Scalable Production of 2,7-Dimethyl-2,4,6-octatriene-1,8-dialdehyde via Novel Grignard Pathway
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways for synthesizing critical carotenoid intermediates, and the recent disclosure in patent CN108752178B represents a significant leap forward in this domain. This patent details a robust, five-step synthesis for 2,7-dimethyl-2,4,6-octatriene-1,8-dialdehyde, commonly known as decarene aldehyde, which serves as a pivotal building block for high-value compounds like beta-carotene, astaxanthin, and lycopene. Unlike previous methodologies that struggled with low selectivity and hazardous reagents, this new approach leverages a streamlined Grignard-based strategy that maximizes atom economy while minimizing waste generation. For R&D directors and procurement specialists alike, the implications are profound, offering a route that not only simplifies the synthetic tree but also drastically improves the overall mass balance of the production process. By shifting away from complex acetal protections and towards direct functionalization, this technology addresses the longstanding bottlenecks that have plagued the supply of high-purity pharma intermediates for decades.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of decarene aldehyde has been fraught with technical challenges that hindered efficient commercial scale-up of complex polymer additives and pharmaceutical precursors. One of the most cited legacy routes, depicted in various US patents such as US20020128520, relies on furan as a starting material, which undergoes a two-step methanol addition to form 1,1,4,4-tetramethoxy-2-butene. This intermediate must then be condensed with propenyl methyl ether under Lewis acid catalysis, a process notorious for generating significant amounts of polymeric byproducts through telomerization side reactions. Furthermore, the reliance on elemental bromine in early stages introduces severe safety hazards and toxicity concerns, necessitating expensive containment and waste treatment protocols. The resulting diacetal structures often require harsh acidic conditions for deprotection, leading to further degradation of the sensitive conjugated system and capping the total yield at unacceptably low levels, often below 25%.
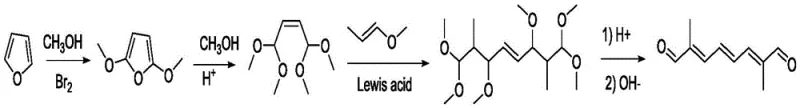
The Novel Approach
In stark contrast, the methodology outlined in CN108752178B bypasses these intricate protection-deprotection cycles entirely by utilizing a direct Grignard coupling strategy. The process initiates with the formation of a bis-Grignard reagent from 1,4-dibromo-2-butene, which is subsequently reacted with ethylene oxide to extend the carbon chain efficiently. This fundamental shift in synthetic logic eliminates the need for unstable bromine additions and avoids the formation of difficult-to-remove polymeric impurities associated with acetal chemistry. By focusing on robust C-C bond forming reactions that proceed with high fidelity, the new route ensures a much cleaner reaction profile. The simplicity of the workup procedures, which primarily involve standard distillation and filtration rather than complex chromatographic separations, translates directly into reduced operational expenditures and a more reliable supply chain for high-purity OLED material and agrochemical intermediate manufacturers who demand consistent quality.
Mechanistic Insights into Grignard Coupling and Catalytic Isomerization
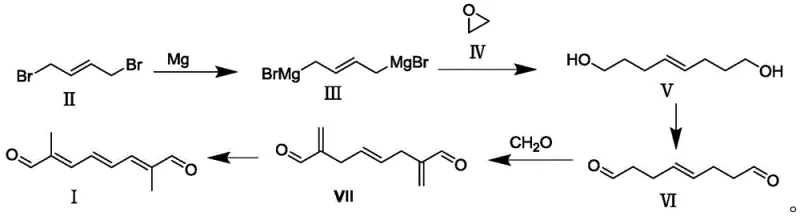
The core of this technological breakthrough lies in the precise control of the Grignard formation and the subsequent oxidative transformations. In the first stage, 1,4-dibromo-2-butene reacts with magnesium turnings in tetrahydrofuran at a controlled temperature of 50-60°C, initiated by trace amounts of iodine to ensure rapid onset without runaway exotherms. The resulting organomagnesium species is highly reactive, allowing for the nucleophilic attack on ethylene oxide to proceed smoothly, extending the chain to form the diol intermediate (Compound V) with yields exceeding 95%. Following this, a selective oxidation step utilizing a TEMPO catalyst and molecular oxygen converts the primary alcohol groups into aldehydes (Compound VI) under mild conditions, preserving the internal double bond geometry. This chemoselectivity is crucial for maintaining the integrity of the molecule before the final conjugation steps, ensuring that the impurity profile remains manageable even at multi-ton scales.
The final stages of the synthesis involve an aldol condensation with formaldehyde followed by a catalytic hydroisomerization, which are critical for establishing the desired conjugated triene system. The aldol reaction, performed in aqueous sodium hydroxide, links two units of the oxidized intermediate, creating the central carbon framework of the target molecule. However, the true elegance of the process is revealed in the final hydroisomerization step, where a supported palladium catalyst facilitates the migration of double bonds under hydrogen pressure (1-5 MPa). This step not only saturates any remaining terminal unsaturation but also thermodynamically drives the formation of the stable, fully conjugated 2,4,6-octatriene system. The use of a heterogeneous catalyst allows for easy removal via filtration, preventing metal contamination in the final API intermediate, a critical specification for downstream pharmaceutical applications.
How to Synthesize 2,7-Dimethyl-2,4,6-octatriene-1,8-dialdehyde Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the Grignard reagent and the control of the oxidation environment to prevent over-oxidation to carboxylic acids. The patent provides detailed parameters for solvent selection, recommending tetrahydrofuran for the Grignard step and dichloromethane for the oxidation to maximize solubility and reaction rates. Operators must maintain strict temperature controls during the exothermic addition of ethylene oxide and the subsequent acidolysis to ensure high purity of the diol intermediate. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and processing times required to replicate the high yields reported in the intellectual property.
- React 1,4-dibromo-2-butene with magnesium in THF at 50-60°C to form the bis-Grignard reagent.
- Add ethylene oxide to the Grignard reagent followed by acidolysis to obtain the diol intermediate (Compound V).
- Oxidize Compound V using oxygen and a TEMPO catalyst to form the dialdehyde (Compound VI).
- Perform aldol condensation with formaldehyde under basic conditions to generate the conjugated system (Compound VII).
- Subject Compound VII to hydroisomerization using a palladium catalyst under hydrogen pressure to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers substantial strategic benefits beyond mere technical superiority. The primary advantage stems from the utilization of commodity feedstocks; 1,4-dibromo-2-butene and ethylene oxide are produced on a massive global scale, ensuring that raw material availability is not a bottleneck for production planning. This stands in sharp contrast to older routes that depended on specialized vitamin A intermediates or custom-synthesized acetals, which often suffered from long lead times and price volatility. By anchoring the supply chain in widely available bulk chemicals, manufacturers can secure long-term contracts at stable prices, effectively insulating their operations from market fluctuations that typically plague niche fine chemical sectors.
- Cost Reduction in Manufacturing: The elimination of expensive protecting group chemistry, such as neopentyl glycol acetals used in competing patents, removes several unit operations from the production schedule. Each removed step corresponds to a direct saving in labor, energy, and solvent consumption, while the high yield of the Grignard coupling minimizes the loss of valuable starting materials. Furthermore, the avoidance of toxic bromine handling reduces the regulatory burden and costs associated with hazardous waste disposal, contributing to a leaner and more cost-effective manufacturing model that enhances overall margin potential.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, particularly the use of standard heterogeneous catalysts like Pd/C in the final step, ensures that the process is less susceptible to batch-to-batch variability. This reliability is paramount for maintaining continuous supply to downstream customers in the nutraceutical and pharmaceutical sectors, where interruptions can be catastrophic. The simplified purification protocols, relying on crystallization and distillation rather than complex chromatography, further accelerate the throughput time, allowing facilities to respond more agilely to sudden spikes in demand without compromising on the stringent purity specifications required by global regulators.
- Scalability and Environmental Compliance: From an environmental perspective, the route generates significantly less saline waste compared to the stoichiometric base usage in older aldol condensations, and the use of molecular oxygen as a terminal oxidant produces water as the only byproduct. This alignment with green chemistry principles facilitates easier permitting and compliance with increasingly strict environmental regulations in major manufacturing hubs. The process is inherently designed for scale, with exotherms that are manageable in large reactors, making the commercial scale-up of complex fragrance and flavor intermediates a straightforward engineering task rather than a scientific risk.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology, derived directly from the comparative data and experimental examples provided in the source documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement officers negotiating supply agreements. The answers reflect the specific advantages in yield, safety, and operational simplicity that distinguish this method from the prior art.
Q: How does this new Grignard-based route compare to traditional furan methods in terms of yield?
A: Traditional furan-based routes often suffer from side reactions like telomerization and require harsh bromination steps, resulting in total yields as low as 15-21%. The novel pathway described in CN108752178B achieves a significantly higher total yield of 78-84% by utilizing readily available 1,4-dibromo-2-butene and avoiding complex protection-deprotection sequences.
Q: What are the key raw material advantages for supply chain stability?
A: The process relies on commodity chemicals such as 1,4-dibromo-2-butene, ethylene oxide, and formaldehyde, which are widely available in the global chemical market. This contrasts with older methods that require specialized intermediates like 1,1,4,4-tetramethoxy-2-butene or vitamin A precursors, thereby reducing procurement risks and lead times.
Q: Is the hydroisomerization step suitable for large-scale manufacturing?
A: Yes, the final step utilizes a supported palladium catalyst (e.g., Pd/C) under moderate hydrogen pressure (1-5 MPa) and temperatures of 80-100°C. These conditions are standard in fine chemical manufacturing, allowing for straightforward scale-up from pilot plants to multi-ton production without requiring exotic high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,7-Dimethyl-2,4,6-octatriene-1,8-dialdehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality that spans from gram-scale development to full industrial output. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory curiosity to market-ready product is seamless and efficient. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2,7-dimethyl-2,4,6-octatriene-1,8-dialdehyde meets the exacting standards required for carotenoid synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your operation, alongside specific COA data and route feasibility assessments that demonstrate our capability to deliver consistent, high-quality supply. Let us collaborate to optimize your supply chain and secure a competitive advantage in the global market for high-value fine chemical intermediates.
