Revolutionizing Aprepitant Intermediate Production via Novel Microbacterium Oxydans Biocatalysis
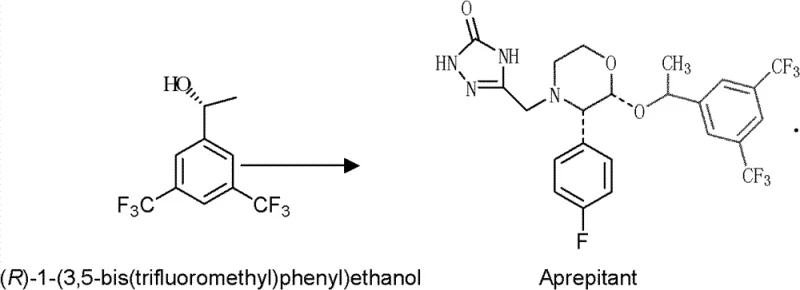
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective pathways for synthesizing chiral intermediates, particularly for blockbuster drugs like Aprepitant. Patent CN102382780B introduces a groundbreaking biocatalytic solution utilizing a novel strain, Microbacterium oxydans C3 (CCTCC M 2010179), to produce optically pure (R)-1-[3,5-bis(trifluoromethyl)phenyl]ethanol. This chiral alcohol serves as a critical precursor for Aprepitant, a neurokinin-1 (NK-1) receptor antagonist used to prevent chemotherapy-induced nausea and vomiting. The patent details a sophisticated whole-cell catalysis method that overcomes the limitations of traditional chemical synthesis and earlier biocatalytic attempts. By leveraging the unique carbonyl reductase activity of this specific bacterial strain, the process achieves exceptional enantioselectivity exceeding 99% ee under mild reaction conditions. This technological advancement represents a significant leap forward for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier partnership that prioritizes both purity and process sustainability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (R)-1-[3,5-bis(trifluoromethyl)phenyl]ethanol has been dominated by chemical catalysis and kinetic resolution strategies, both of which present substantial hurdles for modern green manufacturing. Conventional chemical routes often depend on precious heavy metal catalysts, such as Ruthenium complexes paired with chiral ligands like (S,R)-1-amino-2-indanol, which are not only expensive but also introduce severe toxicity concerns requiring rigorous removal steps to meet regulatory standards. Furthermore, these chemical processes frequently involve hazardous reagents and harsh reaction conditions that complicate waste management and increase operational costs. Alternatively, kinetic resolution of racemic alcohols using lipases is theoretically limited to a maximum yield of 50%, inherently wasting half of the starting material and necessitating complex recycling procedures for the unwanted enantiomer. These inefficiencies create bottlenecks in cost reduction in API manufacturing, making the final drug substance prohibitively expensive and supply chains vulnerable to raw material fluctuations.
The Novel Approach
In stark contrast, the methodology disclosed in CN102382780B utilizes a newly screened strain of Microbacterium oxydans to perform an asymmetric reduction of the prochiral ketone, 3,5-bis-trifluoromethylacetophenone. This biological approach bypasses the need for toxic heavy metals and chiral ligands entirely, relying instead on the intrinsic enzymatic machinery of the microorganism. The process is remarkably versatile, capable of operating with either growing cells or resting cells suspended in buffer, providing flexibility for different production scales. Crucially, this method adheres to the anti-Prelog rule, selectively producing the desired (R)-configuration alcohol, which is often difficult to achieve with common microbial reductases that typically favor the (S)-enantiomer. The reaction system is simplified through the use of inexpensive co-substrates for cofactor regeneration, eliminating the need for external addition of costly NAD(P)H. This streamlined workflow significantly enhances the feasibility of commercial scale-up of complex intermediates, offering a cleaner, safer, and more economically viable alternative to legacy synthetic routes.
Mechanistic Insights into Whole-Cell Asymmetric Reduction
The core of this innovation lies in the specific carbonyl reductase enzymes expressed by the Microbacterium oxydans C3 strain, which exhibit high specificity for the bulky 3,5-bis(trifluoromethyl) phenyl group. Unlike many commercially available oxidoreductases that follow the Prelog rule and generate the (S)-alcohol, this strain possesses a unique stereochemical preference that directly yields the therapeutically active (R)-enantiomer. The mechanism involves the transfer of a hydride ion from the reduced cofactor NAD(P)H to the si-face of the ketone carbonyl group, facilitated by the enzyme's active site architecture. To sustain this reduction continuously without exorbitant costs, the process integrates an efficient cofactor regeneration cycle. By adding isopropanol (Virahol) and glucose to the reaction medium, the microbial cells utilize their endogenous dehydrogenase systems to oxidize these co-substrates, thereby regenerating the consumed NAD(P)H back to its active reduced state. This self-sustaining cycle ensures that only catalytic amounts of cofactors are needed internally, driving the reaction to completion with high atom economy.
Impurity control is another critical aspect where this biocatalytic route excels over chemical counterparts. In chemical hydrogenation, over-reduction or side reactions with the trifluoromethyl groups can occur, leading to difficult-to-remove byproducts. However, the enzymatic active site provides a highly specific pocket that accommodates the substrate precisely, minimizing side reactions and ensuring that the primary byproduct is merely the oxidized form of the co-substrate (e.g., acetone from isopropanol), which is volatile and easily removed. The patent data indicates that the resulting product consistently achieves an enantiomeric excess (ee) value greater than 99%, as confirmed by chiral gas chromatography. This high level of stereochemical purity simplifies downstream purification, often allowing for direct crystallization or simple extraction, thereby reducing the number of unit operations required to obtain high-purity chiral alcohol suitable for pharmaceutical grade applications.
How to Synthesize (R)-1-(3,5-bis(trifluoromethyl)phenyl)ethanol Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a pilot or production setting. The process begins with the cultivation of the Microbacterium oxydans C3 strain in a defined medium containing glucose, peptone, and yeast extract to build sufficient biomass. Following cultivation, the cells are employed either directly as growing cultures or harvested and resuspended as resting cells in a phosphate buffer. The substrate, 3,5-bis-trifluoromethylacetophenone, is introduced into the system, often dissolved in a water-miscible organic solvent like DMSO to enhance solubility, alongside the essential co-substrates for cofactor recycling. The detailed standardized synthesis steps see the guide below.
- Cultivate Microbacterium oxydans C3 in a nutrient-rich medium containing glucose and yeast extract at 27-33°C for 24-48 hours to achieve optimal cell density.
- Prepare the biotransformation system by adding the substrate 3,5-bis-trifluoromethylacetophenone along with co-substrates like isopropanol and glucose for cofactor regeneration.
- Maintain the reaction at controlled temperatures and agitation speeds for 24-88 hours, followed by ethyl acetate extraction to isolate the high-purity chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this biocatalytic platform offers profound strategic benefits beyond mere technical feasibility. The elimination of precious metal catalysts such as Ruthenium removes a significant cost driver and mitigates the risk associated with the volatility of noble metal markets. Furthermore, avoiding heavy metals simplifies the regulatory compliance landscape, as there is no need for extensive and expensive testing to ensure residual metal levels are within strict ICH Q3D guidelines. This inherent safety profile translates directly into cost reduction in API manufacturing by shortening the purification train and reducing the consumption of specialized scavenging resins. The use of fermentation-based production also aligns with global sustainability goals, reducing the environmental footprint associated with hazardous chemical waste disposal, which is increasingly scrutinized by environmental agencies and corporate ESG mandates.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the replacement of expensive chiral ligands and metal catalysts with renewable biological catalysts. The in-situ regeneration of cofactors using cheap bulk chemicals like isopropanol and glucose drastically lowers the raw material cost per kilogram of product. Additionally, the high conversion efficiency reported in the patent embodiments means that less starting material is wasted, improving the overall mass balance and yield. By streamlining the downstream processing through the avoidance of metal removal steps, manufacturers can realize substantial operational savings and improve the gross margin of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Relying on a proprietary microbial strain offers a more secure supply chain compared to sourcing specialized chemical reagents that may be subject to geopolitical trade restrictions or single-source monopolies. The strain can be maintained and propagated indefinitely, ensuring a consistent and renewable source of catalytic activity. The robustness of the whole-cell system allows for flexible production scheduling, as the biocatalyst can be stored or grown on demand. This flexibility helps in reducing lead time for high-purity intermediates, enabling faster response to market demands and preventing stockouts that could disrupt the production of the final drug product.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the scalability of the process, with successful transformations conducted at varying substrate concentrations up to 30g/L. Fermentation technology is a mature and well-understood platform in the fine chemical industry, allowing for seamless scale-up from laboratory flasks to multi-ton industrial fermenters. The aqueous nature of the reaction medium and the biodegradability of the biological components result in a much cleaner waste stream compared to organic synthesis. This facilitates easier wastewater treatment and reduces the environmental compliance burden, making the facility more sustainable and resilient against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this pathway. Understanding these nuances is essential for assessing the fit of this technology within your existing manufacturing infrastructure.
Q: What distinguishes this biocatalytic method from traditional chemical synthesis?
A: Unlike traditional chemical methods that rely on expensive heavy metal catalysts like Ruthenium complexes and chiral ligands, this patent utilizes a whole-cell biocatalyst (Microbacterium oxydans C3) that operates under mild conditions, eliminates heavy metal contamination risks, and achieves superior enantioselectivity (>99% ee) without the theoretical 50% yield limit of kinetic resolution.
Q: How does the process handle cofactor regeneration economically?
A: The process employs an efficient in-situ cofactor regeneration system using inexpensive co-substrates such as isopropanol and glucose. This allows the microbial cells to recycle NAD(P)H internally, removing the need for costly external addition of stoichiometric amounts of expensive cofactors, thereby significantly lowering the overall production cost.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the ease of industrialization. The strain is robust, the fermentation conditions are standard (27-33°C), and the downstream processing involves simple solvent extraction. The high substrate tolerance (up to 30g/L in embodiments) and high conversion efficiency make it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-1-(3,5-bis(trifluoromethyl)phenyl)ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic route described in CN102382780B for the production of this critical Aprepitant intermediate. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities designed to meet stringent purity specifications required by global regulatory bodies. We are committed to delivering high-quality intermediates that support the uninterrupted supply of life-saving medications.
We invite you to collaborate with us to optimize your supply chain and leverage this innovative technology. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your pharmaceutical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
