Advanced Atmospheric Pressure Synthesis of 3-Methyl-2-Pentene-4-Alkyne-1-Alcohol for Commercial Scale-Up
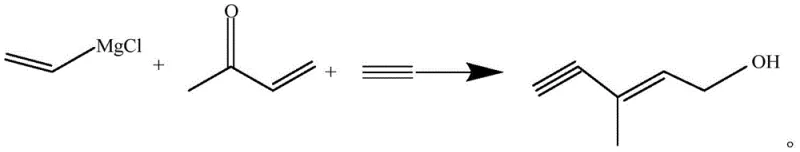
The pharmaceutical and nutraceutical industries rely heavily on the efficient production of key precursors for fat-soluble vitamins, particularly Vitamin A and astaxanthin. Patent CN109896924B introduces a transformative preparation method for 3-methyl-2-pentene-4-alkyne-1-alcohol, a critical building block in these value chains. Historically, the synthesis of such alkynols has been plagued by complex operational requirements, including the need for hazardous high-pressure reactors and cryogenic liquid ammonia systems. This new technical disclosure outlines a streamlined pathway that utilizes a vinylmagnesium chloride Grignard reagent reacting with acetylene under normal atmospheric pressure. By shifting the reaction paradigm from high-pressure forcing to precise temperature-controlled thermodynamics, the inventors have achieved a process that not only simplifies the engineering requirements but also drastically improves the chemical selectivity. The result is a robust manufacturing route that delivers high-purity intermediates with yields reaching up to 96%, addressing long-standing pain points in the supply of fine chemical intermediates for the global vitamin market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating C6 alkynol structures have historically imposed severe constraints on manufacturing flexibility and safety. Prior art methods, such as those utilizing calcium carbide or acetylene in liquid ammonia, often necessitate extremely low temperatures below -40°C and the handling of volatile, toxic solvents. Furthermore, alternative Grignard-based approaches described in patents like CN104744211A typically require the formation of bilateral acetylides under significant pressure, ranging from 0.3 to 1.5 MPa. These high-pressure conditions mandate the use of specialized autoclaves and rigorous safety protocols, which inherently increase capital expenditure and operational complexity. Additionally, the multi-step nature of these legacy processes, involving the isolation of unstable intermediates or the use of excessive reagents to drive equilibrium, frequently leads to compromised yields and difficult purification profiles. The formation of side products, particularly bilateral acetylenic species, is a common occurrence when thermal control is insufficient, resulting in final product purities that often struggle to exceed 88% without extensive downstream processing.
The Novel Approach
The methodology disclosed in CN109896924B represents a significant departure from these cumbersome legacy protocols by leveraging the unique reactivity of vinylmagnesium chloride under ambient pressure conditions. Instead of relying on mechanical pressure to dissolve acetylene into the reaction matrix, this novel approach utilizes precise thermal regulation, specifically maintaining the reaction mixture at approximately -5°C, to facilitate the selective formation of the unilateral alkynyl magnesium Grignard reagent. This subtle yet powerful shift eliminates the need for expensive pressure-rated vessels, allowing the synthesis to proceed in standard glass-lined or stainless steel reactors equipped with efficient cooling jackets. The process flow is remarkably concise: the in situ generated Grignard species reacts directly with methyl vinyl ketone, followed by a straightforward acid-catalyzed transposition to yield the target alcohol. This reduction in unit operations not only accelerates the production cycle but also minimizes the exposure of reactive intermediates to potential degradation pathways, thereby preserving the integrity of the carbon skeleton throughout the synthesis.
Mechanistic Insights into Atmospheric Pressure Grignard Alkynylation
The core mechanistic breakthrough of this patent lies in the controlled metal-halogen exchange and subsequent acetylene insertion that occurs without the driving force of high pressure. In standard Grignard chemistry, the interaction between organomagnesium species and terminal alkynes can be sluggish or non-selective without forcing conditions. However, the inventors discovered that the vinylmagnesium chloride species possesses a specific reactivity profile that allows it to exchange with acetylene efficiently at near-freezing temperatures. At -5°C, the kinetic energy of the system is sufficiently low to prevent the second addition of the Grignard reagent to the formed alkynyl species, which would otherwise lead to the formation of unreactive bilateral dialkynyl magnesium byproducts. This selectivity is paramount; comparative examples within the patent data demonstrate that raising the temperature to just 15°C results in the detection of bilateral peaks via FTIR spectroscopy, correlating with a sharp drop in isolated yield from 96% to roughly 76%. Thus, the mechanism is governed by a delicate balance of thermodynamics where low temperature favors the mono-substituted kinetic product essential for the subsequent condensation step.
Following the formation of the unilateral alkynyl magnesium intermediate, the reaction proceeds through a nucleophilic addition to methyl vinyl ketone (MVK). This step constructs the critical carbon-carbon bond that establishes the six-carbon backbone required for the Vitamin A side chain. The resulting adduct is then subjected to an acid-catalyzed rearrangement or transposition. This final transformation is crucial for establishing the correct conjugation pattern and hydroxyl positioning found in 3-methyl-2-pentene-4-alkyne-1-alcohol. The efficiency of this cascade is heavily dependent on the purity of the preceding Grignard intermediate; because the new method suppresses side reactions effectively, the crude material entering the transposition step is of significantly higher quality. This reduces the burden on the final distillation columns, allowing for the collection of the target fraction with a content of 98% or higher. The ability to control impurity profiles at the molecular level through temperature modulation rather than brute-force pressure application is a hallmark of sophisticated process chemistry design.
How to Synthesize 3-Methyl-2-Pentene-4-Alkyne-1-Alcohol Efficiently
Implementing this synthesis route requires careful attention to solvent selection and thermal management to replicate the high yields reported in the patent literature. The process begins with the generation of the vinylmagnesium chloride reagent in a solvent such as tetrahydrofuran (THF) or diethyl ether, typically initiated at elevated temperatures to ensure complete magnesium consumption before cooling. Once the Grignard reagent is formed, the system must be brought down to the critical window of -5°C prior to the introduction of acetylene gas. It is imperative that the acetylene feed is free of acetone stabilizers, as these can interfere with the Grignard species. The reaction is monitored, ideally using in-line FTIR, to confirm the absence of bilateral acetylide peaks before proceeding to the addition of methyl vinyl ketone. For a comprehensive breakdown of the specific molar ratios, addition rates, and workup procedures, please refer to the standardized protocol below.
- Preparation of vinylmagnesium chloride Grignard reagent in THF or ether solvent at elevated temperatures.
- Reaction with acetylene gas under normal pressure at controlled low temperatures (-5°C) to form unilateral alkynyl magnesium species.
- Condensation with methyl vinyl ketone followed by acid-catalyzed transposition to yield the final alkynol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from high-pressure to atmospheric pressure synthesis offers profound implications for cost structure and vendor reliability. The most immediate impact is the reduction in capital intensity; facilities no longer need to invest in or maintain specialized high-pressure autoclaves rated for 1.5 MPa service, which are subject to rigorous and costly inspection regimes. This simplification of the hardware footprint allows for greater flexibility in manufacturing site selection and significantly lowers the barrier to entry for contract manufacturing organizations. Furthermore, the operational safety profile is markedly improved by eliminating the risks associated with compressing flammable acetylene gas to high pressures, thereby reducing insurance premiums and safety compliance overheads. The streamlined nature of the process also translates to shorter batch cycles, as the time-consuming steps of pressurizing and depressurizing reactors are removed, leading to increased asset utilization and throughput capacity without the need for additional physical infrastructure.
- Cost Reduction in Manufacturing: The economic benefits of this atmospheric pressure route are driven primarily by the elimination of expensive engineering controls and the optimization of raw material efficiency. By avoiding the formation of bilateral byproducts through precise temperature control, the process maximizes the conversion of valuable starting materials like methyl vinyl ketone and acetylene into the desired product, minimizing waste disposal costs. The removal of high-pressure equipment also means lower maintenance costs and reduced downtime for safety certifications, contributing to a leaner overall cost of goods sold. Additionally, the simplified purification sequence, necessitated by the higher crude purity, reduces the consumption of solvents and energy during the distillation phase, further enhancing the margin profile for bulk manufacturers.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of legacy chemical processes that are prone to upset conditions or equipment failure. This novel method enhances reliability by utilizing robust, standard chemical engineering unit operations that are less susceptible to mechanical failure compared to high-pressure systems. The use of common solvents like THF and ether ensures that raw material sourcing remains stable and unaffected by niche supply constraints. Moreover, the higher consistency in product quality, evidenced by the tight control over isomer formation and impurity levels, reduces the likelihood of batch rejection or the need for re-processing. This predictability allows supply chain planners to maintain lower safety stocks while still meeting the demanding delivery schedules of downstream vitamin producers.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production is fraught with challenges, particularly when heat transfer and gas-liquid mass transfer are limiting factors. The atmospheric nature of this reaction simplifies scale-up significantly, as gas sparging at ambient pressure is far easier to manage in large vessels than high-pressure injection. From an environmental perspective, the process generates less hazardous waste due to the absence of heavy metal catalysts often found in alternative coupling reactions and the reduced need for extensive aqueous workups to remove pressure-transmitting fluids. The high atom economy and reduced solvent usage align with modern green chemistry principles, facilitating easier regulatory approval and permitting for new manufacturing lines in jurisdictions with strict environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this atmospheric pressure Grignard technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms traditional high-pressure alternatives. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial production of Vitamin A intermediates.
Q: What is the primary advantage of this synthesis method over traditional high-pressure routes?
A: The primary advantage is the elimination of high-pressure reactors. Traditional methods often require pressures up to 1.5 MPa to force acetylene into solution, whereas this patent demonstrates that unilateral alkynyl magnesium Grignard reagents can be formed efficiently under normal atmospheric pressure by strictly controlling the temperature around -5°C.
Q: How does this process improve product purity compared to prior art?
A: By maintaining the reaction temperature at approximately -5°C during acetylene introduction, the formation of undesirable bilateral acetylide byproducts is suppressed. Comparative data in the patent shows that running this step at higher temperatures (e.g., 15°C) leads to significant bilateral species formation, dropping purity from 98% to 76%.
Q: Is this method suitable for large-scale manufacturing of Vitamin A intermediates?
A: Yes, the method is highly scalable due to its mild conditions. The use of standard solvents like THF or diethyl ether and the avoidance of specialized high-pressure equipment significantly lowers the barrier for commercial scale-up, making it ideal for producing metric ton quantities of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methyl-2-Pentene-4-Alkyne-1-Alcohol Supplier
As the global demand for high-quality nutraceuticals and pharmaceuticals continues to rise, the need for reliable sources of key intermediates like 3-methyl-2-pentene-4-alkyne-1-alcohol has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced process technologies such as the atmospheric pressure Grignard synthesis to deliver superior value to our partners. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for Vitamin A and astaxanthin synthesis, providing you with a secure and consistent supply chain foundation.
We invite you to collaborate with us to optimize your sourcing strategy for this vital intermediate. Our technical team is prepared to provide a Customized Cost-Saving Analysis that demonstrates how switching to our optimized manufacturing route can reduce your overall procurement costs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to not just a chemical supplier, but a strategic ally committed to driving efficiency and innovation in your production lifecycle.
