Industrial Scale-Up of Shogaol Intermediates: A Breakthrough in Room Temperature Catalysis and Yield Optimization
Introduction to Advanced Shogaol Intermediate Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for bioactive compounds that balance high purity with economic viability. Patent CN108191625B introduces a transformative preparation method for (E)-1-(4-hydroxy-3-methoxyphenyl)-4-en-3-decanone, commonly known as Shogaol, a compound renowned for its antipyretic, analgesic, and antitussive properties. This innovation addresses critical bottlenecks in traditional synthesis by replacing hazardous cryogenic conditions with a mild, room-temperature protocol that utilizes inexpensive inorganic bases. For R&D directors and procurement specialists, this represents a pivotal shift towards safer, more scalable pharmaceutical intermediates manufacturing. The structural integrity of the target molecule, characterized by its specific alkene geometry and phenolic functionality, is preserved through a novel enamine-mediated mechanism that avoids the harsh degradation often seen in older methodologies.
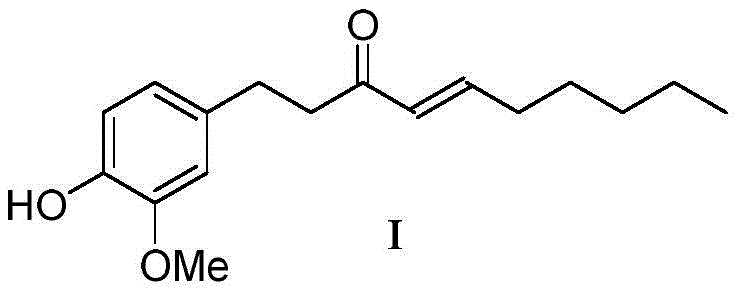
Furthermore, the reported total yield exceeding 78 percent marks a substantial improvement over prior art, which typically struggles to achieve half that efficiency. By operating at ambient temperatures and employing readily available solvents like dichloromethane, this process drastically lowers the barrier to entry for commercial scale-up. The method not only simplifies the operational workflow but also aligns with modern green chemistry principles by minimizing the use of heavy metal catalysts and extreme thermal inputs. As a reliable fine chemical intermediates supplier, understanding these mechanistic nuances is essential for evaluating the long-term viability of supply chains dependent on ginger-derived bioactives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Shogaol has been plagued by complex multi-step sequences that demand rigorous environmental controls and expensive reagents. As illustrated in previous literature, such as the route reported in Food Chemistry (2014), the process necessitates the use of n-butyllithium and LDA at cryogenic temperatures of -78°C to facilitate condensation with n-hexanal. This reliance on pyrophoric reagents introduces significant safety hazards and requires specialized infrastructure that many manufacturing facilities lack. Moreover, subsequent steps involving Pd/C hydrogenation and acidic elimination add further complexity, increasing the risk of side reactions and lowering the overall atom economy. Another approach disclosed in EP1506958A1 utilizes a palladium-catalyzed oxidation and tosyl protection strategy, yet it suffers from a dismal total yield of only 41 percent, rendering it economically unfeasible for large-scale production.
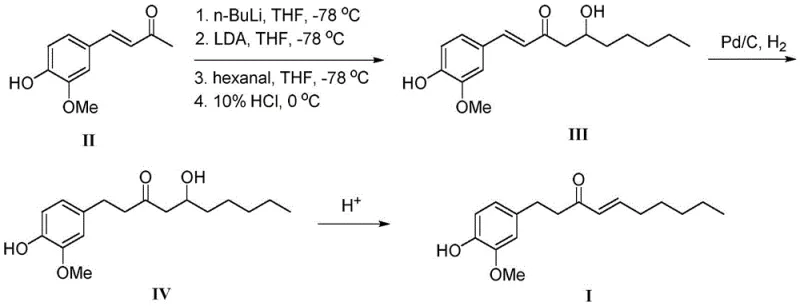
Additionally, Wittig reaction-based strategies, while chemically elegant, often involve cumbersome protection and deprotection steps, such as the use of TBDMS groups, which generate substantial chemical waste. These conventional pathways are not only capital intensive due to the need for low-temperature reactors but also operationally fragile, where minor deviations in temperature or stoichiometry can lead to batch failures. The cumulative effect of these limitations is a supply chain that is vulnerable to disruptions and characterized by high production costs, making the final API intermediates prohibitively expensive for broader therapeutic applications. The industry urgently requires a paradigm shift away from these archaic, high-risk protocols toward more resilient manufacturing technologies.
The Novel Approach
The methodology outlined in CN108191625B offers a decisive break from these constraints by leveraging a room-temperature enamine condensation strategy. Instead of relying on strong organometallic bases, the process utilizes weak organic amines like tetrahydropyrrole or piperidine to activate the ketone precursor, 4-(4-hydroxy-3-methoxyphenyl)-2-butanone. This activation allows for a smooth condensation with n-hexanal in a halogenated hydrocarbon solvent, proceeding efficiently without the need for thermal extremes. The subsequent hydrolysis step employs simple inorganic bases such as sodium hydroxide or potassium carbonate, which are not only cost-effective but also easier to handle and dispose of compared to their hazardous counterparts. This streamlined approach collapses multiple synthetic operations into a cohesive flow, significantly reducing the processing time and equipment footprint required for production.
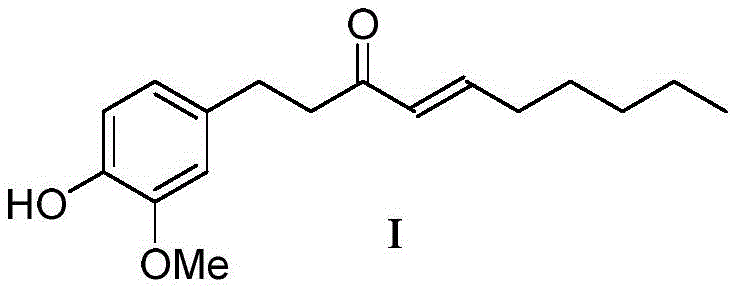
Crucially, this novel route achieves a total yield of over 78 percent, nearly doubling the efficiency of the best-performing prior art methods. The mild reaction conditions preserve the sensitive functional groups on the aromatic ring, minimizing the formation of polymeric byproducts and ensuring a cleaner crude profile. For procurement managers, this translates directly into cost reduction in pharmaceutical intermediates manufacturing through improved material throughput and reduced waste treatment expenses. The simplicity of the workup, involving standard phase separation and acidification, further enhances the operational appeal, making it an ideal candidate for transfer from laboratory bench to multi-ton commercial reactors. This approach exemplifies how strategic reagent selection can unlock substantial value in established chemical syntheses.
Mechanistic Insights into Enamine-Mediated Aldol Condensation
The core innovation of this synthesis lies in the formation and reactivity of the enamine intermediate, which serves as a soft nucleophile in the carbon-carbon bond-forming step. Unlike hard enolates generated by strong bases like LDA, which can lead to uncontrolled polymerization or self-condensation of the aldehyde, the enamine derived from tetrahydropyrrole offers superior regioselectivity and kinetic control. The nitrogen atom of the amine donates electron density into the pi-system of the ketone, activating the alpha-position for nucleophilic attack on the electrophilic carbonyl carbon of n-hexanal. This mechanism proceeds smoothly at room temperature because the energy barrier for the enamine-aldehyde addition is significantly lower than that for direct enolate alkylation under non-cryogenic conditions. The resulting iminium ion intermediate is then susceptible to hydrolysis, regenerating the amine catalyst and releasing the beta-hydroxy ketone precursor.
Following the condensation, the inorganic base plays a dual role in facilitating the final elimination to form the alpha,beta-unsaturated ketone system characteristic of Shogaol. The base promotes the dehydration of the beta-hydroxy intermediate, driving the equilibrium toward the conjugated enone product which is thermodynamically stable. This elimination step is critical for establishing the desired (E)-geometry of the double bond, which is essential for the biological activity of the final molecule. The use of aqueous inorganic base ensures that the reaction medium remains heterogeneous or biphasic, which can help in driving the reaction to completion by sequestering water or byproducts. Understanding this mechanistic pathway allows R&D teams to fine-tune parameters such as amine loading and base concentration to optimize the impurity profile, ensuring that the final product meets the stringent specifications required for high-purity pharmaceutical intermediates.
How to Synthesize (E)-1-(4-hydroxy-3-methoxyphenyl)-4-en-3-decanone Efficiently
The practical execution of this synthesis begins with the preparation of the key ketone precursor, 4-(4-hydroxy-3-methoxyphenyl)-2-butanone, via a classic aldol condensation of vanillin and acetone followed by selective hydrogenation. Once this precursor is secured, the novel protocol dictates dissolving it in a halogenated solvent such as dichloromethane and treating it with a slight excess of tetrahydropyrrole or piperidine at ambient temperature. After a brief incubation period to allow for enamine formation, a solution of n-hexanal is introduced slowly to manage the exotherm and maintain selectivity. The detailed standardized synthesis steps, including precise stoichiometric ratios and workup procedures, are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Preparation of 4-(4-hydroxy-3-methoxyphenyl)-2-butanone via aldol condensation of vanillin and acetone followed by Pd/C hydrogenation.
- Formation of an enamine intermediate by reacting the ketone with tetrahydropyrrole or piperidine in a halogenated hydrocarbon solvent at room temperature.
- Condensation with n-hexanal followed by hydrolysis using an inorganic base solution to yield the final target compound with over 78% total yield.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders, the transition to this room-temperature synthesis model offers profound strategic benefits that extend beyond simple yield metrics. The elimination of cryogenic cooling requirements removes a major bottleneck in batch processing, allowing for faster turnover times and higher utilization rates of existing reactor assets. Furthermore, the substitution of hazardous pyrophoric reagents with stable inorganic bases significantly lowers the regulatory burden and insurance costs associated with chemical storage and handling. This shift enhances the overall resilience of the supply chain by reducing dependency on specialized reagent suppliers and mitigating the risk of production stoppages due to safety incidents. Consequently, manufacturers can achieve substantial cost savings through both direct operational efficiencies and indirect risk mitigation measures.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic simplification of the reaction conditions and the improvement in material efficiency. By avoiding the use of expensive organolithium reagents and palladium catalysts, the direct material costs are significantly lowered, while the higher yield ensures that less raw material is wasted per kilogram of finished product. Additionally, the ability to run reactions at room temperature eliminates the substantial energy costs associated with maintaining -78°C environments, leading to a leaner cost structure. These factors combine to create a highly competitive pricing model for the final intermediate, providing a distinct market advantage.
- Enhanced Supply Chain Reliability: Operational stability is greatly improved by the robustness of the new method, which tolerates a wider range of operating conditions compared to the finicky cryogenic alternatives. The use of commodity chemicals like sodium hydroxide and common solvents ensures that raw material availability is rarely a constraint, safeguarding against supply disruptions. This reliability allows for more accurate forecasting and inventory planning, enabling procurement teams to secure long-term contracts with greater confidence. The simplified process flow also reduces the likelihood of batch failures, ensuring a consistent and uninterrupted flow of goods to downstream customers.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in hazardous waste generation and the avoidance of heavy metal catalysts simplify the effluent treatment process, facilitating compliance with increasingly strict environmental regulations. The process is inherently scalable, as the heat management requirements are minimal, allowing for safe expansion from pilot plant to full commercial production volumes. This scalability ensures that the technology can meet growing market demand without requiring prohibitive capital investment in new infrastructure. Ultimately, this positions the manufacturer as a sustainable partner capable of delivering commercial scale-up of complex pharmaceutical intermediates with a reduced environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own production lines or for sourcing strategies.
Q: What are the primary advantages of the room temperature synthesis method for Shogaol?
A: The primary advantage is the elimination of cryogenic conditions (-78°C) and strong bases like n-BuLi, which significantly reduces energy consumption and safety risks while improving total yield to over 78%.
Q: How does this method improve supply chain reliability for pharmaceutical intermediates?
A: By utilizing common inorganic bases and ambient temperature reactions, the process avoids reliance on specialized cryogenic equipment and hazardous pyrophoric reagents, ensuring more consistent production schedules and lower operational costs.
Q: Is the purity of the final product suitable for pharmaceutical applications?
A: Yes, the described embodiment achieved a purity of 95.34% by HPLC after standard workup, indicating that the method produces high-purity intermediates suitable for downstream pharmaceutical processing without complex purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Shogaol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern pharmaceutical landscape. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the room-temperature Shogaol synthesis can be seamlessly transferred to industrial reality. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of intermediate meets the highest global standards. Our capability to handle complex organic transformations allows us to offer flexible manufacturing solutions tailored to the specific needs of our international partners.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis specific to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can drive value and efficiency in your operations. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing.
