Scalable Synthesis of Propionyl-L-carnitine Hydrochloride: A Technical Breakthrough for Global Supply Chains
The pharmaceutical and nutritional supplement industries are constantly seeking robust, scalable, and environmentally compliant synthetic routes for critical bioactive molecules. Patent CN1995010B represents a significant advancement in the manufacturing of Propionyl-L-carnitine hydrochloride, a vital compound used in cardiovascular therapeutics and metabolic support formulations. This proprietary methodology addresses long-standing challenges associated with toxicity, solvent safety, and purification efficiency that have plagued earlier production techniques. By utilizing L-carnitine inner salt as the starting material and employing propionic acid as both a solvent and reaction medium, the process achieves a streamlined workflow that minimizes hazardous waste generation. The technical breakthrough lies not only in the chemical transformation but in the holistic design of the downstream processing, which ensures that the final active pharmaceutical ingredient (API) intermediate meets stringent quality standards required by global regulatory bodies. For procurement leaders and technical directors, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of propionyl-L-carnitine derivatives has been fraught with significant operational and safety hurdles that impact both cost and environmental compliance. Early methodologies, such as those described in US Patent 4,443,475, relied heavily on trifluoroacetic acid (TFA) as a solvent, which presents severe toxicity risks to personnel and requires complex waste treatment protocols to neutralize fluorinated byproducts. Furthermore, the recrystallization steps in these legacy processes often utilized ternary mixed solvents including ether, a highly volatile and flammable substance that poses explosion hazards in large-scale industrial reactors. Another notable approach, documented in CN 1651402A, attempted to mitigate toxicity by switching to propionic acid but introduced p-toluenesulfonic acid (tosic acid) as a catalyst. While this reduced some toxicity, it necessitated additional purification steps to remove the acidic catalyst residues, thereby complicating the isolation process and potentially introducing organic impurities that are difficult to purge. These conventional methods also suffered from poor solvent recyclability, leading to excessive consumption of raw materials and higher operational expenditures due to waste disposal fees.
The Novel Approach
The innovative strategy outlined in CN1995010B fundamentally reengineers the synthesis pathway to eliminate these critical bottlenecks without compromising reaction kinetics or product integrity. By removing the need for external acid catalysts like tosics acid, the new method simplifies the reaction mixture, allowing for a more direct isolation of the crude product through vacuum distillation and acetone precipitation. The substitution of dangerous ether solvents with a binary ethanol-acetone system for recrystallization drastically improves the safety profile of the manufacturing plant, aligning with modern occupational health and safety standards. This novel approach leverages the dual functionality of propionic acid, which acts effectively as a solvent to dissolve the L-carnitine inner salt while facilitating the acylation reaction with propionyl chloride at moderate temperatures between 50°C and 80°C. The result is a cleaner reaction profile that supports high-throughput manufacturing, offering a compelling value proposition for cost reduction in pharma intermediate manufacturing where safety and purity are paramount.
Mechanistic Insights into Propionyl Chloride Acylation
The core chemical transformation in this process is the nucleophilic acyl substitution where the hydroxyl group of the L-carnitine inner salt attacks the carbonyl carbon of propionyl chloride. In the absence of a strong base or external catalyst, the reaction relies on the inherent nucleophilicity of the alcohol moiety and the electrophilicity of the acid chloride, facilitated by the polar protic environment of the propionic acid solvent. The propionic acid medium helps to stabilize the transition state and solvate the resulting hydrochloride salt byproduct, driving the equilibrium towards the formation of the ester linkage. Maintaining the reaction temperature within the 50°C to 80°C range is critical; temperatures below this threshold may result in incomplete conversion, while excessive heat could promote side reactions or degradation of the sensitive carnitine backbone. The stoichiometry is carefully controlled, typically using a slight excess of propionyl chloride (1.0 to 1.5 equivalents) to ensure complete consumption of the valuable L-carnitine starting material, thereby maximizing atom economy.
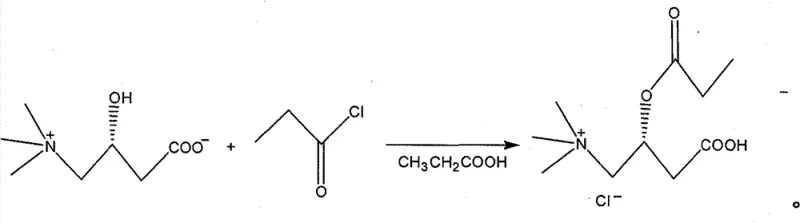
Following the acylation, the purification mechanism is driven by differential solubility and crystallization thermodynamics. The removal of excess propionic acid under reduced pressure concentrates the reaction mass, and the subsequent addition of acetone induces precipitation of the crude hydrochloride salt. Acetone acts as an anti-solvent, reducing the dielectric constant of the medium and forcing the ionic product out of solution. The final recrystallization step utilizes a precise ratio of ethanol to acetone, where the product is dissolved at elevated temperatures (around 50°C) to ensure saturation, followed by controlled cooling to 0°C to 10°C. This thermal gradient promotes the growth of well-defined crystals while excluding impurities that remain soluble in the mother liquor. This rigorous control over crystallization parameters is what enables the process to achieve purity levels exceeding 99%, effectively managing the impurity profile to meet the strict specifications required for high-purity pharmaceutical intermediates.
How to Synthesize Propionyl-L-carnitine Hydrochloride Efficiently
Implementing this synthesis route requires precise adherence to the thermal and stoichiometric parameters defined in the patent to ensure reproducibility and optimal yield. The process begins with the dissolution of L-carnitine inner salt in propionic acid, followed by the controlled addition of propionyl chloride under heating conditions to drive the acylation to completion. Once the reaction is finalized, the solvent is removed via vacuum distillation, and the residue is treated with acetone to isolate the crude solid. The critical final stage involves dissolving this crude material in ethanol, concentrating the solution, and inducing crystallization with additional acetone under cold conditions. For detailed operational parameters, temperature ramps, and specific mass ratios required for GMP-compliant production, please refer to the standardized protocol below.
- React L-carnitine inner salt with propionyl chloride in propionic acid solvent at 50-80°C, then remove solvent under vacuum.
- Add acetone to the residue for dispersion and cooling crystallization at 0-5°C to isolate the crude product.
- Recrystallize the crude product using an ethanol-acetone system, concentrating under vacuum and cooling to obtain high-purity pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented synthesis method offers substantial advantages that directly impact the bottom line and supply chain resilience. By eliminating the need for specialized corrosion-resistant equipment required for handling trifluoroacetic acid, manufacturers can utilize standard stainless steel reactors, significantly lowering capital expenditure and maintenance costs. The removal of ether from the process not only enhances site safety but also reduces insurance premiums and regulatory compliance burdens associated with storing and handling highly flammable Class I solvents. Furthermore, the ability to recycle propionic acid and acetone creates a closed-loop solvent system that minimizes raw material procurement volumes and reduces the environmental footprint of the manufacturing facility. These efficiencies translate into a more stable pricing structure for buyers, shielding them from volatility in raw material markets and ensuring a consistent supply of critical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic catalysts like tosics acid removes an entire unit operation dedicated to catalyst removal and neutralization, thereby reducing labor and utility costs. Additionally, the high recovery rate of solvents means that the effective cost per kilogram of solvent consumed is drastically lowered, contributing to significant overall cost savings in the production budget. The simplified workup procedure also shortens the batch cycle time, allowing for more batches to be produced within the same timeframe, which improves asset utilization rates and throughput capacity without requiring new infrastructure investments.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as propionic acid, ethanol, and acetone ensures that the supply chain is not vulnerable to shortages of exotic or highly regulated reagents. These solvents are widely available from multiple global suppliers, reducing the risk of supply disruption due to geopolitical issues or single-source vendor failures. The robustness of the process, demonstrated by yields consistently above 90%, ensures that production targets are met reliably, preventing delays in the downstream formulation of finished pharmaceutical products and maintaining continuity for the end-user market.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex amino acid derivatives, with exothermic profiles that are manageable in large reactors through controlled dosing of the acid chloride. The avoidance of halogenated solvents and the reduction of hazardous waste streams simplify the permitting process for new manufacturing sites and ensure compliance with increasingly stringent environmental regulations like REACH and TSCA. This environmental stewardship not only mitigates regulatory risk but also aligns with the sustainability goals of major multinational pharmaceutical companies, making the supplier a more attractive partner for long-term contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and quality assurance of Propionyl-L-carnitine hydrochloride using this advanced methodology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this process outperforms legacy technologies. Understanding these details is crucial for technical teams evaluating vendor capabilities and for procurement officers negotiating supply agreements based on quality and safety metrics.
Q: How does this synthesis method improve safety compared to prior art?
A: This method eliminates the use of highly toxic trifluoroacetic acid and flammable ether solvents found in previous patents, replacing them with safer propionic acid and ethanol-acetone systems.
Q: What is the expected yield and purity of the final product?
A: The patented process consistently achieves a total yield of at least 90% with a final purity exceeding 99%, as verified by HPLC analysis.
Q: Is the solvent system recyclable for cost efficiency?
A: Yes, the process allows for the recovery and recycling of both propionic acid and acetone, significantly reducing raw material consumption and waste disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Propionyl-L-carnitine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1995010B are fully realized in our manufacturing facilities. We operate with stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of Propionyl-L-carnitine hydrochloride meets the highest industry standards for identity, assay, and impurity profiles. Our dedication to process optimization allows us to deliver high-purity pharmaceutical intermediates that support the development of life-saving cardiovascular therapies and nutritional supplements.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized synthesis capabilities can enhance your product portfolio while reducing overall manufacturing costs and lead times.
