Scalable Synthesis of 1-Phenyl Vinyl Borate for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for critical building blocks, and the recent patent CN112574244B offers a transformative solution for the production of 1-phenyl vinyl borate. This compound serves as a pivotal intermediate in the synthesis of complex bioactive molecules and functional organic materials, yet its historical manufacturing processes have been plagued by scalability issues and high costs. The disclosed invention introduces a refined palladium-catalyzed cross-coupling strategy that fundamentally alters the economic and operational landscape of producing this valuable organoboron species. By shifting from expensive reagents and laborious purification techniques to a streamlined, cost-effective protocol, this technology addresses the critical bottlenecks that have long hindered the commercial availability of high-purity vinyl boronates. For R&D directors and procurement specialists alike, understanding the nuances of this patented method is essential for securing a reliable supply chain and optimizing the cost structure of downstream API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-phenyl vinyl borate has relied on routes that are inherently difficult to scale and purify, creating significant friction in the supply chain. One prominent prior art method involves the borylation of phenylacetylene using cuprous chloride catalysts in DMF, a process that notoriously generates a mixture of regioisomers which are exceptionally difficult to separate. 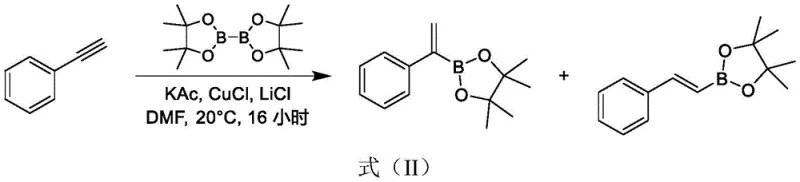 As illustrated in the reaction scheme, the lack of selectivity leads to a product mixture containing different position-substituted boronic acid esters, necessitating complex and yield-loss-inducing column chromatography for purification. Another reported approach utilizes 1-phenylvinyl triflate as a starting material, which presents its own set of challenges regarding raw material availability and cost.
As illustrated in the reaction scheme, the lack of selectivity leads to a product mixture containing different position-substituted boronic acid esters, necessitating complex and yield-loss-inducing column chromatography for purification. Another reported approach utilizes 1-phenylvinyl triflate as a starting material, which presents its own set of challenges regarding raw material availability and cost. 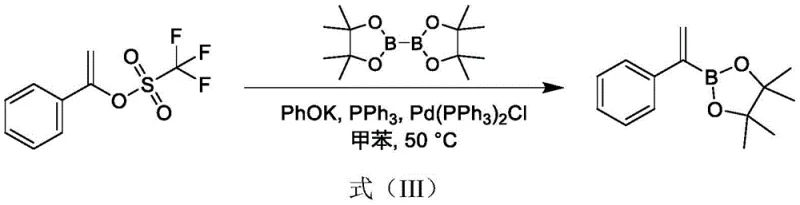 This pathway not only relies on a precursor that is not easily obtained commercially but also employs potassium phenoxide, a reagent that is significantly more expensive than its sodium counterparts. Furthermore, the reliance on column chromatography for final purification in these traditional methods renders them economically unviable for pilot-scale or commercial manufacturing, as the operational expenditure and time required for such separation techniques are prohibitive in an industrial setting.
This pathway not only relies on a precursor that is not easily obtained commercially but also employs potassium phenoxide, a reagent that is significantly more expensive than its sodium counterparts. Furthermore, the reliance on column chromatography for final purification in these traditional methods renders them economically unviable for pilot-scale or commercial manufacturing, as the operational expenditure and time required for such separation techniques are prohibitive in an industrial setting.
The Novel Approach
The innovative method disclosed in patent CN112574244B circumvents these historical limitations by employing a highly selective palladium-catalyzed coupling between (1-bromovinyl)benzene and a diborate ester. 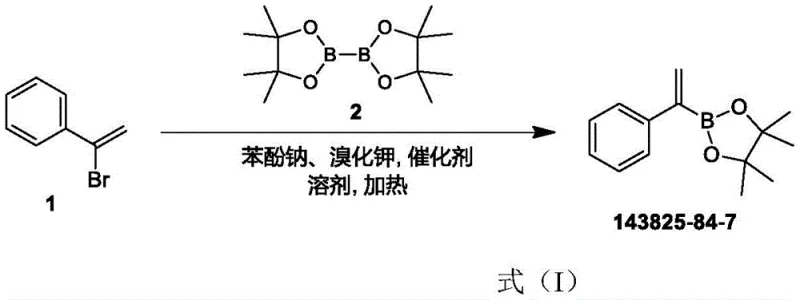 This novel approach strategically replaces the costly potassium phenoxide with sodium phenolate and introduces potassium bromide as a synergistic reagent, drastically reducing the overall reagent cost while maintaining high catalytic activity. The reaction is conducted in toluene, a solvent that is not only effective for the transformation but also facilitates easier workup and solvent recovery compared to polar aprotic solvents like DMF. Crucially, the process eliminates the need for column chromatography entirely; instead, the crude product is purified through simple operations such as pulping and low-temperature recrystallization. This shift from chromatographic separation to crystallization-based purification is a game-changer for commercial scale-up, as it allows for the processing of much larger batches with significantly reduced waste generation and operational complexity, ensuring a consistent supply of high-purity material.
This novel approach strategically replaces the costly potassium phenoxide with sodium phenolate and introduces potassium bromide as a synergistic reagent, drastically reducing the overall reagent cost while maintaining high catalytic activity. The reaction is conducted in toluene, a solvent that is not only effective for the transformation but also facilitates easier workup and solvent recovery compared to polar aprotic solvents like DMF. Crucially, the process eliminates the need for column chromatography entirely; instead, the crude product is purified through simple operations such as pulping and low-temperature recrystallization. This shift from chromatographic separation to crystallization-based purification is a game-changer for commercial scale-up, as it allows for the processing of much larger batches with significantly reduced waste generation and operational complexity, ensuring a consistent supply of high-purity material.
Mechanistic Insights into Palladium-Catalyzed Vinyl Borylation
The core of this synthetic breakthrough lies in the efficient operation of the palladium catalytic cycle, which facilitates the formation of the carbon-boron bond with exceptional regioselectivity. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-bromine bond of the (1-bromovinyl)benzene substrate, forming a stable vinyl-palladium(II) intermediate. This step is critical as the vinyl bromide substrate is more reactive and selective compared to the triflate or alkyne precursors used in older methods, minimizing the formation of side products. Subsequently, the activated diboron species, assisted by the phenolate base, undergoes transmetallation with the vinyl-palladium complex. The presence of potassium bromide in the reaction mixture is believed to play a vital role in stabilizing the catalytic species and enhancing the solubility of the inorganic salts in the organic phase, thereby accelerating the turnover frequency of the catalyst. Finally, reductive elimination releases the desired 1-phenyl vinyl borate product and regenerates the active palladium(0) catalyst, closing the loop for continuous turnover.
From an impurity control perspective, this mechanism offers superior advantages over the copper-catalyzed alkyne borylation routes. In the traditional alkyne pathway, the lack of steric and electronic differentiation often leads to the formation of both E and Z isomers, as well as over-borylated byproducts, which are structurally similar and notoriously difficult to separate. In contrast, the stereospecific nature of the oxidative addition into the vinyl bromide bond preserves the geometry of the double bond, leading to a single, well-defined isomeric product. The use of sodium phenolate as the base further contributes to a cleaner reaction profile by avoiding the harsher conditions or side reactions associated with stronger or more nucleophilic bases. This high level of selectivity means that the crude reaction mixture contains significantly fewer impurities, simplifying the downstream purification process to mere recrystallization and ensuring that the final product meets the stringent purity specifications required for pharmaceutical applications without the need for resource-intensive chromatographic polishing.
How to Synthesize 1-Phenyl Vinyl Borate Efficiently
The practical implementation of this synthesis involves a straightforward sequence of mixing, heating, and crystallization that is designed for ease of operation in a pilot plant environment. The process begins by combining the vinyl bromide starting material and the diboron ester in toluene, followed by the addition of the sodium phenolate, potassium bromide, and the palladium catalyst system under an inert atmosphere. The reaction mixture is then heated to a moderate temperature range, allowing the cross-coupling to proceed to completion over a controlled period. Once the reaction is finished, the workup is remarkably simple, involving filtration to remove insoluble salts, followed by extraction and concentration. The detailed standardized synthesis steps, including specific molar ratios and temperature profiles optimized for maximum yield, are outlined below.
- Dissolve (1-bromovinyl)benzene and bis(pinacolato)diboron in toluene, then add sodium phenolate, potassium bromide, and the palladium catalyst system.
- Heat the reaction mixture to 40-100°C under inert gas for 12-24 hours to complete the cross-coupling reaction.
- Filter the reaction mixture, concentrate the filtrate, extract with ethyl acetate, wash with brine, dry, and purify the crude product via low-temperature recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible strategic benefits that extend far beyond simple chemistry. The primary advantage lies in the substantial cost reduction achieved through the substitution of raw materials; replacing expensive potassium phenoxide with economical sodium phenolate directly lowers the bill of materials without sacrificing quality. Furthermore, the elimination of column chromatography represents a massive reduction in processing time and solvent consumption, which are major cost drivers in fine chemical manufacturing. This streamlined workflow not only reduces the direct manufacturing costs but also minimizes the environmental footprint associated with solvent waste disposal, aligning with modern sustainability goals. By simplifying the purification train, the process becomes inherently more robust and less prone to batch-to-batch variability, ensuring a more predictable and reliable supply of this critical intermediate.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the strategic selection of reagents and the simplification of unit operations. By utilizing sodium phenolate instead of potassium phenoxide, the raw material costs are significantly lowered, as sodium salts are generally more abundant and less expensive to produce than their potassium analogues. Additionally, the removal of the column chromatography step eliminates the need for large quantities of silica gel and elution solvents, which are costly consumables in traditional organic synthesis. This reduction in consumable usage, combined with the ability to recover and recycle the toluene solvent efficiently, results in a leaner manufacturing process with a lower overall cost of goods sold, making the final product more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available and commodity-grade starting materials. Unlike the specialized triflate precursors or specific copper catalysts required in older methods, (1-bromovinyl)benzene and bis(pinacolato)diboron are commercially accessible from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate standard industrial heating and stirring equipment, further ensures that production can be maintained consistently even during periods of high demand. This reliability is crucial for downstream pharmaceutical manufacturers who require uninterrupted access to high-quality intermediates to maintain their own production schedules and meet regulatory deadlines.
- Scalability and Environmental Compliance: The transition from chromatographic purification to crystallization is the key enabler for successful commercial scale-up. Crystallization is a unit operation that scales linearly and predictably from the gram scale to the ton scale, whereas chromatography often faces severe limitations in throughput and efficiency at large volumes. This scalability ensures that the process can meet the growing demand for 1-phenyl vinyl borate as it moves through clinical trials into commercial drug production. Moreover, the reduced solvent usage and waste generation associated with this greener purification method facilitate easier compliance with increasingly stringent environmental regulations, reducing the administrative and operational burden on the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 1-phenyl vinyl borate, based on the detailed specifications provided in the patent literature. These insights are curated to help technical decision-makers evaluate the feasibility of integrating this intermediate into their specific development pipelines. Understanding the nuances of the purification process and the stability of the final product is essential for ensuring successful downstream coupling reactions and maintaining the integrity of the final active pharmaceutical ingredient.
Q: Why is sodium phenolate preferred over potassium phenoxide in this synthesis?
A: Sodium phenolate is significantly more economical and easier to source than potassium phenoxide, directly reducing raw material costs without compromising reaction efficiency or selectivity.
Q: How does this method improve purification compared to prior art?
A: Unlike previous methods requiring complex column chromatography, this process utilizes simple filtration and low-temperature recrystallization, making it highly suitable for large-scale pilot production.
Q: What are the key advantages for supply chain stability?
A: The use of readily available starting materials like (1-bromovinyl)benzene and common solvents like toluene ensures consistent supply continuity and reduces dependency on specialized reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Phenyl Vinyl Borate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the acceleration of drug discovery and development. Our team of expert chemists has extensively analyzed the patented route for 1-phenyl vinyl borate and possesses the technical capability to implement this advanced synthesis method with precision and efficiency. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the early stages of research or full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 1-phenyl vinyl borate we deliver meets the highest standards of quality and consistency required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cost-effective and scalable technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating exactly how this optimized route can improve your bottom line. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your supply chain.
