Scalable Metal-Free Synthesis of 3-Indoleselenyl Alcohols for Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of 3-Indoleselenyl Alcohols for Pharmaceutical Intermediates
The rapid advancement of medicinal chemistry has increasingly highlighted the critical role of organoselenium compounds, particularly 3-indoleselenyl alcohols, as potent scaffolds in drug discovery and development. These asymmetric selenide derivatives exhibit significant physiological activity, making them invaluable building blocks for complex pharmaceutical agents. However, traditional synthetic routes have long been plagued by severe limitations, including the use of toxic reagents and harsh reaction conditions that compromise both safety and scalability. A groundbreaking solution to these challenges is detailed in Chinese Patent CN108047118B, which discloses a novel, transition-metal-free methodology for synthesizing 3-indoleselenyl alcohol organic compounds. This innovative approach utilizes elemental selenium as a cost-effective selenium source and operates under remarkably mild conditions, often employing water as a green solvent. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this technology represents a paradigm shift towards sustainable and efficient manufacturing processes that align with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of asymmetric selenoethers and indole derivatives has relied heavily on methodologies that introduce significant operational hazards and environmental burdens. For instance, earlier reports by Venkataraman et al. described the preparation of asymmetric diaryl monoselenides using a CuI catalytic system; however, this route necessitates the use of phenylselenol, a reagent notorious for its foul smell and high toxicity, rendering it unsuitable for large-scale applications. Furthermore, the requirement for specific bases depending on the electronic nature of the aryl iodide adds complexity to process optimization. Similarly, the Zeni group developed a method utilizing ferric trichloride catalysis with diaryl diselenides. While this approach offered better atom efficiency, it still suffered from the unpleasant odor of diselenides and, more critically, the reliance on transition metal catalysts. The presence of residual metal contaminants in the final product is a major concern for pharmaceutical applications, often necessitating expensive and time-consuming purification steps to meet stringent regulatory purity specifications. These legacy methods collectively highlight an urgent industry need for cleaner, metal-free alternatives.
The Novel Approach
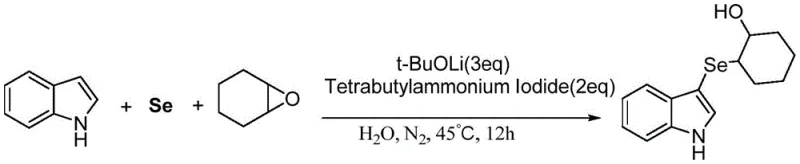
In stark contrast to these conventional techniques, the method disclosed in CN108047118B offers a streamlined, environmentally benign pathway that eliminates the need for transition metals entirely. By employing elemental selenium powder directly with indole derivatives and various epoxides, such as cyclohexene oxide or 1,2-epoxybutane, the reaction proceeds efficiently under the action of an inorganic base. A key feature of this innovation is the versatility of the solvent system; while organic solvents like tetrahydrofuran can be used, the process uniquely supports the use of water as a reaction medium when supplemented with a phase transfer catalyst like tetrabutylammonium iodide. This capability not only drastically reduces solvent costs but also aligns with global sustainability goals by minimizing volatile organic compound (VOC) emissions. The reaction typically operates at mild temperatures around 45°C under a nitrogen atmosphere, ensuring high functional group tolerance and exceptional yields, thereby providing a robust platform for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Transition-Metal-Free Selenylation
The mechanistic elegance of this synthesis lies in its ability to activate elemental selenium without the aid of transition metals, relying instead on a synergistic combination of strong inorganic bases and phase transfer catalysts. The process initiates with the deprotonation of the indole nitrogen or the activation of the selenium species by the base, such as lithium tert-butoxide, generating a reactive nucleophilic selenium intermediate. This species then attacks the electron-deficient carbon of the epoxide ring, facilitating ring opening and the formation of the carbon-selenium bond. The presence of tetrabutylammonium iodide is crucial when water is used as the solvent, as it facilitates the transport of ionic species into the organic phase or stabilizes the transition state, ensuring high reaction rates even in aqueous media. This mechanism avoids the formation of metal-selenium complexes that often lead to difficult-to-remove impurities, thereby simplifying the downstream purification process significantly. The result is a high-purity product profile that is essential for subsequent coupling reactions in multi-step drug synthesis pathways.
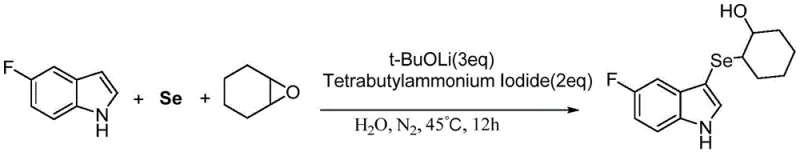
Furthermore, the substrate scope of this reaction demonstrates remarkable robustness, accommodating a wide array of functional groups on the indole ring without compromising yield or selectivity. As illustrated in the patent examples, substituents such as fluoro, chloro, methyl, and methoxy groups at various positions on the indole nucleus are well-tolerated, yielding the corresponding 3-indoleselenyl alcohols in good to excellent yields ranging from 70% to 97%. For instance, the synthesis of 2-(5-fluoro-3-indolylselenyl)-1-cyclohexyl alcohol proceeds smoothly, indicating that electron-withdrawing groups do not inhibit the nucleophilic attack. Similarly, electron-donating groups like methyl and methoxy also participate effectively, showcasing the versatility of this method for generating diverse chemical libraries. This broad tolerance is a critical advantage for medicinal chemists who require rapid access to structurally varied analogs for structure-activity relationship (SAR) studies without needing to re-optimize reaction conditions for each new substrate.
How to Synthesize 3-Indoleselenyl Alcohol Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to atmospheric conditions and reagent stoichiometry to maximize efficiency and safety. The standard protocol involves charging a reaction vessel, such as a Schlenk tube, with selenium powder, the chosen inorganic base, the phase transfer catalyst, and the indole substrate at room temperature. It is imperative to establish an inert nitrogen atmosphere by vacuumizing and backfilling with nitrogen gas multiple times to prevent oxidation of the sensitive selenium intermediates. Once the atmosphere is secured, the epoxide substrate and the solvent, preferably deionized water for green processing, are added. The mixture is initially stirred at room temperature to ensure homogeneity before being transferred to a heating bath maintained at 45°C. Reaction progress is typically monitored via TLC or GC-MS, with completion usually achieved within 12 hours. For detailed operational parameters and specific workup procedures, please refer to the standardized guide below.
- Charge a Schlenk tube with selenium powder, lithium tert-butoxide, tetrabutylammonium iodide, and indole under nitrogen atmosphere.
- Add cyclohexene oxide and deionized water, then stir at room temperature before heating to 45°C for 12 hours.
- Upon completion, dilute with water, extract with ethyl acetate, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement and supply chain perspective, the adoption of this metal-free synthesis route offers profound advantages that extend far beyond simple yield improvements. Traditional methods relying on transition metal catalysts like copper or iron introduce significant hidden costs related to catalyst procurement, specialized handling requirements, and extensive post-reaction purification to remove metal residues. By eliminating these metals entirely, manufacturers can achieve substantial cost savings in raw material acquisition and waste management. Furthermore, the ability to use water as a primary solvent dramatically reduces the expenditure on expensive organic solvents and lowers the environmental compliance burden associated with solvent disposal. This shift not only optimizes the cost structure of the manufacturing process but also enhances the overall sustainability profile of the supply chain, making it more resilient to regulatory changes regarding hazardous chemical usage.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and toxic diselenide reagents directly translates to a leaner bill of materials. Elemental selenium is a commodity chemical that is significantly cheaper and easier to source than specialized organometallic catalysts or malodorous diselenides. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces labor hours and consumable usage during purification. This streamlined process flow allows for a more efficient allocation of resources, driving down the overall cost of goods sold (COGS) for high-purity pharmaceutical intermediates while maintaining competitive pricing structures for downstream clients.
- Enhanced Supply Chain Reliability: Relying on readily available, stable starting materials like elemental selenium and common epoxides mitigates the risk of supply disruptions often associated with specialized or hazardous reagents. The robustness of the reaction conditions, which tolerate a wide range of functional groups and operate at mild temperatures, ensures consistent batch-to-batch quality and reduces the likelihood of production failures. This reliability is crucial for maintaining continuous supply lines to pharmaceutical customers who demand strict adherence to delivery schedules. Moreover, the reduced toxicity of the reagents simplifies logistics and storage requirements, allowing for safer and more flexible inventory management within the manufacturing facility.
- Scalability and Environmental Compliance: The compatibility of this method with aqueous solvent systems positions it as an ideal candidate for large-scale industrial production. Water is non-flammable, non-toxic, and abundant, removing many of the safety hazards associated with scaling up reactions in volatile organic solvents. The absence of heavy metals in the process stream simplifies wastewater treatment and ensures compliance with increasingly stringent environmental regulations regarding heavy metal discharge. This environmental stewardship not only protects the company from potential regulatory fines but also enhances its reputation as a responsible manufacturer, which is a key differentiator when partnering with global pharmaceutical companies that prioritize green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, aiming to clarify the practical benefits and mechanistic details for potential adopters. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this route into their existing production workflows. The answers provided reflect the optimized conditions and observed outcomes from extensive experimentation, ensuring that stakeholders have accurate information for decision-making.
Q: Why is elemental selenium preferred over diselenides in this synthesis?
A: Elemental selenium avoids the foul odor and high toxicity associated with traditional diselenide reagents, while also eliminating the need for expensive transition metal catalysts like copper or iron, resulting in a greener and safer process.
Q: What are the optimal reaction conditions for high yield?
A: The patent specifies that using lithium tert-butoxide as the base in water with tetrabutylammonium iodide as a phase transfer catalyst at 45°C for 12 hours provides excellent yields up to 97%.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method utilizes mild temperatures, inexpensive raw materials like elemental selenium, and potentially water as a solvent, making it highly scalable and compliant with environmental regulations for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Indoleselenyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free synthesis technology described in CN108047118B for the production of high-value organoselenium intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-indoleselenyl alcohol delivered meets the highest standards required for pharmaceutical applications. We are committed to leveraging this green chemistry innovation to provide our partners with a competitive edge through superior product quality and process efficiency.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your supply chain and reduce manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating the tangible economic benefits of switching to this metal-free protocol. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your drug development timeline with a sustainable, scalable, and cost-effective supply solution for your organoselenium intermediate requirements.
