Advanced Boron-Mediated Synthesis Of Quinoline Derivatives For Commercial Scale-Up Of Complex Antibacterial Agents
The pharmaceutical industry continuously seeks robust synthetic routes for critical antibacterial agents, particularly fluoroquinolone derivatives which serve as essential scaffolds for modern antibiotics. Patent CN1025028C introduces a transformative methodology for preparing quinoline carboxylic acid derivatives, specifically targeting the 1-substituted-7-piperazinyl-6-fluoro-4-oxo-1,4-dihydroquinoline-3-carboxylic acid structure. This innovation addresses longstanding challenges in regioselectivity and yield that have historically plagued the manufacturing of these high-value API intermediates. By leveraging a unique boron-complexed intermediate strategy, the disclosed process enables the efficient conversion of precursors into therapeutically active compounds with exceptional purity profiles. For R&D directors and procurement specialists, understanding this technology is vital for securing a reliable API intermediate supplier capable of delivering consistent quality. The method described eliminates the need for harsh halogen exchange conditions often associated with traditional routes, thereby reducing the formation of difficult-to-remove impurities. This technical breakthrough not only streamlines the synthetic pathway but also aligns with modern green chemistry principles by optimizing atom economy and reducing waste generation during the critical substitution steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of 7-piperazinyl quinolones typically involves the direct nucleophilic displacement of a leaving group, such as chlorine or fluorine, on a fully formed quinolone ring system. However, this conventional approach frequently suffers from significant drawbacks, including poor regioselectivity and the formation of multiple byproducts due to the competing reactivity of the 3-carboxyl and 4-keto functionalities. In many historical processes, the harsh conditions required to drive the substitution reaction can lead to degradation of the sensitive quinolone core, resulting in lower overall yields and necessitating extensive downstream purification efforts. Furthermore, the use of unprotected intermediates often requires stoichiometric amounts of expensive reagents to push the equilibrium, driving up the cost reduction in pharmaceutical manufacturing. The presence of residual halogens and metal catalysts in the final product can also pose serious regulatory hurdles, requiring additional processing steps to meet stringent pharmacopeial standards. These inefficiencies create bottlenecks in the supply chain, extending lead times and increasing the risk of batch failures during commercial production.
The Novel Approach
In stark contrast, the methodology outlined in CN1025028C employs a sophisticated boron-chelation strategy to mask the reactive sites of the quinolone nucleus prior to the substitution step. 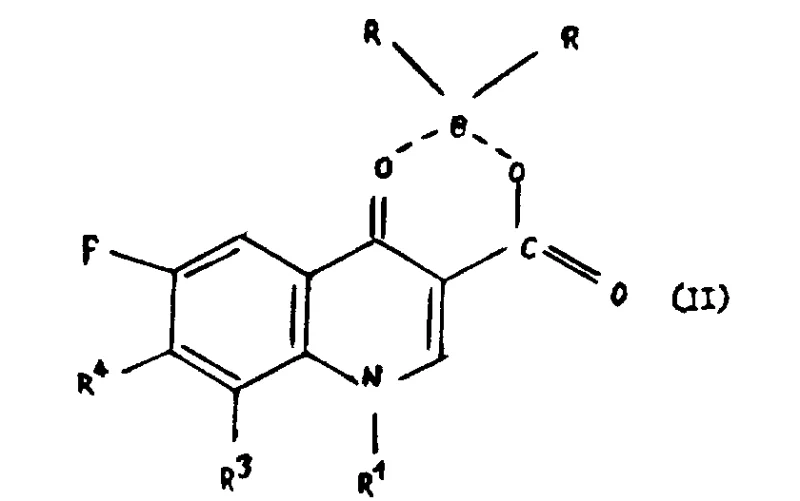 . This pre-activation step generates a stable boron derivative (Formula II) that directs the incoming piperazine nucleophile exclusively to the desired 7-position. By protecting the 3-carboxyl and 4-keto groups as a boron chelate, the process effectively shuts down side reactions that would otherwise consume starting materials and generate impurities. This novel approach allows the reaction to proceed under milder conditions compared to traditional methods, utilizing common polar aprotic solvents like dimethyl sulfoxide (DMSO) and standard organic or inorganic bases. The result is a cleaner reaction profile with significantly higher conversion rates, which translates directly into improved process efficiency and reduced operational costs for manufacturers seeking to optimize their production lines for high-purity OLED material or pharmaceutical intermediates.
. This pre-activation step generates a stable boron derivative (Formula II) that directs the incoming piperazine nucleophile exclusively to the desired 7-position. By protecting the 3-carboxyl and 4-keto groups as a boron chelate, the process effectively shuts down side reactions that would otherwise consume starting materials and generate impurities. This novel approach allows the reaction to proceed under milder conditions compared to traditional methods, utilizing common polar aprotic solvents like dimethyl sulfoxide (DMSO) and standard organic or inorganic bases. The result is a cleaner reaction profile with significantly higher conversion rates, which translates directly into improved process efficiency and reduced operational costs for manufacturers seeking to optimize their production lines for high-purity OLED material or pharmaceutical intermediates.
Mechanistic Insights into Boron-Mediated Cyclization and Substitution
The core mechanistic advantage of this process lies in the electronic modulation of the quinolone ring system achieved through boron complexation. When the starting quinoline carboxylic acid reacts with a boron derivative, it forms a cyclic ester-like structure that locks the conformation of the molecule and alters the electron density distribution across the aromatic system. 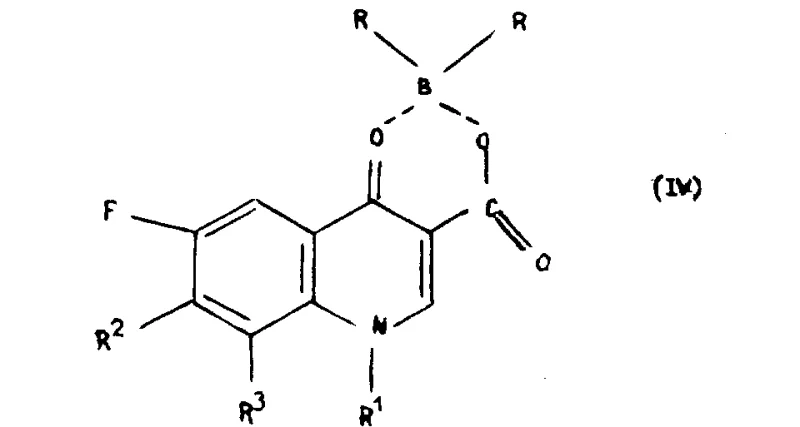 . This electronic activation makes the carbon atom at the 7-position significantly more susceptible to nucleophilic attack by the piperazine derivative (Formula III), while simultaneously rendering the rest of the molecule inert to unwanted side reactions. The reaction proceeds through a concerted mechanism where the boron moiety stabilizes the transition state, lowering the activation energy required for the substitution. Following the successful introduction of the piperazine ring, the boron protecting group is easily removed via hydrolysis under acidic or basic conditions. This deprotection step regenerates the free carboxylic acid and ketone functionalities, yielding the final target compound (Formula I) with high structural integrity.
. This electronic activation makes the carbon atom at the 7-position significantly more susceptible to nucleophilic attack by the piperazine derivative (Formula III), while simultaneously rendering the rest of the molecule inert to unwanted side reactions. The reaction proceeds through a concerted mechanism where the boron moiety stabilizes the transition state, lowering the activation energy required for the substitution. Following the successful introduction of the piperazine ring, the boron protecting group is easily removed via hydrolysis under acidic or basic conditions. This deprotection step regenerates the free carboxylic acid and ketone functionalities, yielding the final target compound (Formula I) with high structural integrity. 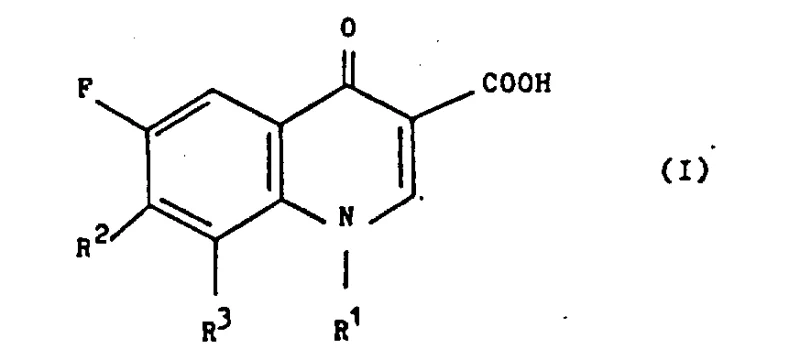 . This precise control over the reaction pathway ensures that the impurity profile remains minimal, a critical factor for R&D teams focused on developing robust analytical methods and stability protocols for new drug formulations.
. This precise control over the reaction pathway ensures that the impurity profile remains minimal, a critical factor for R&D teams focused on developing robust analytical methods and stability protocols for new drug formulations.
Furthermore, the hydrolysis step is designed to be operationally simple, allowing for the isolation of the product through straightforward pH adjustment and crystallization. The patent specifies that the intermediate Formula IV can be hydrolyzed without prior isolation, enabling a telescoped process that saves time and solvent usage. The use of aqueous sodium hydroxide or potassium carbonate for hydrolysis ensures that the reaction environment remains compatible with large-scale stainless steel reactors, avoiding the corrosion issues associated with strong mineral acids. The ability to tune the pH precisely allows for the selective precipitation of the zwitterionic product, leaving soluble impurities in the mother liquor. This crystallization-driven purification is far more scalable and cost-effective than chromatographic techniques, providing a clear pathway for commercial scale-up of complex polymer additives or fine chemical intermediates. The mechanistic robustness of this route means that variations in raw material quality can be accommodated without compromising the final product specification, enhancing the overall reliability of the manufacturing process.
How to Synthesize 1-Ethyl-6,8-Difluoro-4-Oxo-Quinoline-3-Carboxylic Acid Efficiently
The practical implementation of this synthesis involves reacting the boron-complexed precursor with the appropriate amine in a solvent such as DMSO at temperatures ranging from 100°C to 110°C. The reaction mixture is maintained for a period of 1 to 3 hours to ensure complete conversion, after which an aqueous base is added directly to effect hydrolysis.
- React the boron-complexed quinoline derivative (Formula II) with a piperazine derivative in a polar aprotic solvent like DMSO at elevated temperatures.
- Allow the reaction to proceed for 0.5 to 10 hours to ensure complete substitution at the 7-position, forming the intermediate Formula IV.
- Hydrolyze the intermediate using an aqueous alkaline solution such as sodium hydroxide, followed by pH adjustment to isolate the final carboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this boron-mediated synthesis route offers substantial strategic benefits beyond mere technical elegance. The primary advantage lies in the drastic simplification of the purification workflow, which directly correlates to reduced manufacturing costs and shorter cycle times. By eliminating the need for complex chromatographic separations and minimizing the generation of hard-to-remove byproducts, the process significantly lowers the consumption of solvents and consumables. This efficiency gain allows suppliers to offer more competitive pricing structures without sacrificing margin, addressing the constant pressure for cost reduction in electronic chemical manufacturing and related sectors. Additionally, the reliance on commodity chemicals such as DMSO, sodium hydroxide, and piperazine derivatives ensures that the supply chain remains resilient against fluctuations in the availability of exotic reagents. The robustness of the chemistry means that production schedules can be maintained with high predictability, reducing the risk of delays that often plague multi-step synthetic sequences involving unstable intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in solvent usage through telescoped processing steps lead to significant operational expenditure savings. The high regioselectivity of the boron-mediated reaction minimizes the loss of valuable starting materials to side products, thereby improving the overall mass balance and yield of the process. This efficiency translates into a lower cost of goods sold (COGS), enabling manufacturers to remain competitive in a price-sensitive global market. Furthermore, the simplified workup procedure reduces the labor and energy requirements associated with downstream processing, contributing to a leaner and more agile production model.
- Enhanced Supply Chain Reliability: The use of stable boron intermediates allows for potential storage and transport of key precursors, decoupling the synthesis steps and providing greater flexibility in production planning. Since the reagents involved are widely available industrial chemicals, the risk of supply disruption due to raw material scarcity is significantly mitigated. This stability is crucial for maintaining continuous supply to downstream pharmaceutical customers who require just-in-time delivery of critical API intermediates. The process's tolerance to minor variations in reaction conditions also ensures consistent batch-to-batch quality, reducing the incidence of out-of-specification results that can disrupt inventory management.
- Scalability and Environmental Compliance: The aqueous workup and crystallization-based isolation methods are inherently scalable from kilogram to multi-ton scales without requiring specialized equipment modifications. The process generates less hazardous waste compared to traditional halogen-exchange methods, facilitating easier compliance with increasingly stringent environmental regulations. The ability to recycle solvents and recover byproducts further enhances the sustainability profile of the manufacturing operation. This alignment with green chemistry principles not only reduces disposal costs but also strengthens the brand reputation of the supplier as a responsible partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What is the primary advantage of using boron derivatives in this synthesis?
A: The use of boron derivatives (Formula II) protects the reactive 3-carboxyl and 4-keto groups, ensuring highly regioselective substitution at the 7-position and minimizing byproduct formation.
Q: Can this process be scaled for industrial production?
A: Yes, the process utilizes common solvents like DMSO and standard inorganic bases, making it highly suitable for commercial scale-up without requiring exotic reagents or extreme pressure conditions.
Q: How is the final product purified according to the patent?
A: The final product is isolated by adjusting the pH of the hydrolyzed mixture to precipitate the crystalline acid, which is then filtered and washed, avoiding complex chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has extensively evaluated the boron-mediated process described in CN1025028C and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from laboratory benchtop to full industrial manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of quinoline derivative meets the highest international standards. We understand that consistency is key for R&D Directors and Procurement Managers, and our commitment to quality assurance ensures that you receive a product that performs reliably in your downstream applications.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to specific COA data and route feasibility assessments that will help you optimize your supply chain strategy. Whether you are developing new antibiotics or optimizing existing formulations, our expertise in fluoroquinolone chemistry positions us as the ideal partner for your long-term success. Contact us today to discuss how we can support your goals with high-quality, cost-effective solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
