Advanced Catalytic Synthesis of 2,4-Dichloroquinazoline Derivatives for Commercial Scale-up
Advanced Catalytic Synthesis of 2,4-Dichloroquinazoline Derivatives for Commercial Scale-up
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical heterocyclic intermediates, particularly those serving as the backbone for alpha-1 adrenergic receptor antagonists. A pivotal advancement in this domain is detailed in Chinese Patent CN102584721B, which discloses a novel synthetic method for 2,4-dichloroquinazoline derivatives. This technology represents a significant departure from conventional high-temperature and high-pressure methodologies, introducing a milder, catalytic approach that utilizes triphenylphosphine oxide and triphosgene. For R&D directors and procurement managers alike, this patent offers a compelling value proposition by addressing long-standing issues regarding yield optimization, operational safety, and environmental compliance in the manufacturing of quinazoline-based active pharmaceutical ingredients (APIs).
The core innovation lies in the strategic activation of bis(trichloromethyl)carbonate (triphosgene) under alkaline conditions, facilitated by a specific catalytic system. Unlike traditional routes that often suffer from harsh reaction conditions and poor atom economy, this method operates at moderate temperatures ranging from 100°C to 130°C. The ability to synthesize key intermediates such as 6,7-dimethoxy-2,4-dichloroquinazoline with high purity and yield makes this technology particularly attractive for the production of antihypertensive drugs like prazosin and alfuzosin. As a reliable pharmaceutical intermediates supplier, understanding the nuances of such proprietary synthetic pathways is essential for ensuring supply chain continuity and cost-effectiveness.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,4-dichloroquinazoline derivatives has been plagued by significant technical and safety hurdles that impede efficient commercial scale-up. Prior art methods, such as those described in CN101747323A, typically involve the condensation of 2-amino-4,5-dimethoxybenzonitrile with DMF followed by chlorination. These processes often necessitate reaction temperatures exceeding 190°C within high-pressure autoclaves, creating substantial safety risks and energy consumption burdens. Furthermore, alternative routes utilizing veratraldehyde require multiple steps involving toxic reagents like sodium cyanate and phosphorus oxychloride (POCl3). The use of POCl3 is particularly problematic from an environmental standpoint, as it generates large volumes of acidic wastewater that require complex and costly treatment protocols before discharge.
Another reported method by Lee et al. involves the reaction of o-aminobenzonitrile with diphosgene. While this route achieves reasonable yields, it relies on diphosgene, a substance with extreme toxicity that poses severe health hazards to plant personnel. Additionally, this method often requires pressurized conditions and elevated temperatures, limiting its applicability in standard glass-lined reactors commonly found in fine chemical manufacturing facilities. The cumulative effect of these limitations is a production process characterized by high operational expenditures, significant safety liabilities, and a challenging regulatory landscape regarding waste management and worker protection.
The Novel Approach
The methodology presented in patent CN102584721B effectively circumvents these historical bottlenecks by employing a triphosgene-based cyclization strategy mediated by a triphenylphosphine oxide catalyst. This approach allows for the direct conversion of readily available starting materials, such as o-aminobenzonitriles or 3-(oximino)indolin-2-ones, into the desired dichloroquinazoline scaffold. The reaction conditions are markedly milder, typically proceeding at atmospheric pressure and temperatures between 100°C and 130°C, which eliminates the need for specialized high-pressure equipment. This shift not only enhances operational safety but also simplifies the engineering requirements for reactor design, thereby facilitating easier technology transfer and scale-up.
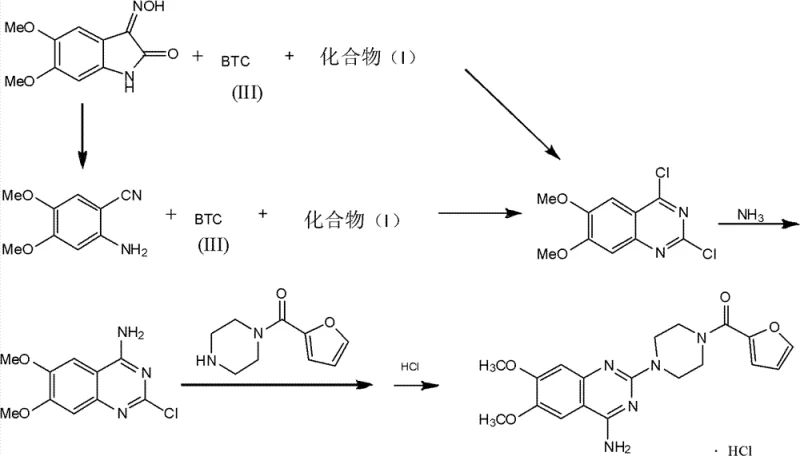
Moreover, the versatility of this synthetic route is demonstrated by its applicability to a wide range of substituted derivatives, including those with nitro, fluoro, chloro, and methoxy substituents. This flexibility is crucial for manufacturers aiming to produce a portfolio of related APIs, such as terazosin and doxazosin, from a common platform technology. By streamlining the synthesis into fewer steps and avoiding the use of highly toxic gaseous reagents, this novel approach offers a more sustainable and economically viable pathway for cost reduction in API manufacturing. The resulting intermediates exhibit high purity profiles, reducing the burden on downstream purification processes and ensuring consistent quality for final drug products.
Mechanistic Insights into Triphosgene-Mediated Cyclization
The mechanistic elegance of this synthesis relies on the in situ generation of reactive chlorinating species from triphosgene, activated by the triphenylphosphine oxide and tertiary amine catalytic system. In the initial phase, the tertiary amine, such as triethylamine, interacts with triphosgene to generate a reactive complex that facilitates the chlorination of the substrate. The triphenylphosphine oxide acts as a crucial promoter, likely stabilizing transition states or enhancing the electrophilicity of the carbonyl carbon in the triphosgene molecule. This cooperative catalysis ensures that the chlorination proceeds efficiently without the need for excessive thermal energy or harsh acidic conditions typically associated with POCl3-mediated reactions.
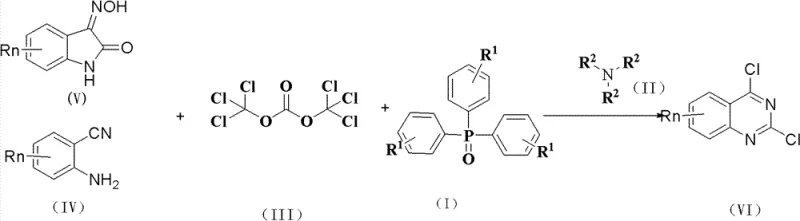
From an impurity control perspective, this mechanism offers distinct advantages over traditional methods. The mild reaction environment minimizes the formation of thermal degradation byproducts and polymerization impurities that often contaminate batches produced at temperatures above 190°C. Furthermore, the stoichiometric control afforded by using solid triphosgene allows for precise management of the chlorination degree, reducing the risk of over-chlorination or incomplete reaction. The subsequent workup procedure, which involves aqueous quenching and solvent extraction, effectively removes polar byproducts and residual catalysts. The ability to recover and recycle the triphenylphosphine oxide catalyst further underscores the efficiency of this mechanism, as it prevents the accumulation of phosphorus-containing impurities in the final product stream.
How to Synthesize 2,4-Dichloroquinazoline Efficiently
Implementing this synthesis requires careful attention to the order of addition and temperature control to maximize yield and safety. The process begins with the preparation of the catalytic mixture in a suitable organic solvent, followed by the controlled addition of the chlorinating agent. Detailed standard operating procedures regarding specific molar ratios, solvent choices, and purification techniques are critical for reproducibility. For a comprehensive guide on the exact experimental parameters and step-by-step execution of this synthesis, please refer to the standardized protocol outlined below.
- Prepare the catalytic system by dissolving triphenylphosphine oxide and a tertiary amine catalyst in an organic solvent such as chlorobenzene under cooling conditions.
- Slowly add a solution of bis(trichloromethyl)carbonate (triphosgene) to the mixture, maintaining low temperature initially before warming to activate the chlorinating species.
- Introduce the substrate (o-aminobenzonitrile or 3-(oximino)indolin-2-one), heat the reaction to 100-130°C, and purify the final product via aqueous workup and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology translates into tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the substantial optimization of the cost structure associated with producing high-purity pharmaceutical intermediates. By eliminating the need for expensive and hazardous reagents like sodium cyanate and phosphorus oxychloride, the raw material costs are significantly reduced. Furthermore, the recovery of the triphenylphosphine oxide catalyst, with reported efficiencies reaching over 90%, means that the effective consumption of this valuable ligand is minimal, leading to drastic savings in material costs over large-scale production campaigns.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the elimination of costly waste treatment associated with acidic effluents from POCl3 usage. Traditional chlorination methods generate vast amounts of phosphoric acid waste, requiring neutralization and disposal, which adds considerable overhead to the manufacturing budget. In contrast, the triphosgene-based route produces cleaner waste streams that are easier and cheaper to manage. Additionally, the operation at lower temperatures reduces energy consumption for heating and cooling, contributing to a lower overall carbon footprint and utility expenditure for the production facility.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable starting materials ensures a robust supply chain that is less susceptible to market volatility. Reagents such as o-aminobenzonitrile and triphosgene are commercially available in bulk quantities from multiple global suppliers, mitigating the risk of single-source dependency. The simplified reaction setup, which does not require specialized high-pressure autoclaves, allows for production to be distributed across a wider network of contract manufacturing organizations (CMOs), thereby increasing supply resilience and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The inherent safety of using solid triphosgene instead of gaseous phosgene or liquid POCl3 simplifies regulatory compliance and permits easier scaling from pilot to commercial production. Facilities can scale up batch sizes without encountering the exponential increase in safety risks associated with high-pressure gas handling. This scalability ensures that the method is suitable for large-scale industrial production, capable of meeting the demands of global markets for alpha-blocker medications while adhering to increasingly stringent environmental, health, and safety (EHS) regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on its practical application in an industrial setting. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary safety advantages of this triphosgene method over traditional POCl3 routes?
A: Traditional methods often rely on phosphorus oxychloride (POCl3) or toxic sodium cyanate, which generate significant hazardous waste and require stringent handling protocols. This patented method utilizes triphosgene, a solid crystalline reagent that is easier to handle and transport than gaseous phosgene, significantly reducing operational risks and environmental burden.
Q: Can this synthesis method be applied to diverse quinazoline derivatives beyond the parent compound?
A: Yes, the method demonstrates broad substrate scope. The patent explicitly details successful synthesis of various substituted derivatives including 6,7-dimethoxy, 7-fluoro, 6-nitro, and 6-methyl variants, making it highly versatile for producing intermediates for drugs like prazosin, terazosin, and doxazosin.
Q: How does the catalyst recovery process impact the overall production cost?
A: The process includes a specific workup step where the triphenylphosphine oxide catalyst precipitates upon addition of petroleum ether. The patent reports a catalyst recovery rate of approximately 92.5%, which drastically lowers the effective cost of goods sold by minimizing the consumption of expensive phosphine ligands.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Dichloroquinazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative methods like the triphosgene-mediated cyclization are translated into reliable supply solutions. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages tailored to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and compliant chemical strategy available.
