Advanced Manufacturing of Esomeprazole Magnesium Trihydrate for Global Pharmaceutical Supply Chains
Advanced Manufacturing of Esomeprazole Magnesium Trihydrate for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for producing critical active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in this domain is detailed in patent CN109912569B, which outlines a novel preparation method for esomeprazole magnesium trihydrate and its key intermediates. This technology addresses long-standing challenges in the synthesis of proton pump inhibitors by offering a route that combines high yield, exceptional purity, and mild reaction conditions. For R&D directors and procurement specialists, understanding the nuances of this process is vital, as it represents a shift away from hazardous, energy-intensive legacy methods toward a more streamlined, industrial-friendly manufacturing paradigm that ensures consistent supply chain reliability.
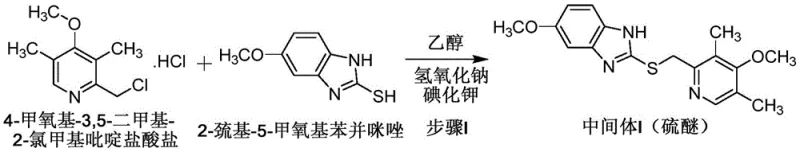
The limitations of conventional synthetic routes for esomeprazole have historically hindered efficient mass production. Traditional approaches often rely on biological enzyme catalytic oxidation, which, while specific, involves complicated operations and prohibitive costs that make it unsuitable for large-scale application. Alternatively, the racemate omeprazole resolution method suffers from inherently low yields and necessitates the use of toxic, expensive resolving agents that pose significant safety risks to personnel and the environment. Furthermore, existing asymmetric catalytic oxidation methods, such as those disclosed in prior art like CN104803978A, often demand extreme temperature controls ranging from -20°C to reflux, making process stability difficult to maintain in a commercial reactor. Other methods require temperatures as high as 70°C and utilize large volumes of volatile, flammable solvents like acetonitrile, creating substantial safety hazards and environmental pollution burdens that complicate regulatory compliance and increase operational overhead.
In stark contrast, the novel approach presented in this patent utilizes a meticulously optimized asymmetric catalytic oxidation pathway that drastically simplifies the operational landscape. The condensation reaction to form the omeprazole thioether intermediate is conducted in 95% ethanol at a moderate temperature of 50-55°C, which is significantly lower than the reflux temperatures required by older technologies, thereby reducing energy consumption. The subsequent chiral oxidation step employs a titanium-based catalyst system in toluene, operating effectively between 25-30°C, avoiding the need for cryogenic cooling or excessive heating. Crucially, the final ion exchange to form the magnesium salt is performed directly in water, eliminating the need for organic solvents in the final isolation step. This holistic optimization results in a process that is not only safer and more environmentally friendly but also easier to control, ensuring that high purity and yield targets are consistently met without the volatility associated with legacy methods.
Mechanistic Insights into Asymmetric Catalytic Oxidation and Ion Exchange
The core of this technological advancement lies in the precise control of the chiral oxidation mechanism and the subsequent salt formation. The process begins with the condensation of 2-mercapto-5-methoxybenzimidazole and 4-methoxy-3,5-dimethyl-2-chloromethylpyridine hydrochloride. By utilizing ethanol as a homogeneous solvent, the reaction kinetics are enhanced, allowing for complete conversion within 30 minutes at 50-55°C. The resulting omeprazole thioether serves as the substrate for the critical chiral oxidation step. Here, a chiral reaction system is formed using diethyl D-tartrate and titanium tetraisopropoxide in toluene. The addition of an oxidant, specifically cumene hydroperoxide, at controlled low temperatures (10-15°C during addition, rising to 25-30°C for reaction) ensures high enantioselectivity. This careful thermal management prevents the formation of unwanted racemates and sulfone byproducts, which are common impurities in less controlled processes.

Following the oxidation, the conversion to the stable magnesium salt involves a strategic two-stage salt formation process designed to maximize purity. First, the esomeprazole is converted to a potassium salt using potassium hydroxide in methanol. This intermediate undergoes a rigorous purification cycle involving acetone and methanol pulping, which effectively removes Impurity D and optical isomers, reducing total impurities to below 0.5%. The final step involves an ion exchange reaction where the purified potassium salt reacts with magnesium chloride hexahydrate in pure water at 35-40°C. Unlike prior art that requires multi-solvent systems, this direct aqueous ion exchange leverages the slight solubility of the magnesium salt to drive crystallization naturally. This mechanism not only simplifies the downstream processing but also ensures that the final trihydrate form is obtained with a purity exceeding 99.9%, meeting the stringent specifications required for global pharmaceutical markets.
How to Synthesize Esomeprazole Magnesium Trihydrate Efficiently
The synthesis of esomeprazole magnesium trihydrate via this patented route involves a sequence of highly controlled chemical transformations designed for reproducibility and scale. The process initiates with the condensation reaction to prepare the omeprazole thioether intermediate, followed by the pivotal chiral oxidation to establish the correct stereochemistry. Subsequent steps involve the formation and purification of the esomeprazole potassium salt, culminating in the ion exchange reaction to yield the final magnesium trihydrate. Each stage is optimized for specific temperature ranges and solvent systems to ensure maximum yield and minimal impurity generation. For a detailed breakdown of the standardized operating procedures, including specific reagent ratios and monitoring techniques, please refer to the technical guide below.
- Condense 2-mercapto-5-methoxybenzimidazole with 4-methoxy-3,5-dimethyl-2-chloromethylpyridine hydrochloride in ethanol at 50-55°C to form omeprazole thioether.
- Perform asymmetric oxidation using titanium tetraisopropoxide, diethyl D-tartrate, and cumene hydroperoxide in toluene to generate esomeprazole.
- React esomeprazole with potassium hydroxide in methanol, followed by purification in acetone/methanol to obtain high-purity esomeprazole potassium.
- Conduct ion exchange in water using magnesium chloride hexahydrate at 35-40°C to crystallize the final esomeprazole magnesium trihydrate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis method offers tangible strategic advantages that extend beyond mere technical feasibility. The primary benefit lies in the significant reduction of manufacturing complexity and associated costs. By eliminating the need for expensive enzymatic catalysts or toxic resolving agents, the raw material costs are inherently lowered. Furthermore, the substitution of hazardous solvents like acetonitrile with safer, more common solvents such as ethanol and toluene reduces the regulatory burden and the cost of solvent recovery and waste disposal. The mild reaction conditions, particularly the avoidance of extreme temperatures (-20°C or 70°C), translate directly into lower energy consumption for heating and cooling, contributing to a more sustainable and cost-effective production profile that enhances overall margin potential.
- Cost Reduction in Manufacturing: The streamlined process eliminates several costly and inefficient steps found in traditional routes. By avoiding the use of large volumes of acetonitrile, which is not only expensive but also requires specialized handling and recovery infrastructure due to its toxicity and flammability, the operational expenditure is significantly reduced. The high yield of the condensation reaction (over 95%) and the efficient purification of the potassium salt minimize material loss, ensuring that the input costs are maximized in the final output. Additionally, the direct aqueous ion exchange removes the need for additional organic solvents in the final crystallization step, further cutting down on solvent procurement and waste treatment costs.
- Enhanced Supply Chain Reliability: The reliance on readily available, commodity-grade chemicals such as ethanol, toluene, and magnesium chloride ensures a robust supply chain that is less susceptible to disruptions compared to routes requiring specialized enzymes or chiral resolving agents. The simplified process flow, characterized by fewer unit operations and milder conditions, reduces the risk of batch failures and production delays. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, ensuring that inventory levels remain stable and that delivery schedules can be met consistently without the unpredictability associated with more fragile synthetic pathways.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial mass production, with reaction conditions that are easily controllable in large-scale reactors. The use of water as the primary solvent for the final ion exchange step dramatically reduces the environmental footprint of the manufacturing process, aligning with increasingly strict global environmental regulations. The reduction in hazardous waste generation and the lower energy intensity of the process facilitate easier compliance with environmental standards, mitigating the risk of regulatory penalties and enhancing the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent data and are intended to clarify the operational benefits and quality assurances provided by this method. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios or sourcing strategies.
Q: How does this novel synthesis method improve upon traditional enzymatic or resolution routes?
A: Unlike enzymatic methods which are operationally complex and costly, or resolution methods which suffer from low yields and toxic reagents, this patented route utilizes asymmetric catalytic oxidation. It operates under milder temperatures (50-55°C vs. reflux or 70°C), uses safer solvents like ethanol and toluene instead of large volumes of acetonitrile, and achieves higher purity (>99.9%) suitable for direct industrial mass production.
Q: What are the critical quality control parameters for the intermediates in this process?
A: Strict control is maintained at each stage. The omeprazole thioether intermediate achieves over 98% purity. The crude esomeprazole potassium must have Impurity D levels below 3.0%, which is further reduced to below 0.2% in the refined potassium salt. The final magnesium trihydrate product demonstrates a purity of 99.977% with optical isomer content controlled below 0.2%.
Q: Why is the water-based ion exchange step significant for commercial scaling?
A: Traditional methods often require forming magnesium salts in methanol before reacting in water. This patent streamlines the process by performing the ion exchange directly in water at mild temperatures (35-40°C). This eliminates the need for additional organic solvents in the final step, significantly reducing environmental impact, simplifying waste treatment, and lowering energy consumption during drying.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Esomeprazole Magnesium Trihydrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our expertise in process chemistry allows us to leverage innovations like the one described in patent CN109912569B to deliver high-quality intermediates and APIs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest international standards, guaranteeing that our esomeprazole magnesium trihydrate meets the exacting requirements of your formulation needs.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your production operations.
