Advanced Bis-N-Substituted Benzimidazole Diamines: Bridging Thermal Stability and Solubility for Next-Gen Polyimides
The landscape of high-performance polymer engineering is constantly evolving, driven by the demand for materials that can withstand extreme thermal environments while remaining processable. Patent CN112778211A introduces a significant breakthrough in this domain with the development of a novel class of bis-N-substituted benzimidazole diamines. These specialized monomers address a critical paradox in polyimide synthesis: the trade-off between thermal stability and solubility. Traditional benzimidazole-based diamines offer exceptional heat resistance due to their rigid aromatic heterocyclic structures, but this same rigidity often leads to poor solubility, making processing and recycling difficult. The innovation detailed in this patent lies in the strategic introduction of side groups on the nitrogen atoms of the benzimidazole rings. By incorporating these N-substituents alongside flexible linking groups, the molecular architecture achieves a delicate balance. The resulting diamines retain the strong affinity and charge transfer effects of terminal amino groups, ensuring excellent thermodynamic performance, while the added steric hindrance from the side groups disrupts molecular packing. This structural ingenuity allows for the creation of polymeric materials that are not only heat-resistant but also soluble in common organic solvents, opening new avenues for applications in electronic materials and advanced coatings.
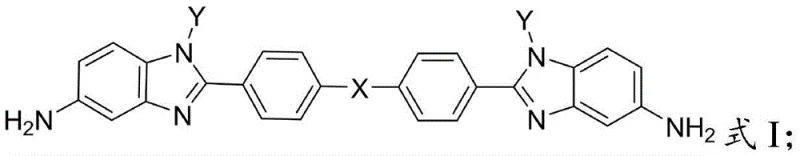
Understanding the limitations of conventional diamine monomers is essential for appreciating the value of this novel approach. Historically, the synthesis of polyimides relied heavily on rigid aromatic diamines which, while providing superior thermal stability, resulted in polymers with high linearity and ordered molecular arrangements. This high degree of order translates to insolubility in most organic solvents, creating significant bottlenecks in manufacturing processes such as film casting or fiber spinning. Engineers often faced the dilemma of sacrificing thermal performance to gain processability, or vice versa. Furthermore, traditional synthesis routes for modified diamines often involved complex multi-step procedures with low overall yields or required expensive, hard-to-source starting materials. The novel approach presented in the patent circumvents these issues by utilizing a modular synthetic strategy. It employs readily available nitro-aniline derivatives and diacid chlorides as building blocks. The key differentiator is the post-synthetic modification capability inherent in the design; by varying the 'X' linker (such as ether bonds or hexafluoroisopropylidene groups) and the 'Y' substituent (such as phenyl groups), manufacturers can fine-tune the polymer properties. This flexibility allows for the precise engineering of free volume within the polymer matrix, effectively breaking the rigidity that plagues conventional benzimidazole diamines without losing the core thermal benefits.
Mechanistic Insights into the Three-Step Synthesis Route
The synthesis of these advanced diamines follows a robust three-step pathway that is both chemically elegant and industrially viable. The process begins with a condensation reaction between an N-substituted nitro-aniline derivative and a diacid chloride. This step is critical as it establishes the central backbone of the molecule. The reaction is typically conducted at mild temperatures ranging from 0°C to 25°C in the presence of an acid-binding agent like triethylamine or pyridine. This controlled environment ensures the formation of the amide intermediate without degrading the sensitive functional groups. Following condensation, the intermediate undergoes a high-temperature ring-closing reaction. This cyclization step, performed at 180°C to 220°C in a high-boiling solvent such as butyrolactone, converts the linear amide structure into the fused benzimidazole ring system. The use of acidic catalysts facilitates this dehydration process, driving the equilibrium towards the desired heterocyclic product. Finally, the nitro groups on the benzimidazole rings are reduced to primary amines via catalytic hydrogenation. This final transformation is crucial for enabling subsequent polymerization. The use of heterogeneous catalysts like palladium on carbon allows for efficient reduction under moderate pressure (0.5-3 MPa) and temperature (40-100°C), ensuring high purity of the final diamine monomer.
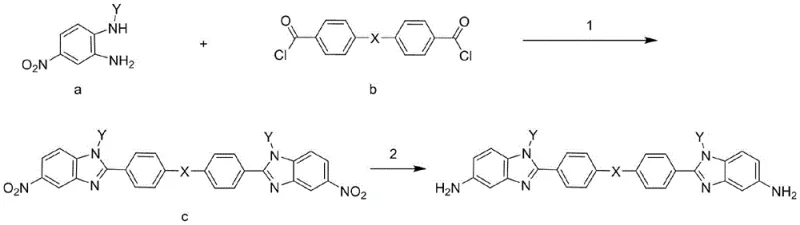
Impurity control is a paramount concern in the production of electronic-grade monomers, and this synthetic route offers inherent advantages in this regard. The stepwise nature of the synthesis allows for intermediate purification, particularly after the ring-closing step where recrystallization can be employed to remove unreacted starting materials or side products. The condensation reaction is monitored via TLC to ensure complete conversion before proceeding, minimizing the carryover of acid chlorides which could interfere with downstream polymerization. Furthermore, the catalytic hydrogenation step is highly selective for nitro group reduction, leaving the robust benzimidazole core and the N-substituents intact. This selectivity prevents the formation of over-reduced byproducts or ring-opening impurities that often plague less optimized routes. The final crystallization from the hydrogenation mixture at low temperatures (0°C) further enhances purity by precipitating the target diamine while keeping soluble impurities in the mother liquor. This rigorous control over the reaction pathway ensures that the resulting diamine meets the stringent quality standards required for high-performance polyimide applications, where even trace impurities can affect the dielectric properties or mechanical strength of the final film.
How to Synthesize Bis-N-Substituted Benzimidazole Diamine Efficiently
The preparation of these high-value monomers requires precise control over reaction parameters to maximize yield and purity. The patent outlines a standardized protocol that begins with the careful addition of diacid chloride to a cooled mixture of the amine precursor and base. Detailed operational guidelines regarding solvent selection, temperature ramping, and workup procedures are essential for reproducibility. For R&D teams looking to replicate or scale this chemistry, understanding the nuances of the cyclization step is particularly important, as the high temperatures involved require appropriate reactor specifications. The following guide summarizes the critical operational phases derived from the patent data, providing a roadmap for efficient laboratory and pilot-scale production.
- Condensation Reaction: Mix N-(2-amino-4-nitrophenyl)-N-aniline with a diacid chloride derivative and an acid-binding agent in a solvent like tetrahydrofuran at 0-25°C.
- Ring Closure: Heat the condensation product with an acidic catalyst in a high-boiling solvent (e.g., butyrolactone) at 180-220°C to form the nitro-benzimidazole intermediate.
- Catalytic Hydrogenation: Reduce the nitro groups to amino groups using a metal catalyst (Pd/C or Pt/C) under hydrogen pressure (0.5-3 MPa) at 40-100°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers compelling economic and logistical benefits. The reliance on commodity chemicals as starting materials significantly de-risks the supply chain. N-substituted anilines and terephthaloyl chloride derivatives are produced globally at large scales, ensuring a stable and competitive pricing structure. Unlike routes that depend on proprietary or scarce catalysts, this method utilizes standard acid-binding agents and recoverable heterogeneous hydrogenation catalysts. This accessibility translates directly into cost reduction in electronic chemical manufacturing, as raw material volatility is minimized. Furthermore, the process safety profile is robust; the reactions do not involve pyrophoric reagents or extreme cryogenic conditions, reducing the need for specialized containment infrastructure and lowering operational expenditures related to safety compliance.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts for C-N bond formation, replaced by a straightforward condensation-cyclization sequence, drastically simplifies the cost structure. The ability to recycle solvents like tetrahydrofuran and butyrolactone further enhances the economic viability. Additionally, the high yields reported in the examples (over 80% for the intermediate and over 90% for the final reduction) indicate a material-efficient process that minimizes waste disposal costs. By avoiding complex protection-deprotection strategies often seen in heterocyclic synthesis, the overall step count is kept low, which inherently reduces labor and utility consumption per kilogram of product.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for flexible sourcing of the 'X' and 'Y' components. If a specific diacid chloride faces supply constraints, the chemistry can be adapted to use alternative linkers without redesigning the entire process flow. This adaptability ensures continuity of supply for downstream polymer manufacturers. Moreover, the final hydrogenation step uses standard equipment found in most fine chemical facilities, meaning that contract manufacturing organizations (CMOs) can easily onboard this production without significant capital investment. This broadens the potential supplier base, reducing dependency on single-source vendors and mitigating geopolitical supply risks.
- Scalability and Environmental Compliance: The process is designed with scalability in mind. The exothermic condensation reaction is managed through controlled dosing, a technique easily translated from gram to ton scale. The high-temperature cyclization is a batch operation that fits well within existing reactor trains. From an environmental standpoint, the atom economy is favorable, and the primary byproduct is salt from the acid-binding agent, which is easily handled in wastewater treatment systems. The use of heterogeneous catalysts eliminates heavy metal contamination in the final product, simplifying purification and reducing the environmental footprint associated with metal scavenging resins. This alignment with green chemistry principles facilitates regulatory approval in markets with strict environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this technology. These answers are derived directly from the experimental data and technical disclosures within the patent, providing clarity on performance metrics and process capabilities. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating these monomers into their existing product lines.
Q: How does N-substitution improve polyimide solubility?
A: The introduction of N-substituted groups on the benzimidazole ring increases the steric hindrance and disrupts the regular packing of polymer chains. This structural modification prevents excessive crystallization, thereby significantly enhancing solubility in organic solvents without compromising the inherent thermal stability provided by the rigid benzimidazole backbone.
Q: What are the key reaction conditions for the cyclization step?
A: The ring-closing reaction requires high temperatures, typically between 180°C and 220°C, to drive the dehydration cyclization effectively. Acidic catalysts such as p-toluenesulfonic acid or concentrated hydrochloric acid are used in high-boiling solvents like butyrolactone or sulfolane to ensure complete conversion to the benzimidazole structure.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process utilizes commercially available raw materials and standard unit operations such as condensation, high-temperature cyclization, and catalytic hydrogenation. The absence of exotic reagents and the use of recoverable heterogeneous catalysts make the route highly suitable for large-scale commercial manufacturing with robust safety profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-N-Substituted Benzimidazole Diamine Supplier
The technical potential of bis-N-substituted benzimidazole diamines is undeniable, offering a pathway to next-generation polyimides that defy the traditional compromises between heat resistance and processability. However, translating patent chemistry into commercial reality requires a partner with deep expertise in process development and scale-up. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific thermal and pressure requirements of the cyclization and hydrogenation steps described in the patent. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of monomer meets the exacting standards required for electronic grade applications, guaranteeing consistent performance in your final polymer products.
We invite you to explore how this innovative chemistry can enhance your product portfolio. Our technical team is ready to collaborate on customizing the 'X' and 'Y' substituents to meet your specific thermal and solubility targets. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how we can become your strategic partner in delivering high-performance polymer solutions efficiently and reliably.
