Scalable Biocatalytic Synthesis of Chiral Alcohol Intermediates for Beta-3 Agonists
Scalable Biocatalytic Synthesis of Chiral Alcohol Intermediates for Beta-3 Agonists
The pharmaceutical industry's relentless pursuit of highly potent and selective therapeutics has placed chiral intermediates at the forefront of modern drug discovery and development. Specifically, the synthesis of beta-3 adrenergic receptor agonists, such as the promising antidepressant and anti-obesity candidate SR58611 and Solabegron, relies critically on the availability of optically pure building blocks. Patent CN101302552B introduces a groundbreaking biocatalytic methodology for the preparation of (R)-2-chloro-1-(3-chlorophenyl) ethanol, a pivotal chiral synthon in these therapeutic pathways. This technology leverages the exquisite stereoselectivity of microbial carbonyl reductases to transform prochiral ketones into high-value chiral alcohols with exceptional optical purity. By shifting away from traditional stoichiometric chemical reductions, this approach offers a sustainable, cost-effective, and scalable solution for the manufacture of complex pharmaceutical intermediates, addressing the growing demand for reliable pharmaceutical intermediate supplier capabilities in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral halogenated aryl alcohols has been plagued by significant inefficiencies inherent to classical chemical synthesis and resolution techniques. Traditional methods often rely on the use of precious metal catalysts, such as rhodium complexes, which not only incur exorbitant raw material costs but also pose severe challenges regarding catalyst recovery and heavy metal contamination in the final API. Furthermore, chemical resolution strategies, while capable of separating enantiomers, suffer from a theoretical maximum yield of only 50%, as the undesired enantiomer is typically discarded or requires complex racemization cycles. These processes frequently necessitate harsh reaction conditions, including extreme temperatures and pressures, and generate substantial volumes of hazardous organic waste, creating a heavy burden on environmental compliance and operational expenditure. The reliance on expensive alkaloids as resolving agents further exacerbates the cost structure, making these conventional routes economically unviable for large-scale commercial production of cost-sensitive generic intermediates.
The Novel Approach
In stark contrast, the biocatalytic route disclosed in the patent data represents a paradigm shift towards green and efficient manufacturing. By utilizing specific microbial strains such as Vibrio fluvialis, Alcaligenes faecalis, and Bacillus thuringiensis, the process harnesses nature's catalytic power to achieve asymmetric reduction under mild aqueous conditions. This biological approach eliminates the need for toxic heavy metals and avoids the 50% yield ceiling of resolution methods by directly synthesizing the desired (R)-enantiomer with enantiomeric excess values exceeding 99%. The integration of whole-cell biocatalysts simplifies the downstream processing, as the enzymes are contained within the biomass, facilitating easier separation. Moreover, the ability to operate in single aqueous phases or benign water-organic biphasic systems drastically reduces the consumption of volatile organic solvents, aligning perfectly with the principles of green chemistry and offering a robust pathway for cost reduction in API manufacturing.
Mechanistic Insights into Microbial Carbonyl Reductase Catalysis
The core of this technological advancement lies in the highly specific activity of carbonyl reductases produced by the screened microbial strains. These enzymes facilitate the hydride transfer from a cofactor (typically NADPH or NADH) to the carbonyl carbon of the substrate, 2-chloro-1-(3-chlorophenyl) ethanone, with strict stereocontrol. What makes this process particularly ingenious is the self-sufficient cofactor regeneration system; the microbial cells utilize added glucose as a hydrogen donor to continuously recycle the oxidized cofactor back to its reduced state, thereby sustaining the catalytic cycle without the need for external addition of expensive coenzymes. This intrinsic regeneration mechanism not only lowers the cost of goods but also simplifies the reaction setup, allowing for high substrate loading concentrations ranging from 50 to 140 mmol/L. The enzyme's active site geometry ensures that the hydride attack occurs exclusively from one face of the planar carbonyl group, resulting in the formation of the (R)-configuration with remarkable fidelity.
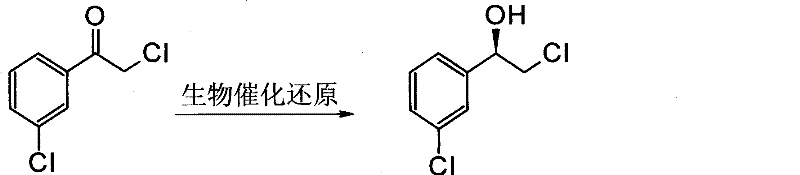
Impurity control in this biocatalytic system is achieved through both enzymatic specificity and process engineering. The high enantioselectivity of the carbonyl reductase minimizes the formation of the (S)-enantiomer, which is the primary chiral impurity of concern. Additionally, the patent describes the strategic use of macroporous resins or organic solvents in a biphasic system to mitigate substrate and product inhibition. Halogenated aromatic compounds can be toxic to microbial cells at high concentrations, potentially leading to cell lysis and the release of intracellular proteases or other contaminants. By adsorbing the hydrophobic substrate and product onto resins like DA201 or partitioning them into an organic phase like dioctyl phthalate, the effective concentration in the aqueous phase surrounding the cells is kept below toxic thresholds. This protects the biocatalyst's integrity throughout the 48-hour reaction window, ensuring consistent conversion rates above 99% and preventing the formation of degradation by-products that often arise from stressed biological systems.
How to Synthesize (R)-2-Chloro-1-(3-Chlorophenyl) Ethanol Efficiently
The implementation of this biocatalytic process requires precise control over fermentation and bioconversion parameters to maximize yield and optical purity. The protocol begins with the cultivation of the selected bacterial strain in a nutrient-rich medium optimized with corn steep liquor and yeast extract to induce high levels of enzyme expression. Following biomass accumulation, the cells are introduced to the reaction vessel containing the ketone substrate, glucose, and a buffering system maintained at pH 6.5. The detailed standardized synthetic steps, including specific incubation times, temperature gradients, and work-up procedures involving ethyl acetate extraction and resin elution, are outlined below to ensure reproducibility and quality consistency for industrial partners seeking to adopt this technology.
- Cultivate selected microbial strains such as Alcaligenes faecalis ATCC 15554 in a optimized medium containing yeast extract and corn steep liquor to induce carbonyl reductase expression.
- Prepare the reaction system using a phosphate buffer (pH 6.5) potentially enhanced with macroporous resin or organic solvents to manage substrate toxicity and improve solubility.
- Conduct the asymmetric reduction of 2-chloro-1-(3-chlorophenyl) ethanone at controlled temperatures (20-50°C) with glucose supplementation, followed by extraction and purification to isolate the high-purity chiral alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this biocatalytic platform offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the risk of supply chain disruptions associated with rare earth mining and refining. Furthermore, the simplified downstream processing, characterized by the absence of complex resolution steps and heavy metal scavenging units, leads to a drastically simplified production workflow. This streamlining translates directly into reduced operational expenditures and shorter batch cycle times, enhancing the overall agility of the manufacturing supply chain. The robustness of the fermentation process also ensures a stable and continuous supply of high-quality intermediates, reducing the lead time for high-purity pharmaceutical intermediates and securing production schedules against raw material volatility.
- Cost Reduction in Manufacturing: The proprietary biocatalytic route fundamentally alters the cost structure by replacing expensive stoichiometric reagents and chiral resolving agents with renewable biological catalysts. Since the process does not require the external addition of costly cofactors like NADH and utilizes inexpensive glucose for regeneration, the variable cost per kilogram of product is significantly lowered. Additionally, the high atom economy of the direct reduction avoids the 50% material loss inherent in resolution methods, effectively doubling the theoretical output from the same amount of starting ketone material. These factors combine to deliver substantial cost savings that can be passed down the value chain, making the final API more competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: Reliance on fermentation-based production decouples the supply of this critical intermediate from the fluctuations of the petrochemical industry that often impact synthetic reagent availability. The microbial strains used are robust and can be stored and propagated easily, ensuring a consistent and reliable source of catalytic activity. The ability to scale fermentation from laboratory shake flasks to large industrial fermenters allows for flexible capacity expansion to meet surging demand without the need for extensive new hardware installation. This scalability ensures that partners can rely on a steady flow of materials, minimizing the risk of stockouts that could halt downstream API synthesis.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a superior profile compared to traditional chemical synthesis. The use of aqueous systems and the avoidance of toxic heavy metals significantly reduce the generation of hazardous waste streams, lowering disposal costs and simplifying regulatory compliance. The mild reaction conditions (ambient pressure and moderate temperatures) reduce energy consumption and enhance operational safety by minimizing the risk of thermal runaways. This green manufacturing footprint not only aligns with corporate sustainability goals but also facilitates smoother regulatory approvals in stringent markets where environmental impact assessments are critical for vendor qualification.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the biocatalytic production of chiral alcohols. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing transparency into the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their existing drug substance manufacturing workflows.
Q: What are the advantages of this biocatalytic method over traditional chemical resolution?
A: Unlike traditional chemical resolution which inherently wastes 50% of the unwanted enantiomer and requires expensive resolving agents, this biocatalytic method achieves direct asymmetric synthesis with >99% e.e. and near-quantitative conversion, significantly reducing raw material costs and waste disposal burdens.
Q: Does this process require the addition of expensive cofactors like NADH?
A: No, a key innovation of this patented process is that the whole-cell biocatalyst utilizes an intrinsic coenzyme regeneration system fueled by glucose, eliminating the need for external addition of costly cofactors like NADH or NADPH during the reaction.
Q: How is substrate toxicity managed during the fermentation process?
A: The process employs a biphasic system (water/organic solvent) or the addition of macroporous resins (such as DA201 or D101) which adsorb the substrate and product, thereby reducing their toxic effects on the microbial cells and allowing for higher substrate loading concentrations up to 140 mmol/L.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-2-Chloro-1-(3-Chlorophenyl) Ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation beta-3 agonists depends on the secure supply of high-quality chiral building blocks. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced chiral chromatography to guarantee that every batch of (R)-2-chloro-1-(3-chlorophenyl) ethanol meets the exacting standards required for clinical and commercial API synthesis. Our commitment to quality assurance ensures that our clients receive materials with consistent optical purity and chemical identity, minimizing the risk of batch failures in downstream processing.
We invite potential partners to engage with our technical procurement team to discuss how this innovative biocatalytic technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to our bio-based route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the performance and profitability of your pharmaceutical development pipeline.
