Advanced Manufacturing of 5-HT2A Antagonist Intermediates for Global Pharmaceutical Supply Chains
Advanced Manufacturing of 5-HT2A Antagonist Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for central nervous system (CNS) active ingredients, particularly those targeting serotonin receptors. Patent CN1213029C discloses a highly efficient process for the preparation of 7-{4-[2-(4-fluorophenyl)-ethyl]-piperazin-1-ylcarbonyl}-1H-indole-3-carbonitrile and its salts, which function as selective 5-HT2A receptor antagonists. These compounds hold significant therapeutic potential for treating neurological disorders such as depression, anxiety, and sleep disturbances, as well as cardiovascular conditions. The disclosed methodology represents a substantial advancement over prior art by optimizing the introduction of the critical cyano group at the 3-position of the indole ring. By leveraging versatile intermediates like indole esters, the process ensures high purity and yield, addressing key concerns for R&D directors focused on impurity profiles. The structural complexity of the final molecule requires precise control over each synthetic transformation to maintain regulatory compliance.
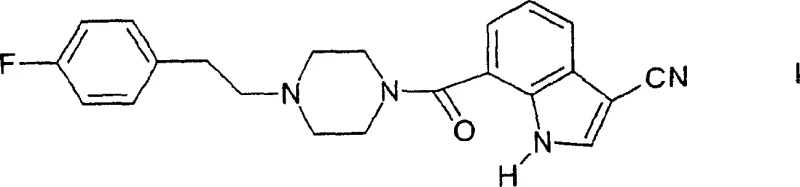
This technical insight report analyzes the proprietary synthesis detailed in the patent, highlighting its implications for cost reduction in pharmaceutical intermediate manufacturing and supply chain stability. The ability to produce high-purity 5-HT2A antagonist intermediates reliably is crucial for downstream drug development. As a leading fine chemical manufacturer, understanding these mechanistic nuances allows us to offer superior contract development and manufacturing organization (CDMO) services. The following sections dissect the chemical logic, compare it with conventional methods, and outline the commercial advantages for procurement and supply chain stakeholders seeking reliable partners for complex organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for functionalizing the indole nucleus at the 3-position often suffer from harsh reaction conditions and poor regioselectivity. Conventional methods might rely on direct cyanation using toxic reagents under extreme temperatures, which can lead to significant decomposition of the sensitive indole core. Furthermore, older protocols frequently struggle with the purification of intermediates, resulting in broad impurity spectra that complicate downstream processing. The use of heavy metal catalysts in some traditional cyanation processes introduces the risk of residual metal contamination, necessitating expensive and time-consuming purification steps to meet stringent pharmaceutical standards. Additionally, multi-step sequences that lack convergent strategies often result in cumulative yield losses, making the overall process economically unviable for commercial scale-up of complex pharmaceutical intermediates. These inefficiencies create bottlenecks in the supply chain, increasing lead times and reducing the reliability of raw material availability for API manufacturers.
The Novel Approach
The process described in CN1213029C introduces a strategic divergence by utilizing a formylation-oximation-dehydration sequence or a halogenation-cyanation sequence to install the cyano group. This novel approach allows for the use of commercially available indole esters as starting materials, significantly simplifying the supply chain logistics. By converting the indole ester into a 3-formyl derivative via Vilsmeier-Haack formylation, the process establishes a versatile handle for subsequent transformation into the nitrile functionality. The conversion of the aldehyde to an oxime and subsequent dehydration provides a mild and controlled pathway to the cyano group, avoiding the harsh conditions associated with direct cyanation. Alternatively, the halogenation route offers a robust fallback option, demonstrating the flexibility of the synthetic design. This modularity ensures that production can continue even if specific reagents face temporary shortages, thereby enhancing supply chain resilience. The final coupling with the piperazine moiety is performed under optimized conditions to ensure high conversion and minimal byproduct formation.
Mechanistic Insights into Vilsmeier-Haack Formylation and Oxime Dehydration
The core of this synthetic strategy lies in the precise manipulation of the indole electronic system to achieve regioselective functionalization. The initial step involves the Vilsmeier-Haack reaction, where phosphorus oxychloride reacts with dimethylformamide (DMF) to generate a reactive chloroiminium ion. This electrophile attacks the electron-rich 3-position of the indole ring, forming an intermediate that hydrolyzes to the 3-formyl indole ester. This step is critical because it sets the stage for the introduction of the nitrogen-containing functionality. The reaction conditions, typically involving temperatures between 0°C and 50°C followed by heating, must be carefully monitored to prevent polymerization or over-chlorination. The choice of solvent, such as DMF or mixtures with aromatic hydrocarbons, plays a pivotal role in solubilizing the intermediates and driving the equilibrium towards the desired aldehyde. Understanding these mechanistic details is essential for R&D teams aiming to replicate or optimize the process for larger scales.
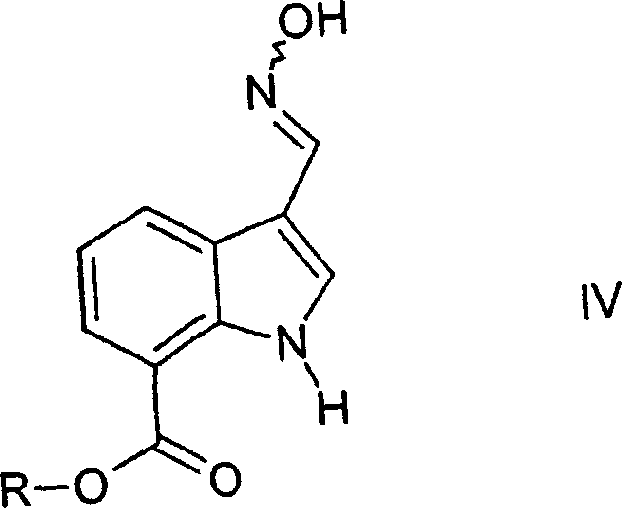
Following formylation, the aldehyde is converted into an oxime derivative through condensation with hydroxylamine. This transformation is generally high-yielding and proceeds under mild conditions in polar aprotic solvents. The subsequent dehydration of the oxime to the nitrile is the key step that defines the efficiency of this route. The patent suggests using acid treatment or dehydrating agents to effect this conversion, transforming the C=N-OH bond into a C≡N triple bond. This mechanism avoids the use of toxic cyanide salts directly on the indole ring, thereby improving the safety profile of the manufacturing process. The resulting cyano indole ester is then saponified to the free acid, which serves as the coupling partner for the piperazine amine. This stepwise construction allows for rigorous quality control at each stage, ensuring that the final API intermediate meets the stringent purity specifications required for clinical applications. The ability to isolate and characterize the oxime intermediate provides an additional checkpoint for impurity control.
How to Synthesize 3-Cyano-1H-indole-7-carboxylic Acid Efficiently
The synthesis of the key 3-cyano-1H-indole-7-carboxylic acid intermediate requires careful attention to reaction parameters to maximize yield and purity. The process begins with the selection of an appropriate indole ester, such as ethyl 1H-indole-7-carboxylate, which balances reactivity and cost. The formylation step must be quenched carefully to avoid exotherms, and the subsequent oxime formation should be driven to completion to minimize residual aldehyde. The dehydration step is particularly sensitive to moisture and acid concentration, requiring optimized workup procedures to isolate the cyano ester cleanly. Finally, the saponification should be conducted under conditions that prevent hydrolysis of the nitrile group while efficiently cleaving the ester. Detailed standardized synthetic steps are provided below to guide process chemists in implementing this route effectively.
- Formylate the indole ester (Formula II) using Vilsmeier-Haack conditions with POCl3 and DMF to generate the 3-formyl intermediate.
- React the formyl intermediate with hydroxylamine to form the oxime derivative (Formula IV), followed by dehydration to yield the cyano ester.
- Saponify the cyano ester to the corresponding carboxylic acid and couple with 1-[2-(4-fluorophenyl)ethyl]piperazine to finalize the API intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits in terms of cost stability and operational efficiency. The reliance on commodity chemicals like DMF, phosphorus oxychloride, and hydroxylamine means that raw material costs are predictable and less susceptible to market volatility compared to processes requiring exotic catalysts. The modular nature of the synthesis allows for flexible manufacturing schedules, enabling producers to stockpile key intermediates like the cyano ester to buffer against supply disruptions. Furthermore, the avoidance of heavy metal catalysts in the primary route simplifies waste treatment and reduces the environmental footprint of the manufacturing process. These factors collectively contribute to a more sustainable and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of readily available starting materials significantly lower the bill of materials. By streamlining the synthesis into fewer distinct operational units, labor and utility costs are also reduced. The high yields reported in the patent examples suggest that material throughput is maximized, further driving down the cost per kilogram of the final intermediate. This economic efficiency allows for competitive pricing strategies without compromising on quality standards.
- Enhanced Supply Chain Reliability: The use of robust chemical transformations that tolerate minor variations in reaction conditions ensures consistent output quality. Since the starting indole esters are commercially available or easily synthesized from nitrobenzoic acids, the risk of raw material shortage is minimized. The process design supports batch production models that can be easily scaled from pilot plant to commercial manufacturing, ensuring that supply can meet demand fluctuations in the pharmaceutical market. This reliability is crucial for maintaining continuous API production lines.
- Scalability and Environmental Compliance: The reaction conditions described, such as moderate temperatures and standard pressures, are well-suited for large-scale reactor operations. The waste streams generated are primarily aqueous and organic solvents that can be treated using standard industrial effluent treatment protocols. The absence of persistent organic pollutants or heavy metals simplifies regulatory compliance and reduces the cost of waste disposal. This alignment with green chemistry principles enhances the long-term viability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 5-HT2A antagonist intermediates. These answers are derived from the detailed experimental data and claims found within the patent documentation. They serve to clarify the feasibility and advantages of the described process for potential partners and stakeholders. Understanding these aspects is vital for making informed decisions about sourcing and development strategies.
Q: What are the critical impurities in the oxime dehydration step?
A: The primary impurities often stem from incomplete dehydration of the oxime or over-reaction during the Vilsmeier formylation. Strict temperature control during the dehydration phase is essential to minimize side products.
Q: Can the cyano group be introduced via halogenation instead of oxime dehydration?
A: Yes, the patent describes an alternative route involving halogenation at the 3-position followed by cyanation with copper cyanide, offering flexibility depending on raw material availability.
Q: Is this process suitable for large-scale commercial production?
A: The process utilizes standard reagents like POCl3 and hydroxylamine and avoids exotic catalysts, making it highly scalable for multi-ton manufacturing with appropriate safety controls.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-HT2A Antagonist Supplier
The technological potential of the synthesis route described in CN1213029C is immense, offering a clear path to high-quality CNS drug intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from lab bench to market. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 5-HT2A antagonist intermediate meets the highest international standards. We understand the critical nature of timeline and quality in the pharmaceutical sector and are committed to delivering excellence.
We invite you to collaborate with us to leverage this advanced synthetic technology for your drug development programs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals. Let us be your partner in bringing innovative therapies to patients worldwide.
