Advancing Organic Electronics via Site-Selective DHR Functionalization and Commercial Scalability
Advancing Organic Electronics via Site-Selective DHR Functionalization and Commercial Scalability
The rapid evolution of the organic electronics sector demands precise molecular engineering to enhance charge mobility and stability in semiconductor materials. Patent CN114516779A introduces a groundbreaking methodology for the site-selective functionalization of Dicycloheptatrienorubicin (DHR), a novel non-benzenoid conjugated skeleton. This innovation addresses a critical bottleneck in the synthesis of advanced optoelectronic materials by providing controlled access to C-2, C-6, and C-7 positions on the DHR core. For R&D directors and procurement specialists, this represents a significant leap forward, offering a reliable pathway to synthesize high-purity OLED material precursors with tailored electronic properties. The ability to selectively install bromine or boronate groups transforms DHR from a static scaffold into a dynamic building block for next-generation displays and sensors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the functionalization of complex polycyclic aromatic hydrocarbons (PAHs) like DHR has been plagued by poor regioselectivity and harsh reaction conditions. Conventional electrophilic aromatic substitution often results in statistical mixtures of isomers, necessitating tedious and yield-depleting separation processes that drive up costs. Furthermore, the unique 5/6/7 ring system of DHR presents steric and electronic challenges that standard reagents fail to navigate effectively, often leading to decomposition or over-reaction. For supply chain managers, these inefficiencies translate into unpredictable lead times and inconsistent batch quality, hindering the commercial scale-up of complex organic semiconductors. The lack of specific handles for cross-coupling reactions has historically limited the structural diversity achievable with the DHR core, restricting its potential application in high-performance devices.
The Novel Approach
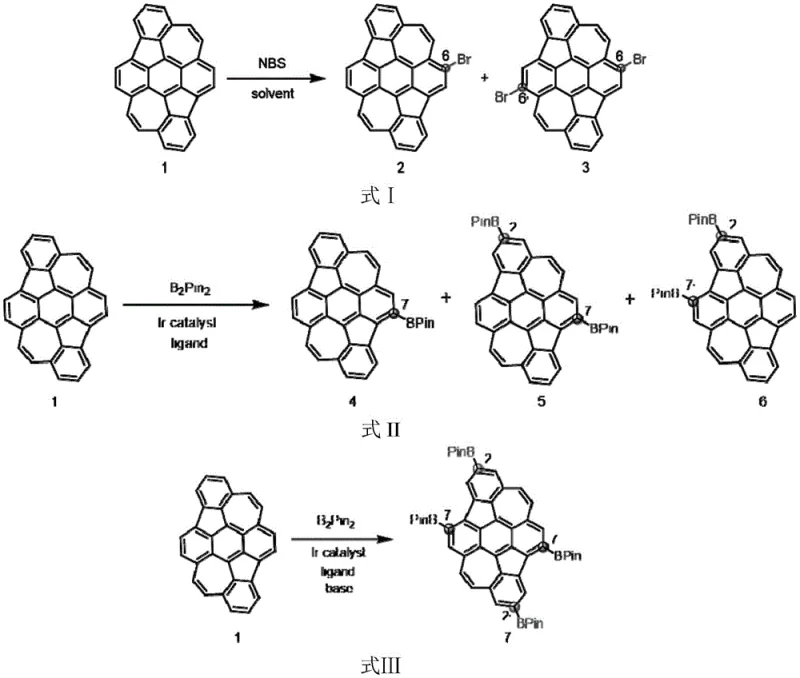
The patented methodology overcomes these hurdles through a sophisticated combination of transition metal catalysis and precise condition control. By employing N-bromosuccinimide (NBS) under optimized thermal conditions, the process achieves exclusive C-6 selective bromination, creating a pristine platform for further modification. More impressively, the utilization of Iridium-catalyzed C-H borylation allows for direct functionalization at the C-7 and C-2 positions without pre-existing directing groups. This dual strategy of halogenation and borylation provides a comprehensive toolkit for molecular architects. As illustrated in the reaction schemes below, this approach not only simplifies the synthetic route but also drastically improves the overall atom economy and purity profile of the resulting intermediates.
Mechanistic Insights into Ir-Catalyzed C-H Borylation and Pd-Cross Coupling
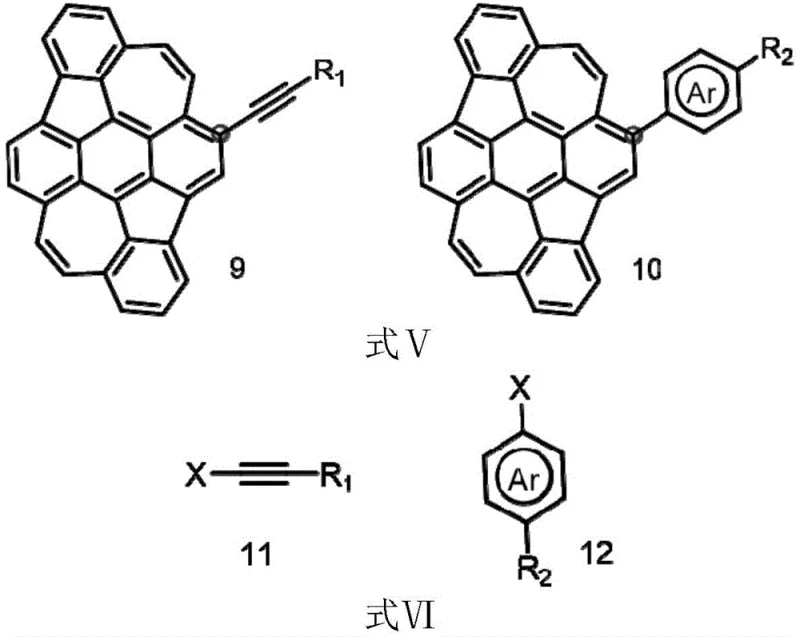
The core of this technological advancement lies in the mechanistic precision of the catalytic cycles employed. The Iridium-catalyzed borylation utilizes a sterically demanding ligand environment, typically involving bipyridine derivatives, to direct the metal center towards the less hindered yet electronically activated C-H bonds at the 2 and 7 positions. This C-H activation step bypasses the need for pre-functionalized substrates, representing a significant cost reduction in electronic chemical manufacturing by eliminating synthetic steps. The subsequent transmetallation with bis(pinacolato)diboron ensures the stable installation of the boronate ester, a crucial functionality for Suzuki-Miyaura coupling. This mechanism is robust enough to tolerate the extended conjugation of the DHR system without disrupting its planarity, which is essential for maintaining the p-type semiconductor characteristics and hole mobility of the final material.
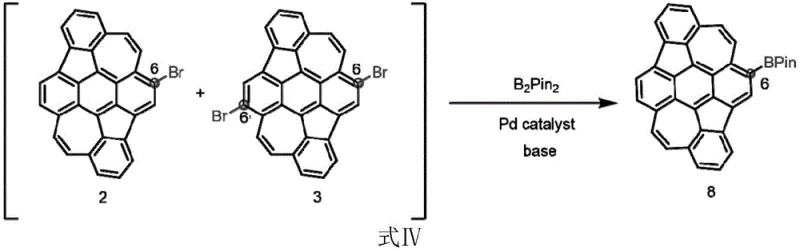
Furthermore, the patent details a seamless transition to Palladium-catalyzed cross-coupling for derivative synthesis. Once the C-6 bromide or boronate is established, it serves as an electrophilic or nucleophilic partner in coupling reactions with aryl halides or alkynes. The use of standard Pd catalysts like Pd(dba)2 or Pd(PPh3)4 in conjunction with mild bases facilitates the formation of C-C bonds under relatively mild temperatures. This modularity allows for the introduction of diverse functional groups, such as carbazole or silyl-protected alkynes, which can fine-tune the HOMO-LUMO energy levels. For technical teams, understanding this mechanism is vital for troubleshooting and optimizing reaction parameters to minimize residual metal impurities, ensuring the final product meets the stringent purity specifications required for commercial display applications.
How to Synthesize Site-Selective Borylated DHR Efficiently
Executing this synthesis requires careful attention to catalyst loading and atmospheric control to maximize yield and selectivity. The process begins with the preparation of the DHR substrate, followed by the addition of the boron source and the specific metal catalyst system under an inert nitrogen atmosphere. Temperature control is critical, with reactions typically proceeding between 120°C and 130°C to activate the C-H bonds without degrading the sensitive conjugated core. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and workup procedures necessary to isolate the target boronate esters in high purity.
- Prepare the reaction vessel with DHR substrate and Bis(pinacolato)diboron under inert atmosphere.
- Add the specific Iridium catalyst (e.g., [Ir(OMe)(cod)]2) and ligand (e.g., dtbpy) for C-H activation, or Pd catalyst for halide conversion.
- Heat the mixture in an appropriate solvent (e.g., dioxane or THF) at 120-130°C, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patent offers substantial advantages that directly impact the bottom line and supply chain resilience. The shift from multi-step functionalization to direct C-H activation significantly reduces the number of unit operations required, leading to lower energy consumption and reduced solvent waste. This streamlining of the process inherently lowers the cost of goods sold (COGS), making high-performance DHR derivatives more accessible for mass-market electronic applications. Additionally, the use of commercially available reagents like NBS and pinacolborane ensures that raw material sourcing remains stable and unaffected by niche supply constraints. For procurement managers, this translates to a more predictable pricing structure and reduced risk of production stoppages due to reagent shortages.
- Cost Reduction in Manufacturing: The elimination of pre-functionalization steps and the high selectivity of the catalytic systems remove the need for expensive chromatographic separations of isomeric byproducts. By achieving high regioselectivity directly, the process minimizes material loss during purification, thereby enhancing the overall yield per batch. This efficiency gain allows manufacturers to produce larger quantities of active pharmaceutical ingredients or electronic intermediates with the same infrastructure investment. The qualitative reduction in waste generation also aligns with increasingly strict environmental regulations, avoiding potential fines and remediation costs associated with hazardous waste disposal.
- Enhanced Supply Chain Reliability: The robustness of the described catalytic systems means that the synthesis is less sensitive to minor fluctuations in reaction conditions, ensuring consistent batch-to-batch quality. This reliability is crucial for long-term supply contracts with major display manufacturers who require uninterrupted material flow. Furthermore, the versatility of the intermediates allows for a "platform chemistry" approach, where a single stock of borylated DHR can be rapidly converted into various end-products based on market demand. This flexibility reduces inventory holding costs and improves the responsiveness of the supply chain to emerging trends in the organic electronics sector.
- Scalability and Environmental Compliance: The reaction conditions described, utilizing standard organic solvents and manageable temperatures, are readily transferable from laboratory scale to pilot and commercial production scales. The absence of exotic or highly toxic reagents simplifies the safety protocols required for large-scale operations. Moreover, the high atom economy of the C-H borylation reaction contributes to a greener manufacturing footprint, supporting corporate sustainability goals. This ease of scale-up ensures that as demand for DHR-based OLED materials grows, production capacity can be expanded without requiring fundamental changes to the process technology or significant capital expenditure on specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this DHR functionalization technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and benefits of adopting this synthetic route. Understanding these aspects is essential for stakeholders evaluating the integration of these intermediates into their existing product pipelines or R&D portfolios.
Q: What distinguishes this DHR functionalization method from traditional electrophilic substitution?
A: Unlike traditional methods that often yield mixtures, this patent utilizes Ir-catalyzed C-H activation to achieve precise site-selectivity at the C-2 and C-7 positions, significantly reducing impurity profiles.
Q: Can these intermediates be scaled for commercial OLED production?
A: Yes, the described protocols use robust catalytic systems and standard workup procedures like filtration and chromatography, which are amenable to kilogram-scale manufacturing for electronic chemicals.
Q: How does the bromination step facilitate further derivatization?
A: The C-6 selective bromination creates a versatile handle for subsequent Pd-catalyzed Suzuki-Miyaura couplings, allowing for the attachment of diverse aryl or alkynyl groups to tune optoelectronic properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dicycloheptatrienorubicin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the DHR scaffold in the realm of organic optoelectronics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate catalytic processes described in CN114516779A can be successfully translated to industrial volumes. We are committed to delivering high-purity DHR derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to manage complex organometallic chemistry guarantees a consistent supply of critical intermediates for your display and semiconductor projects.
We invite you to collaborate with us to leverage these advanced synthetic methodologies for your specific application needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can accelerate your time-to-market while reducing overall production costs. Let us be your partner in bridging the gap between innovative patent chemistry and commercial reality.
