Advanced Synthesis of Allyloxy Polyfluoro Liquid Crystals for Next-Gen Display Manufacturing
Advanced Synthesis of Allyloxy Polyfluoro Liquid Crystals for Next-Gen Display Manufacturing
The rapid evolution of flat-panel display technologies, specifically Vertically-Aligned (VA) and In-Plane Switching (IPS) modes, has created an urgent demand for liquid crystal materials with precise electro-optical properties. Patent CN102559201A introduces a breakthrough class of allyloxy lateral polyfluoro liquid crystal compounds that address the critical limitations of traditional cyano-based or mono-fluorinated mixtures. This technology leverages a sophisticated one-pot synthetic strategy to deliver compounds with large negative dielectric anisotropy, exceptionally low viscosity, and broad nematic phase intervals. For R&D directors and procurement specialists in the electronic chemical sector, this patent represents a pivotal shift towards more efficient, high-performance material sourcing. The disclosed methodology not only enhances the thermal stability and response speed of the final display mixture but also simplifies the manufacturing workflow, offering a compelling value proposition for reliable display material supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the formulation of liquid crystal mixtures for VA and IPS displays relied heavily on laterally substituted cyano compounds to achieve the necessary negative dielectric anisotropy. However, these legacy materials suffer from inherently low resistivity, which compromises the voltage holding ratio and overall display quality in large-screen applications. Furthermore, alternative lateral fluorine-substituted biphenyl compounds, while offering better resistivity, often exhibit insufficient dielectric anisotropy values, typically around 0.1, limiting their utility to basic formulations rather than high-performance mixes. As illustrated in the prior art structures, many existing compounds struggle to balance low melting points with wide liquid crystal phase ranges, often resulting in high viscosity that slows down response times or narrow operating temperature windows that restrict device versatility.
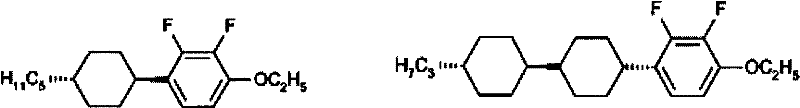
The Novel Approach
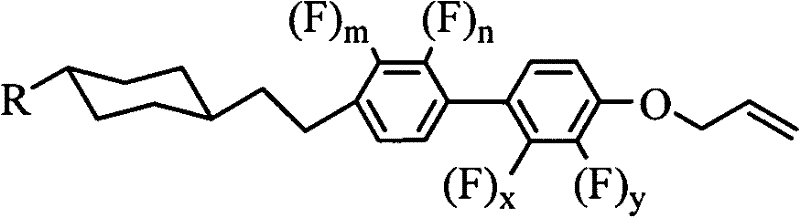
The innovation described in CN102559201A overcomes these hurdles by introducing a multi-fluorine substitution pattern combined with a terminal allyloxy group. This structural modification, depicted in Formula I, allows for fine-tuning of the molecular dipole moment to achieve significant negative dielectric anisotropy without sacrificing resistivity. The incorporation of the allyloxy tail further depresses the melting point and reduces rotational viscosity, directly addressing the response speed requirements of modern high-refresh-rate displays. By systematically varying the fluorine substitution positions (m, n, x, y), manufacturers can create a diverse library of compounds tailored for specific mixture formulations, ensuring cost reduction in electronic chemical manufacturing through optimized performance-to-cost ratios.
Mechanistic Insights into Pd-Catalyzed One-Pot Coupling
The core of this technological advancement lies in its elegant reaction mechanism, which seamlessly integrates a Suzuki-Miyaura cross-coupling reaction with a subsequent nucleophilic substitution in a single reactor vessel. The process initiates with the oxidative addition of a substituted aryl halide, such as 2,3-difluoro-4-bromophenol, to a tetrakis(triphenylphosphine)palladium(0) catalyst. This active Pd(0) species facilitates the transmetallation with the corresponding arylboronic acid derivative, which carries the cyclohexyl-ethyl backbone essential for the mesogenic core. The presence of a phase transfer catalyst, tetra-n-butylammonium bromide, alongside a mild inorganic base like potassium carbonate, ensures efficient activation of the boronic acid in the aqueous-organic solvent interface, driving the biaryl bond formation with high selectivity and minimal homocoupling byproducts.
Following the initial coupling, the reaction mixture undergoes a direct nucleophilic substitution without the need for intermediate isolation or purification. Upon the addition of 3-bromopropene, the phenolic hydroxyl group generated in the first step acts as a nucleophile, displacing the bromide to form the stable allyl ether linkage. This telescoped approach is critical for impurity control, as it prevents the exposure of reactive intermediates to atmospheric moisture or oxygen, which could otherwise lead to oxidation or hydrolysis side reactions. The result is a highly pure crude product that requires only standard extraction and recrystallization to meet stringent purity specifications, demonstrating a robust pathway for the commercial scale-up of complex display materials.
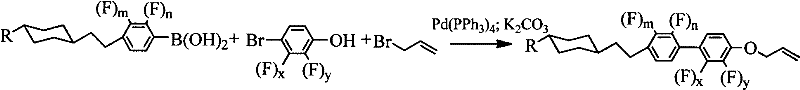
How to Synthesize Allyloxy Polyfluoro Liquid Crystal Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for reproducing these high-value intermediates with consistent quality. The procedure emphasizes the importance of strict inert atmosphere conditions and precise stoichiometric control to maximize yield and minimize palladium residue. Detailed standardized synthesis steps are provided below to guide process chemists in implementing this technology.
- Combine substituted arylboronic acid, substituted halogenated aromatic hydrocarbon, phase transfer catalyst, and base in a mixed solvent system under nitrogen protection.
- Add palladium catalyst at 65°C to initiate the coupling reaction, followed by the addition of 3-bromopropene for nucleophilic substitution without intermediate isolation.
- Perform aqueous workup, extract with petroleum ether, purify via column chromatography, and recrystallize from ethanol to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-pot synthesis route offers transformative benefits that extend beyond mere technical performance. The consolidation of two distinct reaction steps into a single operational unit significantly streamlines the production timeline, reducing the overall equipment occupancy time and labor intensity associated with batch processing. This efficiency gain translates directly into substantial cost savings, as the elimination of intermediate isolation steps removes the need for additional solvent volumes, filtration hardware, and drying cycles, thereby lowering the variable cost per kilogram of the final active pharmaceutical ingredient or electronic chemical.
- Cost Reduction in Manufacturing: The telescoped nature of the reaction eliminates the costly and time-consuming purification of the biaryl phenol intermediate. By avoiding the isolation of this intermediate, the process drastically reduces solvent consumption and waste disposal costs associated with multiple workup procedures. Furthermore, the use of commercially available and relatively inexpensive starting materials, such as substituted bromophenols and boronic acids, ensures a stable and predictable raw material cost structure, shielding the supply chain from volatile pricing fluctuations common in specialty chemical markets.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, utilizing standard reagents like potassium carbonate and common solvents such as DMF or ethanol, minimizes the risk of batch failures due to sensitive reagent degradation. This reliability is crucial for maintaining continuous supply lines to downstream panel manufacturers who operate on just-in-time inventory models. The simplicity of the workup, involving straightforward liquid-liquid extraction and column chromatography, allows for rapid turnaround times between batches, effectively reducing lead time for high-purity electronic chemicals and ensuring that production schedules are met without delay.
- Scalability and Environmental Compliance: The process is inherently scalable, as the reaction exotherms are manageable at 65°C, and the reagent ratios are optimized to prevent excessive byproduct formation. From an environmental perspective, the reduction in solvent usage and the ability to recover and recycle petroleum ether during the purification stage align with increasingly strict global regulations on volatile organic compound (VOC) emissions. This green chemistry approach not only mitigates regulatory risks but also enhances the corporate sustainability profile of the manufacturing entity, a key factor for multinational clients prioritizing eco-friendly supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for stakeholders evaluating this material for integration into their display formulations.
Q: What are the key performance advantages of this allyloxy polyfluoro liquid crystal compound?
A: The compound exhibits large negative dielectric anisotropy, low melting point, and low viscosity, making it ideal for VA and IPS display modes while maintaining a wide nematic phase interval.
Q: How does the one-pot synthesis method impact production costs?
A: By combining the coupling and substitution reactions in a single vessel without isolating intermediates, the process significantly reduces solvent consumption, labor hours, and waste generation.
Q: Is this synthesis route scalable for industrial manufacturing?
A: Yes, the use of standard reagents like potassium carbonate and tetrakis(triphenylphosphine)palladium, along with simple purification steps like recrystallization, ensures excellent scalability from lab to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyloxy Polyfluoro Liquid Crystal Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced liquid crystal intermediates play in the next generation of display technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering materials that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of allyloxy polyfluoro liquid crystal compound performs consistently in your high-end VA and IPS display applications.
We invite you to collaborate with us to leverage this patented technology for your product roadmap. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and innovation in your supply chain.
