Advanced Preparation Method of Obeticholic Acid for Commercial Scale-up and High Purity
The global pharmaceutical landscape is witnessing a surge in demand for effective treatments targeting non-alcoholic steatohepatitis (NASH) and primary biliary cholangitis (PBC), with Obeticholic Acid standing out as a critical therapeutic agent. As the industry seeks more efficient manufacturing pathways, patent CN111138509B emerges as a pivotal technological breakthrough, offering a robust and scalable synthesis route that addresses long-standing challenges in steroid chemistry. This innovative preparation method fundamentally restructures the synthetic approach by leveraging a strategic oxidation-reduction sequence that bypasses the need for complex protecting group manipulations and hazardous cryogenic conditions often associated with traditional methodologies. By focusing on the transformation of key intermediates such as Compound 14 and Compound 15, this technology enables the production of high-purity Obeticholic Acid with significantly reduced impurity profiles, thereby meeting the stringent regulatory requirements of international health authorities. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality at a commercial scale. The following analysis delves deep into the technical merits and commercial implications of this novel process, highlighting its potential to redefine cost structures and supply chain reliability in the hepatology drug sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Obeticholic Acid has been plagued by significant technical hurdles that compromise both economic viability and operational safety. Early approaches, such as those disclosed in patent application WO2002072598, relied heavily on the use of n-butyllithium for ethylation at the C6 position, a reaction that necessitates extremely low temperatures around -78°C to maintain any degree of selectivity. This cryogenic requirement not only imposes a massive energy burden on the manufacturing facility but also introduces severe safety risks associated with handling pyrophoric reagents on a large scale, often resulting in poor selectivity and low yields that are unacceptable for commercial production. Furthermore, other existing routes like those found in WO2006122977 and WO2018010651 frequently involve multi-step protection and deprotection strategies for the hydroxyl groups at the 3 and 7 positions, which drastically lengthens the synthetic timeline and accumulates unnecessary chemical waste. These conventional methods often suffer from side reactions during the reduction of double bonds and carbonyl groups under strong basic conditions, leading to complex impurity profiles that require costly and time-consuming purification processes to resolve. The cumulative effect of these inefficiencies is a fragile supply chain vulnerable to disruptions, high production costs, and inconsistent batch-to-batch quality, creating a pressing need for a more streamlined and robust manufacturing solution.
The Novel Approach
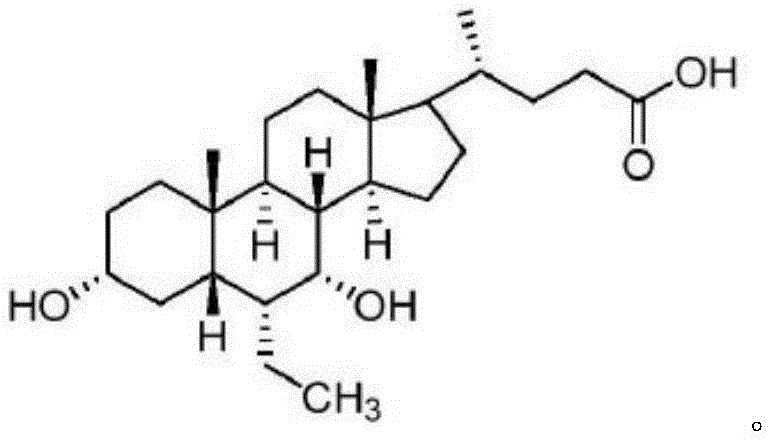
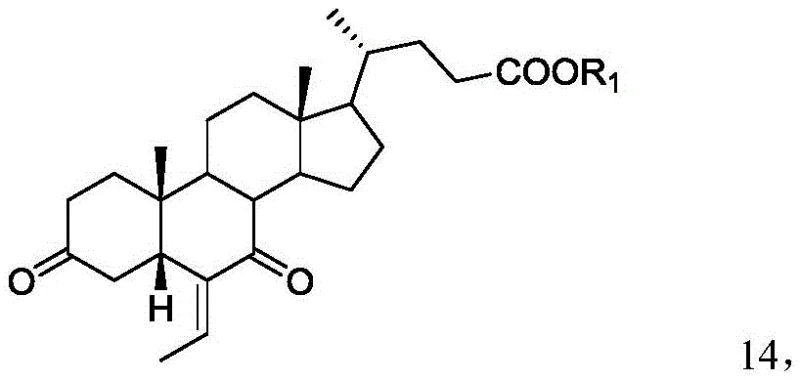
In stark contrast to the cumbersome legacy processes, the method described in patent CN111138509B introduces a remarkably concise and efficient synthetic strategy that directly addresses the core inefficiencies of prior art. This novel approach centers on the preparation of a specific intermediate, Compound 14, which serves as a pivotal junction for introducing the 6α-ethyl group through a controlled catalytic hydrogenation rather than a hazardous alkylation. By utilizing a sequence that involves the oxidation of the C3-hydroxyl group followed by the reduction of the C6-vinyl group and finally the stereoselective reduction of the resulting diketone, the process eliminates the need for cryogenic conditions and toxic organolithium reagents entirely. The strategic decision to defer the final reduction of the 3 and 7 carbonyls until the end of the sequence allows for better control over stereochemistry without the interference of protecting groups, thereby simplifying the workflow and enhancing overall atom economy. This streamlined pathway not only reduces the total number of unit operations but also significantly minimizes the generation of by-products, resulting in a cleaner reaction profile that facilitates easier downstream processing. For a reliable pharmaceutical intermediate supplier, adopting this methodology translates to a more resilient production capability that can consistently meet high-volume demands while adhering to strict environmental and safety standards.
Mechanistic Insights into Oxidation and Stereoselective Reduction
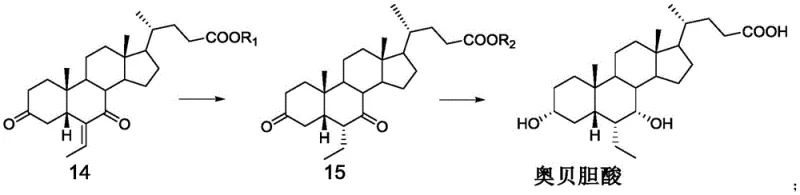
The chemical elegance of this preparation method lies in its precise control over oxidation states and stereochemical outcomes, particularly during the transformation of Compound 13 to Compound 14 and subsequently to the final product. The initial oxidation step typically employs Jones reagent, a mixture of chromium trioxide, sulfuric acid, and water, or alternative oxidants like pyridinium chlorochromate, to convert the 3-hydroxyl group of Compound 13 into a ketone, yielding Compound 14 with high efficiency. This oxidation is critical as it activates the ring system for the subsequent hydrogenation step, where the C6-vinyl group is reduced to an ethyl group using a palladium on carbon catalyst under hydrogen pressure. The choice of solvent system, often a mixture of methanol and water, plays a vital role in solubilizing the steroid substrate while maintaining the activity of the heterogeneous catalyst, ensuring complete conversion without over-reduction of other sensitive functional groups. Following the formation of Compound 15, the final and perhaps most crucial step involves the reduction of the 3,7-diketone system using sodium borohydride in an alkaline aqueous solution. The alkaline environment is not merely a solvent condition but a mechanistic driver that influences the conformational equilibrium of the steroid nucleus, favoring the attack of the hydride ion from the alpha face to establish the thermodynamically stable 3α and 7α hydroxyl configurations. This specific stereocontrol is paramount for the biological activity of Obeticholic Acid, and the ability to achieve it without auxiliary chiral catalysts or protecting groups represents a significant advancement in process chemistry.
Furthermore, the management of impurities throughout this synthetic sequence is inherently superior due to the avoidance of reactive intermediates that typically lead to side products in traditional routes. In the hydrogenation step converting Compound 14 to Compound 15, the use of mild conditions prevents the epimerization of chiral centers, a common issue in base-catalyzed enolization processes found in older methods. The post-treatment procedures, which involve careful pH adjustment and crystallization from solvents like n-butyl acetate, are designed to leverage the solubility differences between the target molecule and potential isomers or unreacted starting materials. By optimizing parameters such as temperature, which is maintained between 40°C and 120°C during the final reduction, and the molar ratio of the reducing agent, the process ensures that the kinetic and thermodynamic factors align to maximize the yield of the desired isomer. This rigorous control over reaction variables results in a final product with an HPLC purity exceeding 99%, demonstrating the robustness of the method against the formation of difficult-to-remove impurities. For R&D teams evaluating technology transfer, this level of mechanistic clarity and impurity control provides the confidence needed to scale the process from laboratory grams to multi-ton commercial batches without compromising quality.
How to Synthesize Obeticholic Acid Efficiently
The practical implementation of this synthesis route requires a disciplined approach to reaction conditions and workup procedures to fully realize the benefits outlined in the patent literature. The process begins with the preparation of the key intermediate Compound 14, followed by its catalytic hydrogenation to Compound 15, and concludes with the stereoselective reduction to the final active pharmaceutical ingredient. Each step is optimized for scalability, utilizing reagents and solvents that are readily available in the global chemical market, thus avoiding supply bottlenecks associated with exotic or highly specialized materials. The detailed standardized synthesis steps provided below serve as a foundational guide for process engineers looking to replicate this high-efficiency pathway in a GMP-compliant environment.
- Oxidize Compound 13 using Jones reagent (CrO3/H2SO4) or alternative oxidants to form the 3-keto intermediate Compound 14.
- Perform catalytic hydrogenation on Compound 14 using Pd/C and hydrogen to reduce the C6-vinyl group to an ethyl group, yielding Compound 15.
- Dissolve Compound 15 in alkaline solution and reduce with sodium borohydride to stereoselectively form the 3α,7α-dihydroxy structure of Obeticholic Acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this novel synthesis method offers profound advantages that extend far beyond simple yield improvements, fundamentally altering the cost and risk profile of Obeticholic Acid manufacturing. By eliminating the need for cryogenic cooling systems and hazardous organolithium reagents, the process drastically reduces the capital expenditure required for specialized reactor infrastructure and lowers the operational costs associated with energy consumption and safety compliance. The removal of protection and deprotection steps further contributes to substantial cost savings by reducing the consumption of auxiliary chemicals and minimizing the volume of waste solvents that require treatment and disposal. This leaner manufacturing footprint not only enhances the economic competitiveness of the final product but also aligns with increasingly stringent environmental regulations, making it a sustainable choice for long-term production. Additionally, the reliance on robust catalytic hydrogenation and standard reduction chemistry ensures that the supply chain is less vulnerable to the volatility of niche reagent markets, providing greater stability and predictability for procurement managers planning annual budgets.
- Cost Reduction in Manufacturing: The elimination of low-temperature reactions and protecting group chemistry removes significant cost drivers from the production ledger, allowing for a more competitive pricing structure without sacrificing margin. By streamlining the synthetic route to fewer steps, the process reduces labor hours, equipment occupancy time, and utility consumption, all of which aggregate to lower the overall cost of goods sold. The high yields reported in the patent examples indicate that raw material utilization is maximized, further driving down the variable costs per kilogram of output. This efficiency creates a buffer against raw material price fluctuations, enabling the supplier to offer more stable pricing contracts to downstream pharmaceutical partners.
- Enhanced Supply Chain Reliability: The use of common industrial reagents such as sodium borohydride, palladium on carbon, and standard organic solvents ensures that the production process is not dependent on single-source or geographically constrained suppliers. This diversification of the supply base mitigates the risk of production stoppages due to material shortages, a critical factor for maintaining continuity of supply for life-saving medications. The robustness of the chemistry also means that the process is less sensitive to minor variations in raw material quality, reducing the frequency of batch failures and the need for reprocessing. Consequently, lead times for high-purity pharmaceutical intermediates can be significantly shortened, allowing customers to maintain leaner inventory levels while ensuring uninterrupted manufacturing of their final dosage forms.
- Scalability and Environmental Compliance: The inherent safety of the reaction conditions, which avoid extreme temperatures and pyrophoric substances, facilitates easier scale-up from pilot plant to full commercial production without the need for complex engineering controls. The reduction in chemical waste and solvent usage aligns with green chemistry principles, simplifying the permitting process and reducing the environmental liability associated with manufacturing operations. This compliance advantage is increasingly valuable as regulatory bodies worldwide tighten restrictions on pharmaceutical manufacturing emissions and waste discharge. The ability to scale efficiently while maintaining a low environmental impact positions this method as a future-proof solution for meeting the growing global demand for Obeticholic Acid.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific preparation method, drawing directly from the detailed specifications and comparative data provided in the patent documentation. These insights are intended to clarify the operational advantages and technical feasibility for stakeholders evaluating this technology for integration into their supply chains. Understanding these details is crucial for making informed decisions about sourcing strategies and process development partnerships.
Q: How does this new method improve upon the WO2002072598 process?
A: Unlike the WO2002072598 process which requires hazardous n-butyllithium at cryogenic temperatures (-78°C) with poor selectivity, this method utilizes mild oxidation and catalytic hydrogenation, significantly improving safety and yield.
Q: What ensures the stereoselectivity of the 3α and 7α hydroxyl groups?
A: The process employs a specific alkaline environment during the final sodium borohydride reduction step, which thermodynamically favors the formation of the desired α-configuration at both the 3 and 7 positions.
Q: Does this route require protecting groups for the hydroxyl functions?
A: No, one of the key advantages of this invention is the elimination of hydroxyl protection and deprotection steps, which shortens the synthetic route and reduces overall production costs and waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Obeticholic Acid Supplier
As the demand for effective NASH and PBC therapies continues to accelerate, partnering with a technically proficient manufacturing partner is essential for securing a stable supply of high-quality Obeticholic Acid. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent results for our global clientele. Our state-of-the-art facilities are equipped to handle the specific requirements of steroid synthesis, including catalytic hydrogenation and precise temperature-controlled reductions, ensuring that every batch meets stringent purity specifications and rigorous QC labs standards. We understand that in the pharmaceutical sector, consistency is key, and our commitment to quality assurance guarantees that our intermediates and APIs perform reliably in your downstream formulation processes.
We invite you to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project needs and timeline. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our optimized production route. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless transition to a more efficient and reliable supply source for your critical hepatology drug portfolio.
