiting the flexibility of production facilities and complicating the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the biocatalytic strategy disclosed in the patent utilizes a highly active mutant dehydrogenase to facilitate the stereoselective reduction of ethyl 4-chloroacetoacetate under mild physiological conditions. This enzymatic pathway operates efficiently at ambient pressure and a moderate temperature of 30°C, effectively eliminating the need for hazardous high-pressure infrastructure. The biological system demonstrates remarkable substrate tolerance, accommodating concentrations up to 100g/L while maintaining a conversion rate exceeding 90% and an enantiomeric excess (ee) value greater than 99.5%. This dramatic improvement in selectivity ensures that the resulting product stream is exceptionally clean, drastically simplifying isolation procedures and enhancing the overall yield of high-purity OLED material or pharmaceutical precursors derived from this chiral pool.
Mechanistic Insights into NADP-Dependent Isopropanol Dehydrogenase Catalysis
The core of this technological breakthrough lies in the rational design and directed evolution of an isopropanol dehydrogenase derived from Caldanaerobacter subterraneus. Unlike typical ketoreductases, this bifunctional enzyme possesses the unique ability to utilize NADPH as a cofactor for the specific reduction of the 3-keto group in the substrate to the desired (R)-hydroxyl configuration. The patent details a specific mutant sequence (SEQ ID NO: 3) containing six critical amino acid substitutions—Y99C, M106I, A121T, H135R, E165K, and R338T—which collectively reshape the active site pocket. These modifications enhance the binding affinity for the bulky chloro-substituted substrate and optimize the hydride transfer mechanism, resulting in a catalytic efficiency that is more than three times superior to the wild-type enzyme, thereby enabling rapid turnover rates essential for industrial throughput.
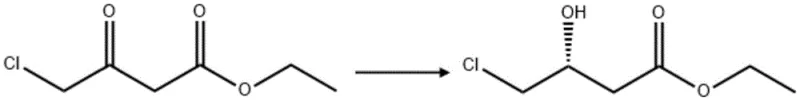
To sustain this high catalytic activity economically, the process integrates an efficient cofactor regeneration system coupled with glucose dehydrogenase (GDH). In this closed-loop cycle, the oxidation of inexpensive glucose drives the reduction of NADP+ back to NADPH, which is immediately consumed by the dehydrogenase to reduce the keto-ester substrate. This coupling ensures that the expensive cofactor is used in catalytic rather than stoichiometric quantities, a critical factor for reducing lead time for high-purity pharmaceutical intermediates by minimizing reagent costs. The reaction proceeds in an aqueous buffer system at pH 6.8, which not only stabilizes the enzyme structure but also suppresses non-enzymatic side reactions such as hydrolysis or racemization, ensuring the integrity of the chiral center throughout the transformation.
How to Synthesize Ethyl (R)-4-chloro-3-hydroxybutyrate Efficiently
The implementation of this biocatalytic route requires precise control over fermentation and reaction parameters to maximize the expression of the recombinant enzyme and its subsequent catalytic performance. The process begins with the cultivation of engineered E. coli BL21 (DE3) strains harboring the mutant dehydrogenase plasmid, followed by the preparation of a whole-cell biocatalyst or immobilized enzyme formulation. The reaction is initiated by introducing the substrate ethyl 4-chloroacetoacetate into a buffered solution containing the biocatalyst, glucose, and trace amounts of NADP+. Detailed standardized synthetic steps for optimizing cell density, substrate feeding strategies, and downstream extraction protocols are outlined in the comprehensive guide below to ensure reproducibility and compliance with GMP standards.
- Construct recombinant E. coli strains expressing the mutant isopropanol dehydrogenase (SEQ ID NO: 3) and glucose dehydrogenase for cofactor regeneration.
- Prepare the reaction system with 10wt% ethyl 4-chloroacetoacetate substrate, glucose, and NADP+ in potassium phosphate buffer at pH 6.8.
- Incubate the mixture at 30°C for 8-20 hours to achieve over 90% conversion and >99.5% ee value, followed by standard extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from chemical hydrogenation to this enzymatic platform offers profound strategic benefits that extend beyond simple unit cost calculations. The elimination of high-pressure processing and precious metal catalysts fundamentally alters the cost structure of production, removing volatile dependencies on ruthenium markets and reducing the energy intensity of the manufacturing process. This shift allows for more predictable budgeting and significantly reduced operational expenditures, as the mild reaction conditions permit the use of standard stainless steel reactors rather than specialized high-pressure vessels. Consequently, this technology enhances supply chain reliability by decentralizing production capabilities to facilities that may lack high-pressure infrastructure but possess standard fermentation and batch processing capabilities.
- Cost Reduction in Manufacturing: The economic model of this biocatalytic process is driven by the substitution of expensive chiral metal catalysts with renewable biological catalysts produced via fermentation. By utilizing glucose as the terminal reductant, the process avoids the procurement of costly stoichiometric reducing agents like sodium borohydride or hydrogen gas compression systems. Furthermore, the high stereoselectivity (>99.5% ee) minimizes the loss of material to unwanted enantiomers, effectively doubling the theoretical yield of the desired isomer compared to resolution methods, which translates to substantial cost savings in raw material utilization and waste disposal.
- Enhanced Supply Chain Reliability: The reliance on recombinant E. coli for enzyme production ensures a scalable and consistent source of biocatalyst that is not subject to the geopolitical supply constraints often associated with rare earth metals. The robustness of the mutant enzyme allows for high substrate loading (10wt%), which increases the volumetric productivity of the reactors and reduces the number of batches required to meet demand. This efficiency shortens production cycles and improves the agility of the supply chain, enabling manufacturers to respond more rapidly to fluctuations in market demand for statin intermediates and other fine chemicals.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this aqueous-based enzymatic process generates significantly less hazardous waste compared to traditional organic solvent-heavy chemical syntheses. The absence of heavy metal residues simplifies wastewater treatment and reduces the regulatory burden associated with discharging metal-contaminated effluents. Additionally, the mild operating conditions lower the risk of thermal runaway or pressure-related accidents, facilitating easier regulatory approval for plant expansions and ensuring long-term operational continuity in increasingly regulated chemical manufacturing environments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dehydrogenase-mediated reduction technology. These insights are derived directly from the experimental data and embodiments described in the patent documentation, providing clarity on enzyme stability, substrate scope, and process scalability. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this biocatalytic route into existing production lines for chiral alcohol synthesis.
Q: What represents the primary advantage of this enzymatic method over traditional chemical hydrogenation?
A: The primary advantage is the elimination of expensive chiral ruthenium catalysts and high-pressure equipment. This biological route operates at mild temperatures (30°C) and atmospheric pressure, achieving superior stereoselectivity (>99.5% ee) while significantly reducing safety risks and energy consumption associated with high-pressure hydrogenation.
Q: How does the mutant enzyme improve upon the wild-type dehydrogenase?
A: Through directed evolution involving error-prone PCR, the mutant enzyme (SEQ ID NO: 3) exhibits a catalytic activity approximately 3.2 times higher than the wild-type strain. Specific mutations such as Y99C, M106I, and R338T enhance the enzyme's affinity for the substrate, allowing for higher substrate loading (up to 100g/L) and faster reaction kinetics suitable for industrial scale-up.
Q: Is the cofactor regeneration system cost-effective for large-scale manufacturing?
A: Yes, the process utilizes a glucose dehydrogenase (GDH) coupled system with inexpensive glucose as the sacrificial reductant. This ensures the continuous recycling of the NADPH cofactor in situ, eliminating the need for stoichiometric addition of expensive cofactors and making the process economically viable for ton-scale production of chiral intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-4-chloro-3-hydroxybutyric acid ethyl ester Supplier
As the pharmaceutical industry increasingly prioritizes green chemistry and cost-efficient manufacturing, the adoption of advanced biocatalytic technologies like the one described in Patent CN114908129B becomes a key differentiator for market leaders. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such innovative processes to reality. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, ensuring that every batch of chiral intermediate meets the exacting standards required for global API registration and commercial distribution.
We invite forward-thinking partners to collaborate with us to harness the full potential of this enzymatic reduction technology for your specific product portfolio. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current manufacturing bottlenecks. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in biocatalysis can drive down your costs while securing a sustainable and high-quality supply of critical pharmaceutical intermediates.
