Scalable Synthesis of Perfluoroalkyl Anilines for Next-Generation Agrochemical Manufacturing
Scalable Synthesis of Perfluoroalkyl Anilines for Next-Generation Agrochemical Manufacturing
The global demand for high-performance agrochemicals continues to drive innovation in fluorine chemistry, specifically regarding the efficient production of perfluoroalkyl-substituted anilines. Patent CN114394903A introduces a groundbreaking synthetic methodology that addresses critical bottlenecks in current manufacturing processes. By utilizing 2-bromoheptafluoropropane as a primary alkylating agent in conjunction with inexpensive ammonium salts as catalysts, this technology achieves exceptional yields exceeding 97% under optimized conditions. This represents a paradigm shift from traditional methods that rely on prohibitively expensive perfluoroisopropyl iodides or complex transition metal catalytic systems. For R&D directors and procurement strategists, this patent offers a clear pathway to reducing the cost of goods sold (COGS) while maintaining rigorous purity standards required for active pharmaceutical and agricultural ingredients.
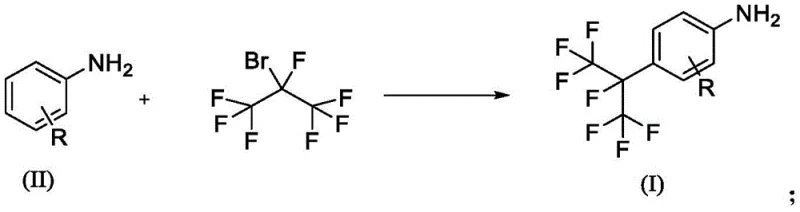
The strategic value of this invention lies in its ability to transform a historically low-yielding, high-cost reaction into a robust industrial process. Conventional approaches often struggle with the reactivity of perfluoroalkyl halides, necessitating harsh conditions or exotic reagents that complicate supply chains. In contrast, the disclosed method operates within a moderate temperature range of 60°C to 125°C using readily available solvents like acetonitrile. This accessibility of reagents ensures supply chain continuity, a crucial factor for supply chain heads managing long-term production schedules. Furthermore, the use of sodium dithionite as a radical initiator provides a controlled reaction environment that minimizes side reactions, thereby enhancing the overall impurity profile of the final product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of perfluoroalkyl-substituted anilines has been plagued by economic and technical inefficiencies. Prior art, such as JP201215363A, relies on perfluoroisopropyl iodide, a raw material that is not only exponentially more expensive than its bromide counterpart but also poses significant handling challenges due to the potential formation of free iodine and subsequent corrosion issues. Additionally, methods described in WO2020169768A utilize complex catalytic systems involving zinc, nickel nitrate, and specialized phosphine ligands like DPEPhos. These transition metal catalysts introduce severe downstream processing burdens, requiring extensive purification steps to remove trace metals to meet regulatory limits for agrochemical intermediates. The reliance on biphasic systems in some older technologies further complicates mass transfer, often resulting in inconsistent yields hovering around 77% to 85%, which is suboptimal for large-scale commercialization.
The Novel Approach
The methodology presented in CN114394903A fundamentally disrupts these legacy constraints by substituting expensive iodides with cost-effective 2-bromoheptafluoropropane. This switch alone drastically reduces the raw material input costs, a key metric for procurement managers evaluating vendor competitiveness. Moreover, the replacement of sophisticated transition metal complexes with simple inorganic ammonium salts, such as ammonium chloride or ammonium sulfate, eliminates the need for expensive ligand synthesis and metal scavenging operations. The reaction proceeds efficiently in a homogeneous phase when using solvents like acetonitrile, ensuring uniform heat distribution and reaction kinetics. This simplification of the chemical architecture allows for a streamlined workflow where the reaction can be driven to near-quantitative conversion, as evidenced by yields reaching 97.9% in specific embodiments, thereby maximizing asset utilization and throughput.
Mechanistic Insights into Radical Substitution Catalysis
The core of this technological advancement rests on a finely tuned radical substitution mechanism initiated by sodium dithionite. In this system, the dithionite acts as a single-electron transfer agent, generating the necessary perfluoroalkyl radicals from the 2-bromoheptafluoropropane precursor. The presence of ammonium chloride plays a subtle yet critical role, likely acting as a phase-transfer facilitator or stabilizing the transition state through hydrogen bonding interactions, which enhances the nucleophilic attack of the aniline derivative on the generated radical species. Unlike traditional Lewis acid catalysts that might coordinate too strongly with the nitrogen lone pair of the aniline, potentially deactivating the substrate, the ammonium salt maintains a delicate balance that promotes reactivity without inhibiting the amine functionality. This mechanistic elegance ensures that the reaction proceeds with high selectivity for the mono-alkylated product, minimizing the formation of di-alkylated impurities that are common in uncontrolled radical processes.
Impurity control is another hallmark of this catalytic system. The use of a closed pressure vessel system under inert gas protection, typically nitrogen, prevents oxidative degradation of the sensitive aniline starting materials and the perfluoroalkyl intermediates. The patent data indicates that by carefully controlling the molar ratio of the initiator to the substrate, specifically keeping the sodium dithionite ratio between 0.5 and 1.2 relative to the aniline, the generation of unwanted radical byproducts is suppressed. Furthermore, the specific temperature window of 60°C to 70°C identified in the examples is critical; temperatures that are too low fail to activate the bromide sufficiently, while excessive heat can lead to decomposition of the perfluoroalkyl chain or polymerization side reactions. This precise thermal management results in a clean reaction profile, simplifying the subsequent crystallization or distillation steps required to achieve high-purity specifications.
How to Synthesize Perfluoroalkyl Substituted Aniline Efficiently
Implementing this synthesis route requires strict adherence to the sequential addition protocols and thermal profiles outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the reaction mixture under an inert atmosphere to exclude oxygen, followed by a critical pre-cooling step that manages the exotherm upon introduction of the gaseous or volatile perfluoroalkyl bromide. Detailed operational parameters, including specific stirring rates and pressure monitoring, are essential for scaling this chemistry from laboratory glassware to industrial autoclaves. The following guide summarizes the standardized operational procedure derived from the patent's most successful embodiments, providing a roadmap for process engineers to validate this technology in their own facilities.
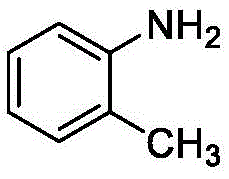
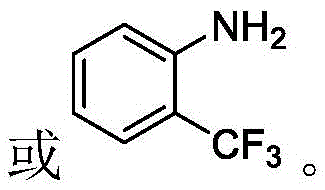
- Charge the reactor with acetonitrile solvent, o-aminotrifluorotoluene, sodium dithionite initiator, and ammonium chloride catalyst under nitrogen protection.
- Pre-cool the reaction mixture to -5°C before introducing 2-bromoheptafluoropropane gas into the pressure vessel.
- Heat the mixture to 60-70°C for 15 hours, then cool, add water to dissolve salts, separate layers, and extract the organic phase.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative economic benefits that extend beyond simple yield improvements. The primary driver of value is the substitution of raw materials; shifting from perfluoroisopropyl iodide to 2-bromoheptafluoropropane leverages a commodity chemical with a much broader global supply base, reducing vulnerability to price volatility and supply disruptions. This raw material swap inherently lowers the baseline cost of production without compromising the quality of the final intermediate. Additionally, the elimination of precious metal catalysts removes a significant line item from the bill of materials, as well as the associated costs of waste treatment and metal recovery. The simplified downstream processing, characterized by a straightforward aqueous wash and extraction, reduces solvent consumption and energy usage during purification, contributing to a leaner and more sustainable manufacturing footprint.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the use of ammonium chloride, a ubiquitous and low-cost industrial chemical, replacing expensive proprietary ligands and transition metals. This change drastically simplifies the supply chain for catalysts, as ammonium salts are available from multiple global suppliers, ensuring competitive pricing and reliable delivery. Furthermore, the high conversion rates achieved mean that less starting material is wasted, directly improving the material efficiency of the plant. The reduction in purification complexity also translates to lower utility costs, as fewer distillation columns or chromatography steps are required to meet purity specifications, resulting in substantial overall cost savings in perfluoroalkyl aniline manufacturing.
- Enhanced Supply Chain Reliability: Reliance on 2-bromoheptafluoropropane mitigates the risks associated with sourcing specialized fluorinated iodides, which often have limited production capacity and long lead times. By utilizing a bromide feedstock, manufacturers can tap into a more resilient supply network, ensuring continuous operation even during market fluctuations. The robustness of the reaction conditions, which tolerate a range of solvents including acetonitrile and glycol ethers, provides flexibility in sourcing solvents based on regional availability and price. This adaptability is crucial for maintaining production schedules and meeting the just-in-time delivery requirements of downstream agrochemical formulators, thereby strengthening the overall reliability of the supply chain for high-purity agrochemical intermediates.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard pressure reactors and avoiding hazardous reagents that would require specialized containment infrastructure. The absence of heavy metals in the catalyst system significantly reduces the environmental burden, simplifying wastewater treatment and solid waste disposal compliance. This aligns with increasingly stringent global environmental regulations, reducing the risk of regulatory shutdowns or fines. The ability to run the reaction in a single phase with simple workup procedures facilitates easy scale-up from pilot plants to multi-ton commercial production, allowing companies to rapidly respond to market demand for complex fluorine materials without extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on catalyst selection, reaction safety, and product quality. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: Why is ammonium chloride preferred over transition metal catalysts for this synthesis?
A: Ammonium chloride serves as a highly effective, inexpensive inorganic catalyst that eliminates the need for costly transition metals like zinc or nickel and complex phosphine ligands, significantly reducing raw material costs and simplifying downstream metal removal processes.
Q: What are the safety advantages of using 2-bromoheptafluoropropane over iodides?
A: 2-Bromoheptafluoropropane is significantly cheaper and more commercially available than perfluoroisopropyl iodide. Additionally, the reaction conditions described allow for high conversion rates without the extreme sensitivity often associated with iodide-based radical initiators.
Q: Can this process be scaled to multi-ton production?
A: Yes, the patent explicitly demonstrates the use of standard pressure vessels (autoclaves) and common solvents like acetonitrile. The single-phase reaction system and simple aqueous workup make it highly amenable to large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perfluoroalkyl Substituted Aniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient fluorine chemistry in the modern agrochemical landscape. Our technical team has extensively analyzed the capabilities presented in CN114394903A and is fully equipped to translate this intellectual property into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. Our facilities are outfitted with state-of-the-art pressure reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of perfluoroalkyl aniline delivered meets the highest industry standards for performance and consistency.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By leveraging our expertise in process development, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to request specific COA data and route feasibility assessments to verify the compatibility of this technology with your downstream applications. Together, we can drive down costs and enhance the reliability of your agrochemical intermediate supply, securing a competitive advantage in the global market.
