Revolutionizing Beta-Cyano Intermediate Production via Metal-Free Electrochemical Synthesis
Revolutionizing Beta-Cyano Intermediate Production via Metal-Free Electrochemical Synthesis
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards greener, more sustainable methodologies, a trend vividly exemplified by the breakthrough technology disclosed in patent CN113862708A. This pivotal intellectual property introduces a novel electrochemical synthesis method for producing beta-cyano-bis-phenylsulfonyl imide compounds, a class of versatile intermediates critical for the development of advanced pharmaceuticals and functional materials. Unlike traditional synthetic pathways that rely heavily on stoichiometric amounts of toxic reagents and precious metal catalysts, this innovation leverages direct electrical energy to drive chemical transformations under remarkably mild conditions. By utilizing simple olefins, cyanobenzoxazolone, and bisbenzenesulfonimide as feedstocks in an acetonitrile solvent system, the process achieves high efficiency without generating hazardous waste streams. For R&D directors and procurement strategists alike, this represents a significant opportunity to streamline supply chains and enhance the environmental profile of complex molecule production. The core structural motif of these valuable intermediates is depicted below, highlighting the precise arrangement of the cyano and sulfonimide functionalities that confer their unique reactivity.
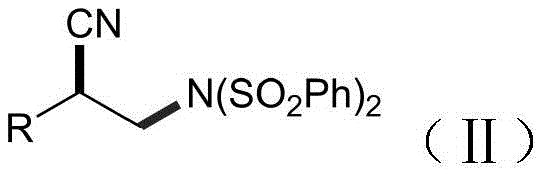
Furthermore, the operational simplicity of this electrochemical approach cannot be overstated, as it proceeds at room temperature under an inert atmosphere without the need for external initiators or complex light sources. The absence of transition metals in the catalytic cycle ensures that the final product is devoid of heavy metal contamination, a critical quality attribute for pharmaceutical intermediates intended for human therapeutic applications. This technological advancement not only addresses the growing regulatory pressure to reduce toxic waste but also offers a robust platform for the diversification of chemical libraries through the modification of the R group on the olefin substrate. As the industry moves towards more sustainable manufacturing practices, the adoption of such electrode-driven methodologies positions forward-thinking organizations at the forefront of innovation, enabling them to meet stringent purity specifications while minimizing their ecological footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-cyano-bis-phenylsulfonyl imide compounds has been fraught with significant technical and safety challenges that hinder efficient large-scale production. Conventional protocols typically depend on the conjugate cyanidation of alpha,beta-unsaturated ketones using highly toxic cyanide sources such as trimethylsilyl cyanide (TMSCN), potassium cyanide, or hydrocyanic acid. These reagents pose severe health risks to laboratory personnel and require specialized containment infrastructure to prevent accidental exposure, thereby inflating operational costs and complicating regulatory compliance. Moreover, the prevailing state-of-the-art methods often necessitate the use of transition metal catalysts, specifically copper-based systems paired with ligands like phenanthroline, to facilitate the carbon-carbon bond formation. While effective in small-scale settings, these metal-catalyzed processes introduce a persistent impurity profile that demands rigorous and costly downstream purification steps to meet the stringent residual metal limits imposed by global pharmacopoeias. The reliance on such catalysts also limits the substrate scope, as sensitive functional groups may be incompatible with the harsh oxidative conditions or the metal center itself. A representative example of this traditional copper-catalyzed pathway is illustrated in the following reaction scheme, which underscores the complexity and reagent burden associated with legacy manufacturing techniques.
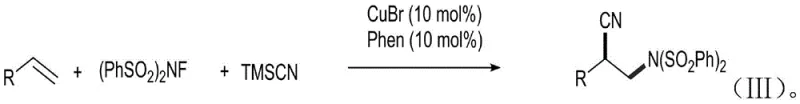
The Novel Approach
In stark contrast to these cumbersome legacy methods, the electrochemical synthesis strategy outlined in patent CN113862708A offers a transformative solution that fundamentally reimagines the reaction mechanism. By replacing chemical oxidants and metal catalysts with electrons as the primary reagent, this new approach eliminates the root causes of toxicity and contamination inherent in conventional routes. The reaction utilizes readily available olefins or their derivatives, which serve as abundant and cost-effective starting materials, reacting them with cyanobenzoxazolone and bisbenzenesulfonimide in a unified electrochemical cell. This methodology operates under ambient conditions, specifically at room temperature, which drastically reduces energy consumption compared to thermal processes requiring high heat or cryogenic cooling. The elimination of copper catalysts not only simplifies the workup procedure by removing the need for metal scavengers but also broadens the functional group tolerance, allowing for the synthesis of a wider array of derivatives including those with halogen substituents. Consequently, this novel approach delivers a cleaner, safer, and more economically viable pathway for producing high-value intermediates, aligning perfectly with the principles of green chemistry and sustainable manufacturing.
Mechanistic Insights into Electrochemical Oxidative Functionalization
The success of this electrochemical transformation lies in its elegant mechanism, which facilitates the formation of carbon-nitrogen and carbon-carbon bonds through a proton-coupled electron transfer (PCET) process. At the anode, typically constructed from platinum, the oxidation of the bisbenzenesulfonimide generates a reactive nitrogen-centered radical species via the loss of an electron and a proton. This radical intermediate is stabilized by the adjacent sulfonyl groups, allowing it to selectively attack the electron-rich double bond of the olefin substrate. Simultaneously, the cyanobenzoxazolone serves as a safe and stable source of the cyano group, participating in the radical addition cascade to construct the beta-cyano framework. The use of tetrabutylammonium acetate as the electrolyte plays a dual role: it ensures sufficient conductivity within the acetonitrile medium and acts as a base to facilitate the deprotonation steps essential for the catalytic cycle. This intricate interplay of electrochemical potentials and radical chemistry allows for precise control over the reaction trajectory, minimizing side reactions such as polymerization or over-oxidation that often plague traditional free-radical processes. For technical teams, understanding this mechanism is crucial for optimizing parameters such as current density and electrode spacing to maximize yield and selectivity.
From an impurity control perspective, the metal-free nature of this electrochemical system provides a distinct advantage in managing the quality of the final product. In traditional copper-catalyzed reactions, trace amounts of metal can coordinate with the product or intermediates, leading to difficult-to-remove complexes that compromise purity. By entirely excluding transition metals from the reaction matrix, the electrochemical method inherently produces a cleaner crude mixture, significantly reducing the burden on downstream purification units like chromatography or crystallization. Furthermore, the mild reaction conditions prevent the degradation of sensitive functional groups on the olefin substrate, such as the halogen atoms found in chloro- or bromo-styrenes, which might otherwise undergo unwanted substitution or elimination under harsher thermal or acidic conditions. This high level of chemoselectivity ensures that the impurity profile remains predictable and manageable, facilitating easier validation and regulatory approval for pharmaceutical applications. The ability to tune the reaction potential also offers a unique handle for process engineers to suppress specific side pathways, further enhancing the robustness of the manufacturing process.
How to Synthesize Beta-Cyano-Bis-Phenylsulfonyl Imide Efficiently
Implementing this electrochemical synthesis route requires careful attention to the preparation of the electrolytic cell and the precise control of reaction parameters to ensure reproducibility and safety. The process begins with the dissolution of the olefin derivative, cyanobenzoxazolone, and bisbenzenesulfonimide in dry acetonitrile under a strictly inert nitrogen atmosphere to prevent interference from oxygen or moisture. Once the homogeneous mixture is formed, the supporting electrolyte, tetrabutylammonium acetate, is introduced to establish the necessary ionic conductivity for the electrochemical cell. The reaction is then driven by applying a constant current across a platinum anode and a graphite cathode, a setup that balances reactivity with cost-effectiveness. Detailed standard operating procedures regarding electrode dimensions, current density, and reaction duration are critical for achieving the optimal yields reported in the patent data. For a comprehensive guide on the exact molar ratios and specific operational steps required to replicate this high-efficiency synthesis, please refer to the standardized protocol below.
- Prepare the reaction mixture by dissolving olefin derivatives, cyanobenzoxazolone, and bisbenzenesulfonimide in acetonitrile under a nitrogen atmosphere.
- Add tetrabutylammonium acetate as the electrolyte and perform constant current electrolysis using a platinum anode and graphite cathode.
- Upon completion, quench the reaction with water, extract with ethyl acetate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrochemical technology translates into tangible strategic benefits that extend far beyond the laboratory bench. The most immediate impact is observed in the realm of cost reduction, where the elimination of expensive transition metal catalysts and toxic cyanide reagents directly lowers the bill of materials. Traditional methods often incur hidden costs associated with the disposal of hazardous waste and the extensive purification required to remove metal residues, expenses that are effectively nullified by this metal-free approach. By simplifying the raw material list to commodity chemicals like olefins and acetonitrile, manufacturers can leverage existing supply chains and mitigate the risk of price volatility associated with specialized reagents. This streamlined input profile enhances the overall economic resilience of the production process, allowing for more competitive pricing in the global market for fine chemical intermediates.
- Cost Reduction in Manufacturing: The removal of copper catalysts and toxic cyanide sources fundamentally alters the cost structure of production by eliminating the need for expensive metal scavengers and complex waste treatment protocols. Without the requirement for rigorous heavy metal clearance steps, the downstream processing becomes significantly faster and less resource-intensive, leading to substantial savings in both time and consumables. Additionally, the use of electricity as the primary driving force is inherently more energy-efficient than thermal heating or high-pressure systems, further contributing to lower utility costs per kilogram of product. These cumulative efficiencies result in a leaner manufacturing process that maximizes margin potential while maintaining high product quality standards.
- Enhanced Supply Chain Reliability: Relying on widely available starting materials such as substituted styrenes and common solvents reduces dependency on single-source suppliers of exotic catalysts or hazardous reagents. This diversification of the supply base minimizes the risk of production stoppages due to raw material shortages or logistical disruptions, ensuring a more consistent flow of goods to customers. The robustness of the electrochemical method also means that production can be easily scaled up or down in response to market demand without the need for specialized reactor configurations, providing greater agility in inventory management. Consequently, partners can enjoy shorter lead times and more reliable delivery schedules, strengthening the overall resilience of the pharmaceutical supply chain.
- Scalability and Environmental Compliance: The simplicity of the electrochemical setup, which does not require complex photochemical equipment or high-pressure vessels, makes it exceptionally well-suited for commercial scale-up from pilot plants to multi-ton facilities. The absence of toxic byproducts and heavy metal waste aligns perfectly with increasingly stringent environmental regulations, reducing the regulatory burden and liability associated with chemical manufacturing. This green profile not only facilitates smoother permitting processes but also enhances the brand reputation of companies committed to sustainable practices. As the industry pivots towards net-zero goals, adopting such energy-efficient and waste-minimizing technologies positions organizations as leaders in responsible chemical stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of beta-cyano-bis-phenylsulfonyl imide compounds, drawing directly from the experimental data and beneficial effects described in the patent literature. These insights are designed to clarify the operational feasibility and strategic value of this innovative method for stakeholders evaluating its integration into their manufacturing portfolios. Understanding the nuances of substrate compatibility, reaction conditions, and product quality is essential for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of this electrochemical method over traditional copper-catalyzed routes?
A: The electrochemical method eliminates the need for toxic cyanide reagents and expensive transition metal catalysts like copper, resulting in products free from heavy metal residues and significantly reducing purification costs.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process utilizes standard electrolytic cells and common solvents like acetonitrile, operating at room temperature without complex photochemical equipment, making it highly suitable for commercial scale-up.
Q: What is the substrate scope for the olefin starting materials?
A: The method demonstrates broad compatibility with various styrene derivatives, including those with electron-donating groups like methyl and electron-withdrawing groups such as fluoro, chloro, bromo, and trifluoromethyl.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Cyano-Bis-Phenylsulfonyl Imide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the electrochemical synthesis method detailed in patent CN113862708A and are fully equipped to bring this technology to commercial reality for our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of beta-cyano intermediates meets the highest standards required for pharmaceutical applications. Our commitment to excellence extends beyond mere production; we actively collaborate with clients to optimize process parameters and ensure that the environmental and economic benefits of this metal-free route are fully realized in the final supply chain.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can address your specific sourcing challenges and cost objectives. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the financial advantages associated with switching to this electrochemical method. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project requirements. Let us help you secure a reliable supply of high-purity intermediates while advancing your sustainability goals through cutting-edge chemical innovation.
