Advanced Carbonylation Strategy for Commercial Scale-up of Complex Chiral Statin Intermediates
The pharmaceutical industry's relentless pursuit of efficient pathways for cardiovascular therapeutics has found a significant breakthrough in the technology disclosed in patent CN103420870A. This intellectual property introduces a streamlined, two-step synthetic methodology for producing chiral 4-cyano-3-hydroxybutyrate, a critical building block for blockbuster statin drugs such as Atorvastatin and Rosuvastatin. Unlike conventional multi-step sequences that often suffer from low overall yields and complex purification requirements, this novel approach leverages advanced carbonylation chemistry to transform readily available chiral epichlorohydrin directly into the desired ester intermediate with exceptional stereochemical fidelity. For R&D directors and process chemists, this represents a paradigm shift towards atom-economical synthesis, while supply chain leaders will recognize the strategic value of a route dependent on bulk commodity feedstocks rather than scarce chiral pool resources. The ability to maintain high enantiomeric excess throughout the transformation without rigorous resolution steps underscores the robustness of this technology for modern GMP manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 4-cyano-3-hydroxybutyrate has been plagued by significant inefficiencies that hinder cost-effective commercialization. Traditional chemical routes often rely on the kinetic resolution of racemic mixtures or the use of expensive chiral starting materials like L-malic acid, which inherently limits the maximum theoretical yield to 50% or imposes high raw material costs. Furthermore, biological methods, while selective, frequently encounter challenges related to substrate inhibition, low volumetric productivity, and the complexities of enzyme stabilization and recycling on an industrial scale. These legacy processes typically involve four or more distinct synthetic steps, each introducing potential points of yield loss, impurity generation, and operational complexity. The cumulative effect of these inefficiencies results in prolonged production cycles, elevated waste generation, and a supply chain vulnerable to the volatility of specialized reagent markets, creating substantial bottlenecks for manufacturers aiming to meet the global demand for lipid-lowering medications.
The Novel Approach
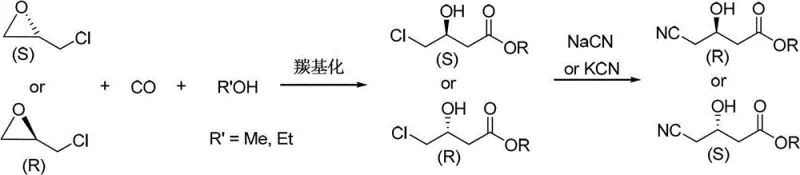
In stark contrast, the methodology outlined in CN103420870A collapses the synthetic timeline into a concise two-step sequence that maximizes efficiency and minimizes waste. By utilizing a cobalt-catalyzed carbonylation reaction, the process directly inserts a carbonyl group into the chiral epichlorohydrin backbone, effectively constructing the four-carbon ester skeleton in a single operation with complete retention of configuration. This innovative strategy bypasses the need for protective group manipulations and tedious resolution steps that characterize older technologies. The subsequent cyanation step is equally streamlined, proceeding under mild conditions to deliver the final nitrile product with high purity. This reduction in step count not only accelerates the time-to-market for new drug formulations but also drastically simplifies the manufacturing footprint, allowing for higher throughput in existing reactor trains. The result is a agile, responsive production capability that aligns perfectly with the needs of a reliable statin intermediate supplier seeking to optimize their portfolio.
Mechanistic Insights into Cobalt-Catalyzed Carbonylation
The core of this technological advancement lies in the sophisticated catalytic cycle driven by octacarbonyldicobalt (Co2(CO)8) in conjunction with specific ligands such as ZnBr2(pyridine)2 or 3-hydroxypyridine. Mechanistically, the reaction initiates with the coordination of the cobalt species to the epoxide ring of the chiral epichlorohydrin, facilitating a regioselective ring-opening and subsequent migratory insertion of carbon monoxide. This delicate balance of electronic and steric factors ensures that the nucleophilic attack by the alcohol occurs with precise stereocontrol, preserving the chiral information encoded in the starting material. The presence of the pyridine-based ligands is crucial, as they modulate the electrophilicity of the metal center and stabilize the active catalytic species against decomposition under the reaction pressures of 4 to 9 MPa. Understanding this mechanism is vital for process optimization, as it explains why the system tolerates a wide range of temperatures (30-90°C) without compromising the optical integrity of the product, a common failure point in less robust catalytic systems.
Beyond the primary transformation, the impurity control mechanism inherent in this route offers profound advantages for quality assurance. The high selectivity of the carbonylation step minimizes the formation of regioisomers and oligomeric byproducts that are notoriously difficult to separate in traditional syntheses. Because the reaction proceeds with such high fidelity, the crude product profile is significantly cleaner, reducing the burden on downstream purification units like distillation columns or crystallization tanks. This intrinsic purity is maintained through the cyanation step, where the use of aqueous alcohol solvents facilitates the smooth displacement of the chloride ion without inducing elimination side reactions that could generate unsaturated impurities. For regulatory affairs teams, this consistent impurity profile simplifies the validation process and ensures that the final high-purity chiral 4-cyano-3-hydroxybutyrate meets the stringent specifications required for API synthesis, thereby mitigating the risk of batch failures and ensuring supply continuity.
How to Synthesize Chiral 4-Cyano-3-Hydroxybutyrate Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters to fully realize its potential for cost reduction in pharmaceutical intermediate manufacturing. The process begins with the charging of chiral epichlorohydrin, the selected alcohol (methanol or ethanol), and the catalyst system into a pressure-rated autoclave, followed by pressurization with carbon monoxide. Detailed operational protocols regarding temperature ramping, pressure maintenance, and workup procedures are critical to achieving the reported yields of up to 97% for the intermediate and high conversion rates for the final nitrile. While the general chemistry is robust, scaling this reaction requires precise engineering controls to manage the exothermic nature of carbonylation and the safe handling of carbon monoxide gas. The following guide outlines the standardized steps derived from the patent examples to ensure reproducibility and safety during technology transfer.
- Perform carbonylation of chiral epichlorohydrin with CO and alcohol using a Co2(CO)8 catalyst system under 4-9 MPa pressure at 30-90°C to obtain chiral 4-chloro-3-hydroxybutyrate with retained configuration.
- Conduct cyanation of the resulting chloro-ester using sodium or potassium cyanide in an aqueous alcohol solution under heating to yield the final chiral 4-cyano-3-hydroxybutyrate.
- Purify the final product through extraction, drying, and vacuum distillation to achieve high purity and enantiomeric excess suitable for pharmaceutical applications.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented process offers transformative benefits that directly address the pain points of procurement managers and supply chain heads. The fundamental shift away from complex, multi-step syntheses towards a direct carbonylation route fundamentally alters the cost structure of the molecule. By eliminating the need for expensive chiral resolving agents, precious metal catalysts, or biologically derived enzymes, the variable cost of goods sold is substantially reduced. Furthermore, the reliance on carbon monoxide and simple alcohols as C1 and alkyl sources leverages the stability and abundance of the petrochemical supply chain, insulating the production process from the volatility associated with specialty agricultural or fermentation-based feedstocks. This stability translates into more predictable pricing models and stronger negotiation leverage when securing long-term supply agreements for key statin precursors.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the drastic simplification of the process flow. By condensing the synthesis into just two steps, manufacturers eliminate the capital expenditure and operational overhead associated with multiple isolation and purification stages. The removal of transition metal catalysts that require expensive scavenging resins further lowers the cost per kilogram. Additionally, the high atom economy of the carbonylation reaction means that a greater proportion of the raw material mass ends up in the final product, minimizing waste disposal costs and maximizing raw material utilization efficiency. These factors combine to create a lean manufacturing process that delivers substantial cost savings without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of universally available bulk chemicals. Unlike niche intermediates that may have single-source suppliers or long lead times, the inputs for this process—epichlorohydrin, alcohols, and carbon monoxide—are produced globally at massive scales. This ubiquity ensures that production schedules are not held hostage by the availability of a single specialized reagent. Moreover, the robustness of the reaction conditions allows for flexible manufacturing across different geographic locations, enabling a diversified supply base that can mitigate regional disruptions. This reliability is crucial for reducing lead time for high-purity statin precursors, ensuring that downstream API production lines remain operational and responsive to market demand.
- Scalability and Environmental Compliance: The environmental profile of this synthesis aligns with modern green chemistry principles, facilitating easier regulatory approval and community acceptance. The process generates significantly less solvent waste and hazardous byproducts compared to traditional routes involving heavy metals or stoichiometric oxidants. The mild reaction temperatures and pressures are well within the capabilities of standard stainless-steel reactors, meaning that commercial scale-up of complex chiral building blocks can be achieved without requiring exotic, high-cost infrastructure. This ease of scalability allows manufacturers to rapidly ramp up production volumes from pilot batches to multi-ton annual capacities, ensuring that the supply chain can flexibly adapt to the growing global demand for cardiovascular therapies.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis method, we have compiled answers to common inquiries regarding its implementation and performance. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering a transparent view of the technology's capabilities. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this route for their own manufacturing operations or supply partnerships. The following questions address critical aspects of stereochemistry, cost drivers, and industrial viability.
Q: How does this carbonylation method improve optical purity compared to traditional routes?
A: The patented process utilizes chiral epichlorohydrin as a starting material and employs a specific cobalt catalyst system that ensures configuration retention during the carbonylation step, preventing racemization and maintaining ee values above 99.9%.
Q: What are the primary cost drivers reduced in this manufacturing process?
A: The process eliminates the need for expensive chiral pool materials like L-malic acid or complex enzymatic resolutions, relying instead on bulk commodity chemicals such as carbon monoxide and simple alcohols, which significantly lowers raw material costs.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild (30-90°C) and the equipment requirements are standard for fine chemical production, making it highly suitable for commercial scale-up from pilot plants to multi-ton annual capacity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 4-Cyano-3-Hydroxybutyrate Supplier
As the global demand for cardiovascular medications continues to rise, the need for efficient, scalable, and cost-effective synthesis routes for key intermediates has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced technologies like the carbonylation process described in CN103420870A to deliver superior value to our partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of high-purity chiral 4-cyano-3-hydroxybutyrate meets the exacting standards required for pharmaceutical applications, providing you with a secure and reliable foundation for your drug development pipeline.
We invite you to explore how our optimized manufacturing capabilities can enhance your supply chain efficiency and reduce your overall production costs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to innovation and quality can support your strategic goals and drive success in the competitive landscape of statin manufacturing.
