Scalable Synthesis of 3,4-Pyridinediimide Derivatives via Lewis Acid-Promoted Cyclization for Commercial API Production
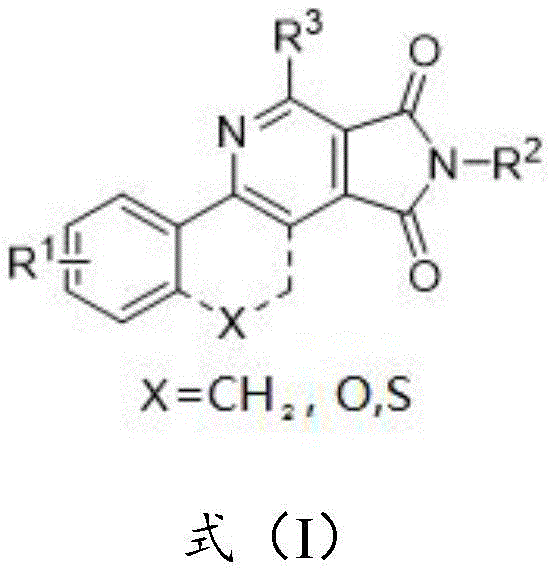
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex nitrogen-containing heterocycles, which serve as the backbone for numerous active pharmaceutical ingredients (APIs) and advanced functional materials. Patent CN113480533B introduces a groundbreaking preparation method for 3,4-pyridinediimide derivatives, addressing critical bottlenecks in current synthetic routes. This innovation leverages a Lewis acid-promoted [4+2] cyclization strategy that merges an enamide and a maleimide scaffold, resulting in a fused pyrrolo[3,4-c]pyridine-1,3-dione core. The significance of this chemistry lies in its ability to simultaneously construct two vital heterocyclic systems—pyridine and maleimide—in a single operational step. These structural motifs are ubiquitous in bioactive molecules, including potential treatments for HIV and chronic myeloid leukemia, as well as in high-performance OLED materials. By offering a pathway that avoids expensive transition metal catalysts while maintaining high atom economy, this technology represents a substantial leap forward for reliable pharmaceutical intermediate supplier networks aiming to optimize their supply chains.
![General reaction scheme showing Lewis acid-promoted [4+2] cyclization of enamide and maleimide](/insights/img/3-4-pyridinediimide-synthesis-pharma-supplier-20260305065517-09.webp)
Traditional methods for synthesizing pyridine derivatives often rely heavily on transition metal catalysis, which introduces significant challenges regarding cost, toxicity, and downstream purification. The removal of residual heavy metals like palladium or platinum to meet stringent pharmaceutical standards (often below 10 ppm) requires additional processing steps, such as scavenging or extensive recrystallization, which drastically increases manufacturing lead times and operational expenses. Furthermore, many conventional routes suffer from limited substrate scope, failing to tolerate sensitive functional groups or requiring harsh reaction conditions that compromise yield and safety. In contrast, the novel approach detailed in the patent utilizes readily available enamides and maleimides as building blocks, promoted by inexpensive Lewis acids or protonic acids. This shift eliminates the dependency on precious metals entirely, streamlining the workflow and enhancing the overall sustainability of the manufacturing process. The reaction proceeds under relatively mild thermal conditions, typically between 80°C and 140°C, which reduces energy consumption compared to high-temperature or high-pressure alternatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyridine core has been dominated by methods that, while effective, impose heavy burdens on commercial production teams. Transition metal-catalyzed couplings, for instance, are notorious for their sensitivity to air and moisture, necessitating inert atmosphere handling that complicates scale-up operations. Additionally, the cost volatility of noble metals can severely impact the financial predictability of long-term supply contracts. Beyond economic factors, the environmental footprint of these processes is considerable, generating heavy metal waste that requires specialized disposal protocols. For procurement managers, these factors translate into higher costs of goods sold (COGS) and increased supply chain risk. Moreover, traditional cyclization strategies often struggle with regioselectivity, leading to complex mixtures of isomers that are difficult to separate, thereby reducing the overall yield of the desired high-purity pharmaceutical intermediate.
The Novel Approach
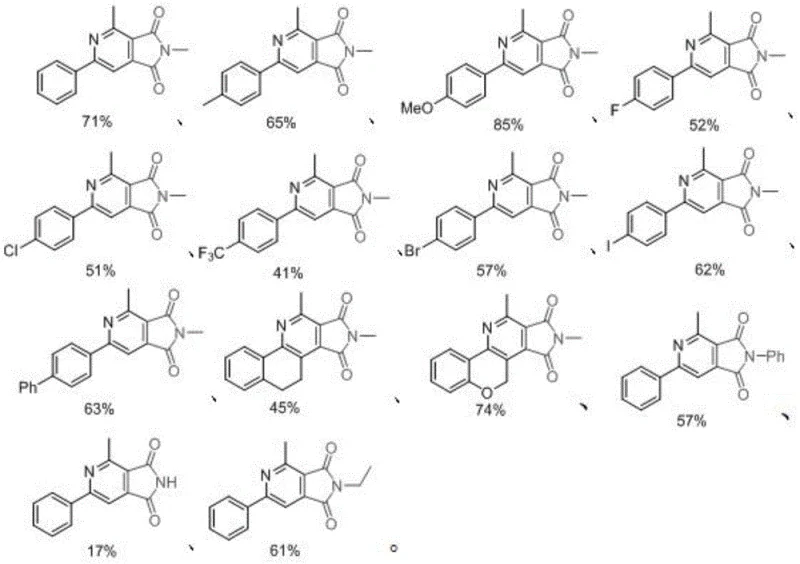
The methodology described in CN113480533B circumvents these issues by employing a direct [4+2] cycloaddition mechanism facilitated by promoters such as manganese acetate, copper salts, or iron chloride. This approach is characterized by its operational simplicity; the reagents are simply mixed in a solvent system like DMF/PhCl and heated. The absence of sensitive catalysts means the reaction is more robust and forgiving, allowing for easier handling in standard reactor vessels. From a technical perspective, this route offers exceptional functional group tolerance, accommodating electron-withdrawing groups like trifluoromethyl and halogens, as well as electron-donating groups like methoxy, without significant loss in efficiency. This versatility is crucial for medicinal chemists who need to rapidly generate diverse libraries of analogs for structure-activity relationship (SAR) studies. The result is a streamlined process that delivers the target 3,4-pyridinediimide scaffold with impressive efficiency, positioning it as a superior choice for cost reduction in API manufacturing.
Mechanistic Insights into Lewis Acid-Promoted [4+2] Cyclization
The core of this synthetic breakthrough is the concerted [4+2] cyclization between the enamide dienophile and the maleimide diene equivalent, driven by the activation of the carbonyl or imine functionalities by the promoter. The promoter A, which can range from transition metal salts like Cu(OAc)2 and Mn(OAc)2 to Lewis acids like BF3·Et2O, plays a pivotal role in lowering the activation energy of the cycloaddition. By coordinating with the electron-rich centers of the reactants, the promoter facilitates the formation of the new carbon-carbon and carbon-nitrogen bonds required to close the pyridine ring. This mechanistic pathway ensures high regioselectivity, minimizing the formation of unwanted byproducts. The reaction likely proceeds through a polarized transition state where the electron density is redistributed to favor the formation of the fused bicyclic system. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as temperature and stoichiometry, to maximize conversion rates.
Impurity control is another critical aspect where this mechanism excels. The use of specific solvent systems, particularly the binary mixture of DMF and chlorobenzene (PhCl), appears to stabilize the intermediate species and prevent side reactions such as polymerization or hydrolysis. The patent data indicates that maintaining a molar ratio of enamide to maleimide between 1.0:2.0 and 1.0:4.0 ensures that the limiting reagent is fully consumed, driving the equilibrium towards the product. Furthermore, the choice of promoter influences the purity profile; for instance, manganese acetate has shown to provide clean reaction profiles with minimal metal contamination compared to other salts. This inherent cleanliness reduces the burden on downstream purification, meaning less solvent is used for chromatography or recrystallization, which aligns with green chemistry principles and reduces waste disposal costs for the supply chain.
How to Synthesize 3,4-Pyridinediimide Derivatives Efficiently
To implement this synthesis effectively, operators should adhere to the optimized conditions outlined in the patent examples. The process begins with the precise weighing of the enamide substrate and the maleimide derivative, ensuring the molar ratios fall within the specified range to drive the reaction to completion. The selection of the promoter is flexible, but manganese acetate is highlighted as a preferred candidate due to its balance of activity and cost. The reaction mixture is then subjected to thermal energy in an oil bath, typically maintained at 120°C for a duration of 24 hours, although this can be adjusted between 12h and 36h depending on the specific reactivity of the substrates. Upon completion, standard workup procedures involving ethyl acetate extraction and drying over anhydrous sodium sulfate are sufficient to isolate the crude product, which can then be purified via column chromatography.
- Mix enamide, maleimide, and promoter A (e.g., manganese acetate) in a solvent system such as DMF/PhCl.
- Heat the reaction mixture in an oil bath at 120°C for 24 hours to facilitate the [4+2] cyclization.
- Upon completion, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond simple yield metrics. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for expensive and supply-constrained precious metal catalysts, manufacturers can secure a more stable and predictable supply chain. The promoters used, such as manganese acetate or copper chloride, are commodity chemicals available in bulk quantities at a fraction of the cost of palladium or rhodium complexes. This substitution directly contributes to substantial cost savings in the final API production without compromising quality. Additionally, the robustness of the reaction conditions means that the process is less prone to batch failures caused by minor fluctuations in temperature or atmosphere, thereby enhancing overall production reliability and on-time delivery performance.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes a significant cost driver from the bill of materials. Furthermore, the simplified purification process, necessitated by the cleaner reaction profile, reduces solvent consumption and labor hours associated with metal scavenging steps. This leads to a leaner manufacturing process with lower operational expenditures, allowing for more competitive pricing in the global market for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The starting materials, enamides and maleimides, are commercially available and can be synthesized from basic feedstocks, reducing dependency on single-source suppliers for exotic reagents. The tolerance for various functional groups means that a single platform technology can be used to produce a wide array of derivatives, increasing supply flexibility. This adaptability is crucial for mitigating risks associated with raw material shortages or geopolitical disruptions in the chemical supply chain.
- Scalability and Environmental Compliance: The reaction operates at atmospheric pressure and moderate temperatures, making it inherently safer and easier to scale from kilogram to multi-ton batches using standard glass-lined or stainless steel reactors. The reduced generation of heavy metal waste simplifies environmental compliance and waste treatment protocols, aligning with increasingly strict global regulations on industrial emissions and effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when integrating this method into their production workflows. Understanding these details is essential for R&D directors evaluating the feasibility of this route for their specific pipeline candidates.
Q: What are the key advantages of this synthesis method over traditional transition metal catalysis?
A: This method utilizes inexpensive Lewis acids or protonic acids instead of precious transition metals, significantly reducing raw material costs and simplifying the removal of metal residues, which is critical for pharmaceutical compliance.
Q: What is the substrate scope for the enamide and maleimide components?
A: The process demonstrates excellent functional group tolerance, accommodating various substituents on the phenyl ring including fluoro, chloro, bromo, iodo, trifluoromethyl, and methoxy groups, as well as different N-substituents on the maleimide.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction conditions involve standard heating (oil bath 80-140°C) and common solvents (DMF, PhCl), making it highly scalable from laboratory to multi-ton production without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Pyridinediimide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Lewis acid-promoted cyclization technology for the development of next-generation therapeutics and functional materials. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies, guaranteeing that every batch of 3,4-pyridinediimide derivatives meets the highest standards of quality and consistency.
We invite you to collaborate with us to leverage this innovative synthesis route for your upcoming projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you optimize your supply chain for the future.
