Scalable Synthesis of Flunixin Meglumine Intermediate for Veterinary Pharmaceutical Manufacturing
The pharmaceutical landscape for veterinary medicine is constantly evolving, demanding intermediates of exceptional purity and structural integrity to ensure the efficacy of final active pharmaceutical ingredients (APIs). A pivotal development in this sector is detailed in patent CN113372264A, which outlines a robust synthetic method for producing 2-[2-methyl-3-(trifluoromethyl)phenylamino]nicotinic acid. This compound serves as a critical precursor for Flunixin Meglumine, a potent non-steroidal anti-inflammatory drug (NSAID) widely used in equine and bovine medicine for treating colic, respiratory diseases, and mastitis. The patented methodology addresses long-standing industry challenges regarding low yields and impurity accumulation by introducing a streamlined five-step sequence that leverages electrophilic substitution and high-pressure halogen exchange. By optimizing reaction conditions such as temperature gradients and catalyst loading, this approach ensures a consistent supply of high-quality intermediates essential for downstream API manufacturing.
![Chemical structure of the final product 2-[2-methyl-3-(trifluoromethyl)phenylamino]nicotinic acid](/insights/img/2-methyl-3-trifluoromethyl-phenylamino-nicotinic-acid-supplier-20260307015707-05.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex fluorinated aniline derivatives like the target molecule has been plagued by inefficient reaction pathways that struggle to balance yield with purity. Traditional routes often rely on multiple protection and deprotection steps or utilize expensive fluorinating reagents that are difficult to handle on a large scale, leading to significant batch-to-batch variability. Furthermore, conventional nitration and reduction sequences frequently generate isomeric byproducts that are structurally similar to the desired intermediate, making purification via crystallization or chromatography both costly and time-consuming. These inefficiencies result in a final product that may contain residual heavy metals or organic impurities, posing risks for regulatory compliance in veterinary drug applications. The reliance on less selective catalysts in older methods also necessitates extensive downstream processing, which erodes profit margins and extends the overall production lead time for pharmaceutical manufacturers.
The Novel Approach
In contrast, the methodology disclosed in the patent introduces a highly efficient pathway starting from readily available commodity chemicals like 3,4-dichlorotoluene and carbon tetrachloride. The innovation lies in the strategic use of electrophilic substitution to install the trichloromethyl group, followed by a direct halogen exchange with hydrogen fluoride under controlled high-pressure conditions. This specific sequence avoids the need for exotic reagents and simplifies the molecular construction, allowing for higher throughput in standard reactor setups. The subsequent nitration and hydrogenation steps are optimized with specific solvent systems and catalyst ratios, such as palladium-carbon, to maximize conversion rates while minimizing side reactions. This holistic approach not only enhances the overall yield but also ensures that the final 2-[2-methyl-3-(trifluoromethyl)phenylamino]nicotinic acid meets stringent purity specifications required for GMP manufacturing environments.
Mechanistic Insights into Electrophilic Substitution and Fluorination
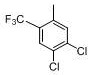
The core of this synthetic strategy relies on a sophisticated understanding of aromatic substitution mechanisms and halogen exchange dynamics. The initial step involves a Friedel-Crafts type electrophilic substitution where aluminum trichloride acts as a Lewis acid catalyst to activate carbon tetrachloride, generating a reactive electrophile that attacks the electron-rich aromatic ring of 3,4-dichlorotoluene. This regioselective attack is crucial for positioning the trichloromethyl group ortho to the methyl group, setting the stage for the subsequent fluorination. The precision of this step determines the structural fidelity of the entire molecule, as any misplacement of the substituent would render the intermediate useless for the final coupling reaction. Careful temperature control during the exothermic addition phase prevents poly-substitution and ensures the formation of the mono-substituted Intermediate A with high selectivity.
Following the substitution, the transformation of the trichloromethyl group into a trifluoromethyl moiety represents the most chemically demanding phase of the synthesis. This halogen exchange reaction proceeds via a nucleophilic substitution mechanism where hydrogen fluoride, under elevated pressure and temperature, displaces the chlorine atoms. The high electronegativity of fluorine drives this equilibrium forward, but it requires robust equipment capable of withstanding corrosive conditions. The resulting trifluoromethyl group significantly alters the electronic properties of the aromatic ring, deactivating it towards further electrophilic attack and thereby stabilizing the molecule for the subsequent nitration step. This electronic modulation is vital for controlling the position of the nitro group in the next stage, ensuring it lands precisely where needed for the final amine formation.
How to Synthesize 2-[2-Methyl-3-(Trifluoromethyl)Phenylamino]Nicotinic Acid Efficiently
Executing this synthesis requires strict adherence to the defined operational parameters to maintain safety and product quality throughout the five distinct stages. The process begins with the preparation of the trichloromethyl intermediate, followed by the high-pressure fluorination which demands specialized reactor infrastructure. Once the fluorinated core is established, the nitration must be conducted with careful acid management to prevent over-oxidation, followed by a catalytic hydrogenation that requires precise hydrogen pressure control to reduce the nitro group without affecting other sensitive functionalities. The final coupling with 2-chloronicotinic acid completes the assembly of the pharmacophore. For a detailed breakdown of the specific mass ratios, temperature profiles, and workup procedures for each step, please refer to the standardized guide below.
- Perform electrophilic substitution on 3,4-dichlorotoluene using carbon tetrachloride and aluminum trichloride to generate the trichloromethyl intermediate.
- Execute high-pressure halogen exchange with hydrogen fluoride to convert the trichloromethyl group into a trifluoromethyl group.
- Conduct nitration followed by catalytic hydrogenation reduction to introduce the amino functionality, concluding with amide coupling using 2-chloronicotinic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial advantages by leveraging low-cost, high-volume starting materials that are readily available in the global chemical market. The elimination of complex protecting group strategies and the use of standard unit operations significantly simplify the manufacturing process, reducing the capital expenditure required for specialized equipment. This simplicity translates directly into a more resilient supply chain, as the reliance on niche reagents is minimized, mitigating the risk of shortages that can disrupt production schedules. Furthermore, the high purity achieved at each intermediate stage reduces the burden on quality control laboratories and minimizes the waste associated with re-processing off-spec batches, leading to a more sustainable and cost-effective operation overall.
- Cost Reduction in Manufacturing: The utilization of commodity chemicals such as carbon tetrachloride and aluminum trichloride as primary feedstocks drastically lowers the raw material cost basis compared to routes requiring custom-synthesized fluorinated building blocks. By avoiding expensive transition metal catalysts beyond standard palladium on carbon and eliminating the need for cryogenic conditions, the operational expenditure is significantly optimized. The high yield and purity profile mean that less material is lost to purification losses, effectively increasing the mass efficiency of the entire process and delivering a lower cost per kilogram of the final active intermediate.
- Enhanced Supply Chain Reliability: Because the synthesis relies on well-established chemical transformations like nitration and hydrogenation, it can be easily replicated across multiple manufacturing sites without requiring proprietary technology transfers that often delay timelines. The robustness of the halogen exchange step under defined pressure conditions ensures consistent output, allowing procurement teams to forecast demand with greater accuracy. This reliability is critical for maintaining continuous production of Flunixin Meglumine, ensuring that veterinary clinics and farms have uninterrupted access to essential pain management medications.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are compatible with large-scale stainless steel reactors commonly found in fine chemical facilities. Waste streams are manageable through standard neutralization and extraction protocols, and the high atom economy of the coupling steps reduces the volume of organic solvent waste generated. This alignment with green chemistry principles facilitates easier regulatory approval and environmental permitting, accelerating the time to market for new batches and supporting long-term sustainability goals for pharmaceutical manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this veterinary pharmaceutical intermediate. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on process capabilities and quality standards. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this supply source into their existing manufacturing workflows.
Q: What are the critical control points in the fluorination step of this synthesis?
A: The halogen exchange reaction requires precise high-pressure conditions (approximately 8kg pressure) and temperature control between 60-80°C to ensure complete conversion of the trichloromethyl group to the trifluoromethyl group while maintaining safety standards.
Q: How does this method improve purity compared to conventional routes?
A: By utilizing a specific sequence of electrophilic substitution followed by rigorous purification steps like recrystallization and rectification at each stage, the process achieves final product purity levels exceeding 99%, significantly reducing impurity profiles common in older methods.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process utilizes standard industrial unit operations such as high-pressure reactors for fluorination and standard hydrogenation vessels, making it highly adaptable for commercial scale-up from pilot plants to multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-[2-Methyl-3-(Trifluoromethyl)Phenylamino]Nicotinic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and production of life-saving veterinary medicines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and potency of every batch, guaranteeing that our 2-[2-methyl-3-(trifluoromethyl)phenylamino]nicotinic acid meets the highest industry standards.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthetic capabilities can support your long-term business goals.
