Advanced Alpha-Functionalization of N-Phenyl-3-Butenamide for High-Value Pharmaceutical Intermediates
Introduction to Next-Generation Amide Functionalization
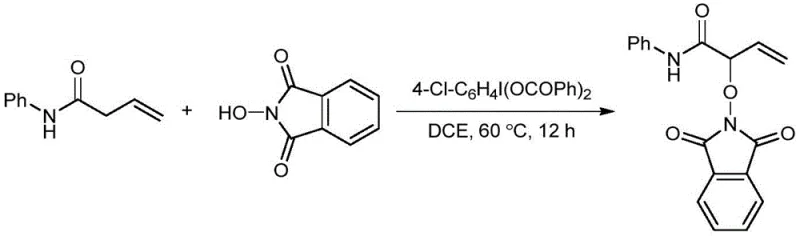
The landscape of fine chemical synthesis is constantly evolving towards greener, more efficient methodologies that satisfy the rigorous demands of modern pharmaceutical manufacturing. A significant breakthrough in this domain is documented in patent CN113845466A, which discloses a novel preparation method for alpha-oxo-N-phenyl-3-butenamide compounds. This technology leverages the unique reactivity of hypervalent iodine reagents to achieve selective oxidation at the alpha-position of N-phenylbut-3-enamine derivatives. For R&D directors and process chemists, this represents a pivotal shift away from harsh, non-selective oxidation protocols towards a controlled, catalytic system that operates under remarkably mild thermal conditions. The ability to install complex oxygenated functionality with high fidelity opens new avenues for constructing diverse libraries of bioactive molecules, particularly those required for advanced drug discovery programs where structural complexity is paramount.
Furthermore, the environmental profile of this synthesis aligns perfectly with the increasing global pressure to reduce the ecological footprint of chemical manufacturing. By utilizing organic solvents like 1,2-dichloroethane and avoiding stoichiometric amounts of toxic heavy metals, the process mitigates the burden on wastewater treatment facilities. The reported yields, reaching as high as 87% in optimized examples, demonstrate that sustainability does not have to come at the cost of efficiency. For procurement managers, this translates to a more reliable supply of high-value intermediates with reduced risk of batch failure due to impurity profiles often associated with cruder oxidation methods. This patent serves as a foundational document for understanding how modern organocatalysis and hypervalent iodine chemistry can be harnessed to solve long-standing challenges in the production of complex amide scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of functionalized aniline derivatives and related amide intermediates has relied on archaic and often hazardous methodologies. Traditional pathways, such as the iron powder reduction method, are plagued by severe environmental drawbacks, including the generation of massive quantities of iron sludge that require costly disposal procedures. Similarly, electrolytic reduction methods, while technically feasible, demand high energy inputs and strict control parameters that are difficult to maintain on a large industrial scale, leading to inconsistent product quality. Biological fermentation, though green, often suffers from prolonged production cycles and low space-time yields, making it economically unviable for high-volume commodity chemicals. Moreover, conventional oxidation strategies frequently employ stoichiometric chromium or manganese reagents, which introduce toxic heavy metal residues into the final product, necessitating expensive and time-consuming purification steps to meet stringent pharmaceutical purity specifications. These legacy methods create significant bottlenecks in the supply chain, increasing lead times and exposing manufacturers to regulatory risks associated with heavy metal limits.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a sophisticated oxidative functionalization strategy that circumvents these historical pitfalls. By employing a hypervalent iodine reagent, specifically p-chloro(dibenzoic acid)iodobenzene, in conjunction with N-hydroxyphthalimide (NHPI), the process achieves highly selective activation of the alpha-C-H bond. This approach operates at a moderate temperature of 60°C, eliminating the need for extreme thermal stress that can degrade sensitive functional groups like the terminal alkene present in the substrate. The reaction mechanism proceeds through a radical pathway that is tightly controlled by the iodine species, ensuring that over-oxidation to carboxylic acids or degradation of the amide backbone is minimized. This precision allows for the direct conversion of simple N-phenyl-3-butenamides into valuable alpha-oxygenated derivatives in a single pot. The result is a streamlined workflow that reduces the number of unit operations, lowers solvent consumption, and delivers a product with a superior impurity profile, directly addressing the pain points of both production efficiency and environmental compliance.
Mechanistic Insights into Hypervalent Iodine-Mediated Alpha-Oxidation
The core of this technological advancement lies in the synergistic interaction between the hypervalent iodine oxidant and the N-hydroxyphthalimide mediator. Mechanistically, the hypervalent iodine species acts as a potent one-electron oxidant capable of generating phthalimide-N-oxyl (PINO) radicals from N-hydroxyphthalimide under thermal conditions. These PINO radicals are highly effective hydrogen atom transfer (HAT) agents that selectively abstract the relatively weak allylic or benzylic hydrogen at the alpha-position of the amide substrate. This abstraction generates a stabilized carbon-centered radical intermediate, which is subsequently trapped by the iodine species or the phthalimide moiety to form the new C-O or C-N bond. The use of 4-Cl-C6H4I(OCOPh)2 is particularly strategic; the electron-withdrawing chloro substituent enhances the oxidizing power of the iodine center, facilitating the reaction at lower temperatures compared to unsubstituted analogues. This mechanistic pathway avoids the formation of high-energy cationic intermediates that often lead to rearrangement by-products, thereby ensuring high regioselectivity for the alpha-position.
From an impurity control perspective, this radical mechanism offers distinct advantages over ionic oxidation pathways. Because the reaction does not rely on strong Brønsted or Lewis acids, acid-sensitive groups such as the terminal vinyl group remain intact, preventing polymerization or hydration side reactions that are common in acidic media. Furthermore, the by-products of the hypervalent iodine reduction are typically iodobenzene derivatives, which are organic-soluble and easily separated from the polar product during aqueous workup or chromatography. The patent data indicates that column chromatography using ethyl acetate and n-hexane effectively separates the target alpha-functionalized product from any unreacted starting material or minor over-oxidized by-products. This clean reaction profile is critical for pharmaceutical applications, where the presence of genotoxic impurities or heavy metals must be kept below parts-per-million levels. The ability to achieve yields up to 87% with such high selectivity underscores the robustness of this catalytic cycle and its suitability for generating high-purity intermediates required for GMP manufacturing.
How to Synthesize Alpha-Oxo-N-Phenyl-3-Butenamide Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and thermal management to maximize the efficiency of the hypervalent iodine cycle. The process begins with the precise mixing of the oxidant, the amide substrate, and the NHPI mediator in a suitable chlorinated solvent, followed by a controlled heating period. The reaction kinetics are dependent on the concentration of the radical mediator and the stability of the iodine reagent at the operating temperature. While the patent provides specific laboratory-scale examples, scaling this reaction involves optimizing the heat transfer rates to maintain the uniform 60°C environment necessary for consistent radical generation. Detailed standardized synthetic steps, including specific molar ratios and workup procedures, are essential for reproducibility and are outlined in the technical guide below.
- Prepare the reaction mixture by combining the hypervalent iodine oxidant (4-Cl-C6H4I(OCOPh)2), N-phenyl-3-butenamide substrate, and N-hydroxyphthalimide catalyst in an organic solvent such as 1,2-dichloroethane.
- Heat the mixture in an oil bath at 60°C with continuous stirring (approx. 600 r/s) for 12 hours to facilitate the alpha-position oxidation.
- Quench the reaction with water, extract the organic phase using ethyl acetate or dichloromethane, and purify the crude product via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hypervalent iodine-mediated synthesis offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of transition metal catalysts removes a significant cost center associated with the purchase of precious metals like palladium or rhodium, as well as the downstream costs of metal scavenging and validation. This shift to organocatalytic reagents stabilizes raw material costs, insulating the supply chain from the volatility of the precious metals market. Additionally, the mild reaction conditions reduce energy consumption significantly compared to high-temperature reflux or high-pressure hydrogenation processes, contributing to lower overall manufacturing overheads. The simplified purification process, driven by the clean reaction profile, reduces solvent usage and waste disposal fees, further enhancing the cost-effectiveness of the production route.
- Cost Reduction in Manufacturing: The replacement of expensive and toxic heavy metal oxidants with recyclable or cheaper hypervalent iodine reagents leads to substantial cost savings in raw materials. By avoiding the need for specialized equipment to handle corrosive acids or high-pressure hydrogen, capital expenditure for plant infrastructure is also minimized. The high selectivity of the reaction reduces the loss of valuable starting materials to side products, improving the overall mass balance and atom economy of the process. Consequently, the cost per kilogram of the final high-purity intermediate is drastically reduced, allowing for more competitive pricing in the global market for pharmaceutical building blocks.
- Enhanced Supply Chain Reliability: The reagents used in this process, such as N-hydroxyphthalimide and substituted iodobenzenes, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by utility fluctuations or minor variations in raw material quality. This reliability ensures consistent delivery schedules for downstream customers, which is critical for maintaining uninterrupted drug manufacturing lines. Furthermore, the stability of the intermediates allows for safer storage and transportation, mitigating risks associated with hazardous chemical logistics.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard batch reactor operations without the need for exotic flow chemistry setups or extreme conditions. The reduced generation of hazardous waste, particularly heavy metal sludge, simplifies compliance with increasingly stringent environmental regulations such as REACH and TSCA. This environmental compatibility facilitates faster regulatory approvals for new manufacturing sites and reduces the administrative burden on EHS teams. As a result, companies can scale production from pilot plants to multi-ton commercial volumes with greater confidence and speed, meeting market demand efficiently.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alpha-oxidation technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of the synthesis. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of using hypervalent iodine reagents for this synthesis?
A: The use of hypervalent iodine reagents, specifically p-chloro(dibenzoic acid)iodobenzene, offers mild reaction conditions (60°C) and high selectivity compared to traditional heavy metal oxidants. This results in yields up to 87% while minimizing toxic waste and simplifying downstream purification processes.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes common organic solvents like dichloroethane and standard heating equipment (oil baths), avoiding extreme pressures or cryogenic temperatures. The absence of expensive transition metal catalysts significantly reduces raw material costs and supply chain risks associated with critical metals.
Q: How does this method compare to traditional aniline derivative production methods?
A: Unlike traditional electrolytic reduction or iron powder reduction methods which suffer from high energy consumption, severe pollution, and difficult scale-up, this oxidative functionalization pathway is environmentally friendlier. It avoids the generation of large amounts of iron sludge and operates under much milder thermal conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Oxo-N-Phenyl-3-Butenamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the hypervalent iodine-mediated synthesis described in patent CN113845466A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle the specific requirements of hypervalent iodine chemistry, including precise thermal control and advanced separation technologies. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alpha-functionalized amides meets the exacting standards required by global pharmaceutical clients.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this metal-free approach can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data for our reference standards and comprehensive route feasibility assessments. Let us help you secure a sustainable and cost-effective supply chain for your critical amide intermediates.
