Revolutionizing Apremilast Intermediate Production with Advanced Asymmetric Catalysis and Precision Purification
The pharmaceutical industry constantly demands higher standards for intermediate purity, particularly for complex small molecule inhibitors like Apremilast. Patent CN112645854B introduces a groundbreaking refinement method for the key chiral intermediate, designated as Formula (I), which serves as a critical building block in the synthesis of this PDE4 inhibitor. The traditional challenges in producing this intermediate often revolve around the difficulty of removing structurally similar impurities and enantiomers that persist even after standard synthetic procedures. This new technology addresses these pain points by combining a highly selective asymmetric catalytic hydrogenation step with a novel recrystallization protocol using specific ketone-nitrile solvent systems. By implementing this refined approach, manufacturers can achieve product purities exceeding 99.5% while maintaining robust yields, effectively solving the bottleneck of residual process impurities that typically hover around 2% in conventional methods. 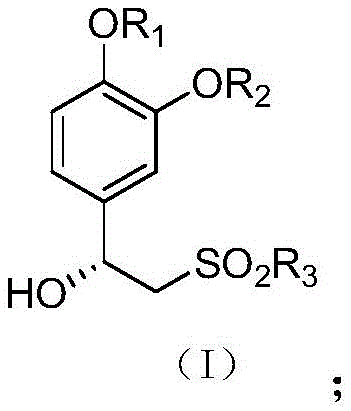
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the chiral amine precursor for Apremilast has relied on routes that involve the separate preparation of chiral amines and anhydrides, followed by coupling reactions. A significant drawback of these legacy pathways is the reliance on chiral resolution or the use of expensive chiral auxiliaries, which inherently limits the overall yield and increases the cost of goods. Furthermore, even when asymmetric hydrogenation is employed to generate the chiral alcohol precursor, the resulting crude product often contains approximately 2% of unreacted starting material or enantiomeric impurities that are chemically very similar to the target molecule. Conventional purification techniques struggle to differentiate between these closely related species, leading to a final purity that rarely exceeds 93% without extensive and wasteful chromatographic separations. This inability to drive single impurity levels below the critical 0.1% threshold poses a severe risk for regulatory compliance in API manufacturing. 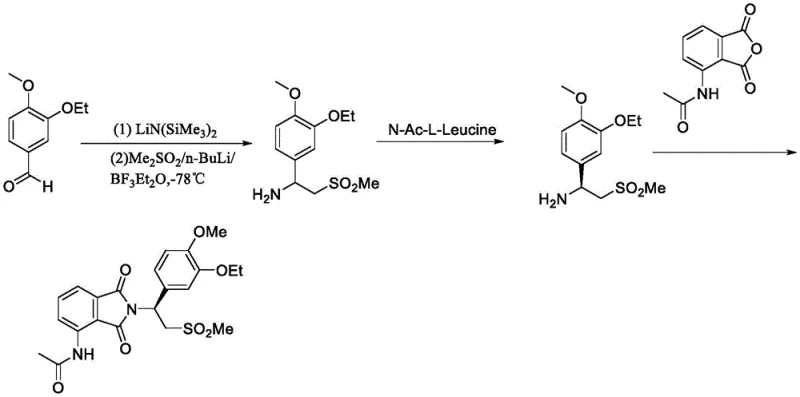
The Novel Approach
The innovative strategy outlined in the patent data fundamentally shifts the paradigm by integrating a specialized solid-phase chiral catalyst with a precision solvent engineering technique for purification. Instead of relying on brute-force chromatography, the process utilizes a polyethylene glycol-modified ruthenium catalyst to establish the chiral center with high fidelity during the hydrogenation of the ketone precursor. Following the reaction, the crude intermediate is subjected to a controlled recrystallization using a binary solvent system comprising methyl isobutyl ketone and acetonitrile, or butanone and acetonitrile. This specific solvent combination exploits subtle solubility differences to exclude impurities from the crystal lattice, consistently delivering a refined product with purity levels above 99.5% and single impurities controlled below 0.07%. 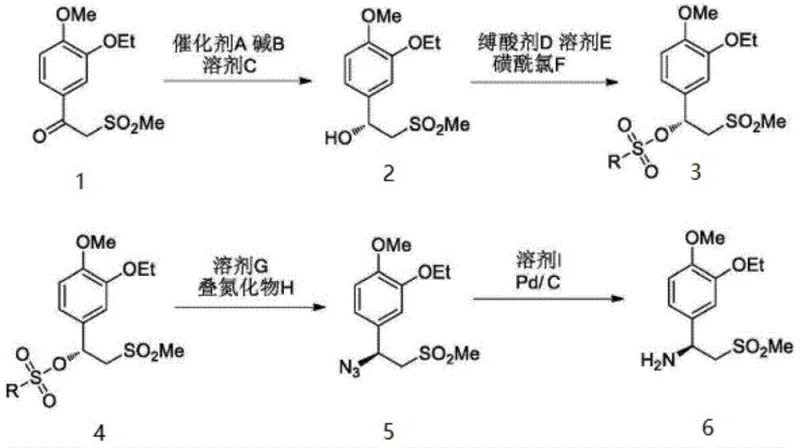
Mechanistic Insights into PEG-Modified Ruthenium Catalysis and Solvent Engineering
The core of this technological advancement lies in the unique structure of the catalyst employed, specifically the polymer-supported ruthenium complex depicted in Formula (C). This catalyst features a polyethylene glycol (PEG) chain with a degree of polymerization (n) ranging from 12 to 65, which serves a dual purpose in the reaction mechanism. Firstly, the long PEG chain acts as a phase-transfer mediator that enhances the interaction between the hydrophobic substrate and the catalytic center, thereby boosting the asymmetric conversion rate. Secondly, and perhaps more critically for pharmaceutical applications, the PEG moiety facilitates the immobilization of the heavy metal complex, allowing it to be easily separated from the reaction mixture via simple filtration post-reaction. This design drastically minimizes the leaching of ruthenium ions into the product stream, addressing one of the most stringent requirements for API intermediate specifications regarding heavy metal residues. 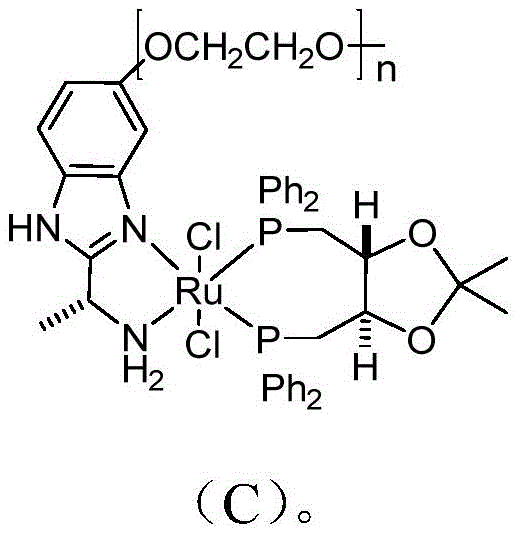
Complementing the catalytic efficiency is the sophisticated solvent selection strategy used in the purification stage. The patent identifies that impurities affecting the final purity are primarily the starting ketone compound and the enantiomer of the target alcohol, both of which possess physicochemical properties nearly identical to the desired product. By employing a mixed solvent system of methyl isobutyl ketone and acetonitrile in specific volume ratios ranging from 1:1 to 10:1, the process creates a thermodynamic environment where the target intermediate has optimal solubility at elevated temperatures (50-80°C) but poor solubility at low temperatures (0-10°C). Conversely, the impurities remain soluble in the mother liquor even upon cooling, preventing their co-crystallization. This mechanism ensures that the crystal growth is highly selective, effectively scrubbing the product of trace contaminants without the need for complex equipment or specialized operational skills.
How to Synthesize High-Purity Apremilast Intermediate Efficiently
The synthesis and purification workflow described in the patent offers a streamlined pathway for producing the critical chiral intermediate required for Apremilast. The process begins with the asymmetric hydrogenation of the ketone precursor using the specialized PEG-Ru catalyst under hydrogen pressure, followed by a straightforward workup to isolate the crude alcohol. The subsequent purification step is the key differentiator, involving dissolution in the optimized solvent blend and controlled cooling to induce high-purity crystallization. This methodology is designed to be robust and scalable, minimizing the number of unit operations while maximizing the removal of critical impurities. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide provided below. 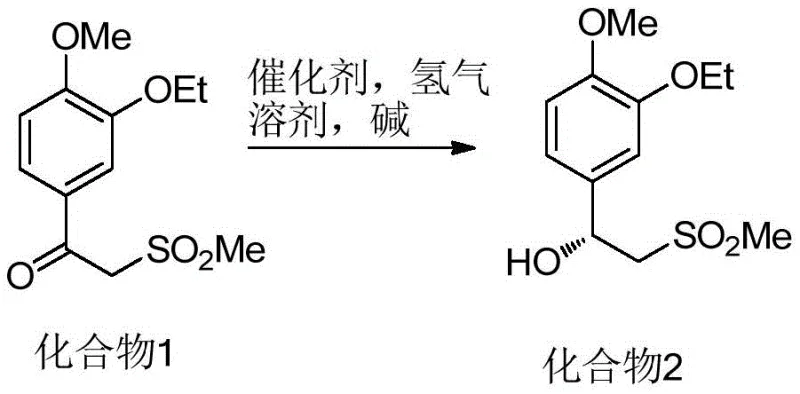
- Provide the crude intermediate of formula (I) obtained from asymmetric hydrogenation, ensuring initial impurity levels are within manageable ranges.
- Dissolve the crude intermediate in a refining solvent mixture, such as methyl isobutyl ketone and acetonitrile, at a temperature between 50°C and 80°C.
- Cool the solution gradually to 0°C to 10°C to induce crystallization, then perform solid-liquid separation and dry the crystals to obtain the refined product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented purification method translates into tangible strategic benefits beyond mere technical specifications. The elimination of complex chiral resolution steps and the reduction in purification cycles significantly simplify the manufacturing workflow, which directly correlates to reduced operational expenditures and shorter production lead times. By utilizing a catalyst that can be recovered via simple filtration due to its solid-phase nature, the process mitigates the loss of expensive precious metals and reduces the environmental burden associated with heavy metal waste disposal. Furthermore, the reliance on commodity solvents like methyl isobutyl ketone and acetonitrile ensures that raw material sourcing remains stable and cost-effective, avoiding the supply chain volatility often associated with exotic or highly specialized reagents.
- Cost Reduction in Manufacturing: The integration of a recoverable solid-phase catalyst eliminates the need for expensive downstream metal scavenging processes, which are typically required to meet strict pharmaceutical limits. Additionally, the high efficiency of the recrystallization step reduces the need for multiple purification passes, thereby lowering solvent consumption and energy costs associated with distillation and drying. The overall simplification of the synthetic route removes the necessity for costly chiral resolving agents, resulting in a leaner and more economically viable production model for high-volume API intermediates.
- Enhanced Supply Chain Reliability: By relying on widely available industrial solvents and a robust catalytic system, the manufacturing process becomes less susceptible to disruptions caused by the scarcity of niche chemicals. The ability to consistently achieve high purity levels reduces the risk of batch failures and reprocessing, ensuring a steady and reliable flow of materials to downstream API synthesis units. This stability is crucial for maintaining continuous production schedules and meeting the rigorous delivery commitments expected by global pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reactor equipment and straightforward crystallization techniques that do not require specialized infrastructure. The reduction in heavy metal residues and the efficient recovery of the catalyst contribute to a greener manufacturing profile, aligning with increasingly stringent environmental regulations and corporate sustainability goals. This eco-friendly approach not only minimizes waste treatment costs but also enhances the marketability of the final drug product as being produced through responsible chemical practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced purification technology. Understanding these details is essential for R&D teams evaluating the feasibility of adopting this route for commercial production. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to ensure accuracy and relevance.
Q: How does this purification method control single impurities below 0.1%?
A: The method utilizes a specific solvent system, such as methyl isobutyl ketone mixed with acetonitrile, which leverages the principle of like dissolves like to selectively crystallize the target intermediate while keeping structurally similar impurities and enantiomers in the mother liquor.
Q: What are the advantages of the PEG-modified catalyst used in the synthesis?
A: The polyethylene glycol-modified ruthenium catalyst allows for solid-phase immobilization, facilitating easy recovery via simple filtration and significantly reducing the risk of heavy metal residues in the final pharmaceutical product.
Q: Can this process be scaled for commercial API manufacturing?
A: Yes, the process avoids complex chiral resolution steps and uses common industrial solvents like methyl isobutyl ketone and acetonitrile, making it highly suitable for large-scale commercial production with consistent quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Apremilast Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful development of life-saving medications like Apremilast. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of intermediate meets the exacting standards required for global regulatory submissions. Our expertise in handling complex chiral syntheses and advanced purification techniques positions us as a strategic partner capable of delivering consistent value.
We invite you to engage with our technical procurement team to discuss how this patented purification method can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits specific to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the quality and cost-efficiency of your pharmaceutical manufacturing operations.
