Optimizing Tenibulin Production: A Strategic Shift to Negishi Coupling for Commercial Scale-Up
Optimizing Tenibulin Production: A Strategic Shift to Negishi Coupling for Commercial Scale-Up
The pharmaceutical landscape for dermatological treatments has been significantly advanced by the development of Tenibulin (Tirbanibulin), a potent microtubule inhibitor approved for treating actinic keratosis. However, the commercial viability of such complex molecules often hinges on the efficiency of their synthetic routes. Patent CN113999166B introduces a transformative methodology that replaces traditional cross-coupling strategies with a robust Negishi coupling protocol. This innovation addresses critical bottlenecks in prior art, specifically targeting the challenges associated with pyridine substrate reactivity and harsh reaction conditions. By leveraging the superior nucleophilicity of organozinc reagents, this method achieves high purity and yield under mild thermal conditions, marking a pivotal shift towards more sustainable and economically viable pharmaceutical intermediate manufacturing.
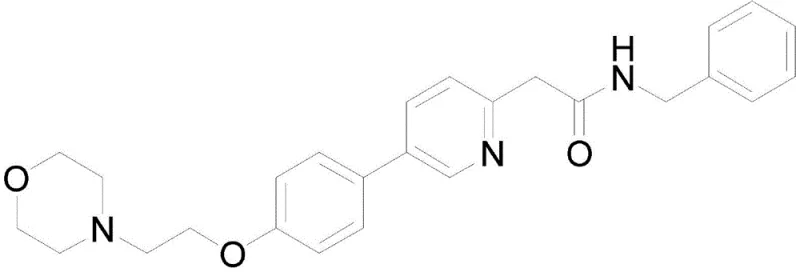
The structural complexity of Tenibulin, characterized by a substituted pyridine ring linked to a phenoxy-morpholine system, presents unique synthetic challenges. Traditional approaches often struggle with the electron-deficient nature of the pyridine ring, necessitating aggressive reaction parameters that compromise safety and scalability. The disclosed invention circumvents these issues by strategically employing a palladium-catalyzed Negishi coupling as the key bond-forming step. This approach not only streamlines the synthesis but also aligns with modern green chemistry principles by reducing energy consumption and hazardous waste generation, positioning it as an ideal candidate for commercial scale-up of complex APIs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tenibulin has relied heavily on Suzuki-Miyaura coupling or multi-step sequences involving hazardous reagents, both of which present significant drawbacks for industrial application. One prominent prior art route, disclosed in WO2006071960, utilizes a Suzuki coupling between a boronic ester and a bromopyridine derivative. While Suzuki coupling is a staple in medicinal chemistry, it often suffers from low yields when applied to electron-deficient heteroaromatic systems like pyridines. Furthermore, this specific route necessitates microwave irradiation at elevated temperatures of 150°C to drive the reaction to completion, a condition that is energetically inefficient and difficult to replicate safely on a multi-kilogram or tonne scale. Additionally, the reliance on pinacol boronic esters as starting materials introduces unnecessary cost burdens due to the high price of these specialized reagents.
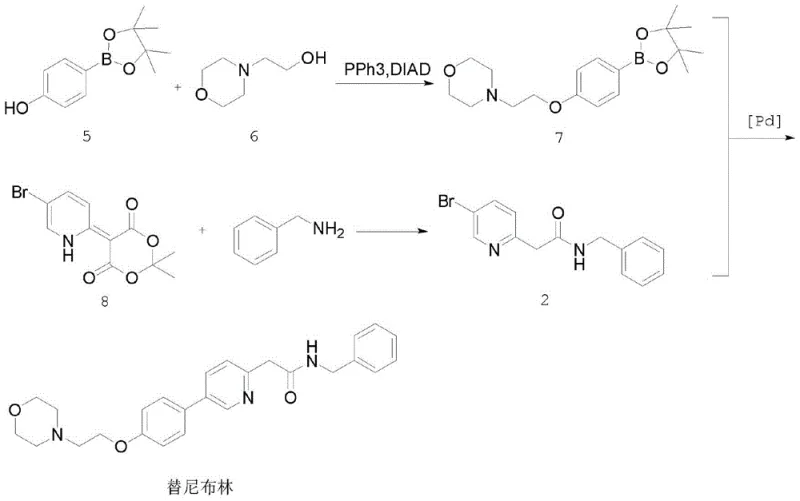
Another existing pathway, described in WO2008082637, attempts to bypass the boronic ester issue but introduces even more severe operational hazards. This route involves the use of n-butyllithium (n-BuLi) to generate a lithiated intermediate at cryogenic temperatures of -60°C. The requirement for such extreme cold demands specialized refrigeration equipment and rigorous exclusion of moisture and oxygen, drastically increasing capital expenditure and operational complexity. Moreover, this lengthy sequence involves multiple protection and deprotection steps, along with a low-yielding nucleophilic aromatic substitution of fluorine by acetonitrile. The cumulative effect of these inefficiencies results in poor overall atom economy and a supply chain vulnerable to disruptions caused by the handling of pyrophoric reagents.
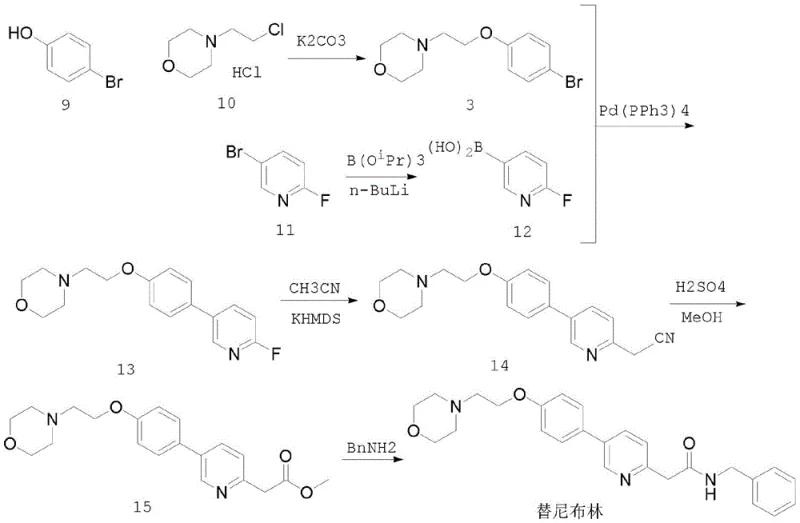
The Novel Approach
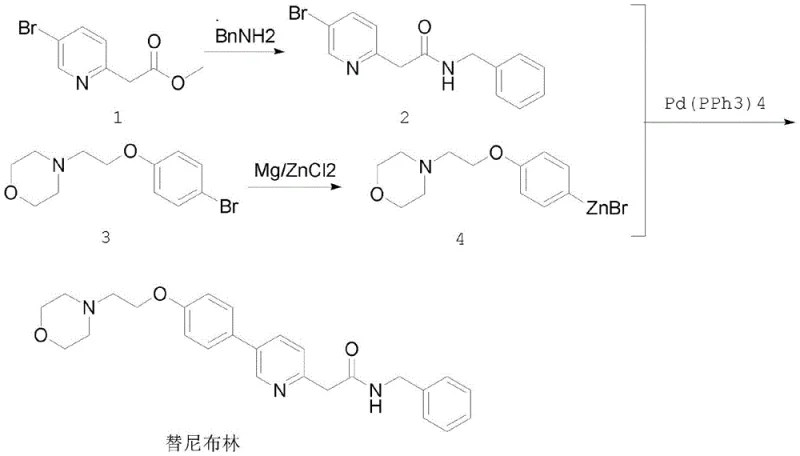
In stark contrast to these legacy methods, the novel approach detailed in CN113999166B leverages the distinct advantages of Negishi coupling to forge the critical carbon-carbon bond between the pyridine and phenyl rings. By converting the bromophenoxy-morpholine precursor into an organozinc reagent via a Grignard intermediate, the synthesis capitalizes on the higher reactivity of zinc species compared to their boron counterparts. This increased nucleophilicity allows the coupling with the bromopyridine acetamide to proceed efficiently at mild temperatures ranging from 0°C to 40°C, completely eliminating the need for cryogenic cooling or high-energy microwave heating. The result is a streamlined, three-step process that delivers the target molecule with exceptional purity and yield, exemplifying a breakthrough in cost reduction in API manufacturing.
Mechanistic Insights into Pd-Catalyzed Negishi Coupling
The success of this synthetic strategy lies in the fundamental mechanistic advantages of the Negishi coupling cycle, particularly when dealing with electron-deficient heterocycles. The catalytic cycle initiates with the oxidative addition of the palladium(0) catalyst, typically tetrakis(triphenylphosphine)palladium, into the carbon-bromine bond of the pyridine acetamide intermediate. Unlike Suzuki coupling, where the transmetallation step with boronic acids can be sluggish and often requires strong bases that might degrade sensitive functional groups, the transmetallation with organozinc reagents is rapid and irreversible. The zinc-carbon bond is highly polarized, making the carbon atom significantly more nucleophilic, which facilitates a smooth transfer to the palladium center even on the electron-poor pyridine ring. This kinetic advantage ensures that the desired cross-coupling product is formed preferentially over potential side reactions, such as homocoupling of the zinc reagent.
Furthermore, the choice of generating the zinc reagent in situ via a Grignard precursor offers precise control over impurity profiles. The initial formation of the Grignard reagent from the bromophenoxy-morpholine and magnesium powder is a well-understood exothermic process that can be managed effectively in standard reactor vessels. Subsequent transmetallation with anhydrous zinc chloride converts the highly reactive Grignard species into the more stable and selective organozinc halide. This stability is crucial for minimizing the formation of des-halogenated byproducts or biaryl impurities that often plague cross-coupling reactions. By maintaining the reaction temperature below 20°C during the coupling phase, the process further suppresses thermal degradation pathways, ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications without the need for extensive chromatographic purification.
How to Synthesize Tenibulin Efficiently
The implementation of this novel route requires careful attention to the preparation of the organometallic species and the stoichiometry of the coupling partners. The process begins with the amination of methyl 2-(5-bromopyridin-2-yl)acetate with benzylamine in a high-boiling solvent like xylene, establishing the pyridine core. Following isolation, the focus shifts to the generation of the zinc nucleophile, where the ratio of magnesium to zinc chloride is critical for maximizing conversion. Finally, the coupling step demands an inert atmosphere to protect the palladium catalyst and the organozinc species from oxidation. For a comprehensive guide on the specific molar ratios, solvent choices, and workup procedures validated by experimental data, please refer to the standardized protocol below.
- Perform amination of methyl 2-(5-bromopyridin-2-yl)acetate with benzylamine in xylene under reflux to obtain the pyridine acetamide intermediate.
- Generate the organozinc reagent by reacting N-[2-(4-bromophenoxy)ethyl]morpholine with magnesium powder to form a Grignard reagent, followed by transmetallation with zinc chloride.
- Execute the Negishi coupling reaction between the pyridine intermediate and the zinc reagent using tetrakis(triphenylphosphine)palladium(0) catalyst at mild temperatures (0-40°C).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this Negishi-based synthesis represents a strategic opportunity to optimize the cost structure and reliability of the Tenibulin supply chain. By eliminating the dependency on expensive boronic ester starting materials and avoiding the logistical complexities associated with cryogenic reagents like n-butyllithium, manufacturers can achieve substantial cost savings. The simplified workflow reduces the number of unit operations and minimizes the consumption of specialized solvents and energy-intensive cooling utilities. This operational efficiency translates directly into a more competitive pricing model for the final active pharmaceutical ingredient, allowing partners to secure a reliable pharmaceutical intermediate supplier relationship that is resilient to market fluctuations in raw material costs.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the use of commodity-grade starting materials and the elimination of costly purification steps. The high yield of the Negishi coupling step means that less raw material is wasted, improving the overall atom economy of the process. Additionally, the avoidance of microwave reactors and cryogenic chillers reduces capital investment and maintenance costs, leading to a lower cost of goods sold (COGS) without compromising on quality standards.
- Enhanced Supply Chain Reliability: From a logistics perspective, the removal of hazardous pyrophoric reagents significantly de-risks the supply chain. Transporting and storing n-butyllithium requires specialized containers and strict safety protocols, which can introduce delays and regulatory hurdles. By substituting these with stable zinc salts and magnesium powder, the manufacturing process becomes more robust and less susceptible to disruptions. This stability ensures consistent delivery schedules and reducing lead time for high-purity pharmaceutical intermediates, which is critical for meeting clinical and commercial demand.
- Scalability and Environmental Compliance: The mild reaction conditions (0-40°C) make this process inherently scalable from pilot plant to commercial production. There is no need for specialized high-pressure or high-temperature equipment, allowing the synthesis to be performed in standard glass-lined or stainless steel reactors. Furthermore, the reduced energy footprint and the potential for solvent recycling align with increasingly stringent environmental regulations, facilitating smoother regulatory approvals and enhancing the sustainability profile of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Tenibulin using this patented Negishi coupling methodology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this route is superior for industrial applications. Understanding these nuances is essential for technical teams evaluating process feasibility and for commercial teams negotiating supply agreements.
Q: Why is the Negishi coupling preferred over Suzuki coupling for Tenibulin synthesis?
A: The Negishi coupling utilizes organozinc reagents which exhibit higher nucleophilicity compared to boronic acid derivatives used in Suzuki coupling. This is particularly advantageous for electron-deficient pyridine substrates, allowing the reaction to proceed under much milder conditions (0-40°C) without the need for harsh microwave heating or expensive catalysts, resulting in significantly improved yields and operational safety.
Q: What are the safety advantages of this new synthetic route?
A: Unlike prior art routes that require cryogenic conditions (-60°C) using hazardous n-butyllithium or high-energy microwave irradiation at 150°C, this patented method operates at near-ambient temperatures. The elimination of pyrophoric reagents and extreme thermal inputs drastically reduces process hazards, making it inherently safer for large-scale industrial production.
Q: How does this method impact the cost of goods for Tenibulin?
A: The process reduces costs by shortening the synthetic sequence and utilizing cheaper starting materials compared to pinacol boronic esters. Furthermore, the higher reaction yields and simplified post-treatment procedures (avoiding complex purification steps associated with low-yield couplings) contribute to substantial overall manufacturing cost reductions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tenibulin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the fast-paced pharmaceutical industry. Our team of expert chemists has thoroughly analyzed the technological advancements presented in CN113999166B and is fully equipped to implement this Negishi coupling strategy for the commercial production of Tenibulin. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to manufacturing plant is seamless. Our state-of-the-art facilities are designed to handle organometallic chemistry safely, and our rigorous QC labs guarantee that every batch meets stringent purity specifications required for global regulatory submissions.
We invite potential partners to engage with us to explore how this optimized synthesis can benefit your specific project needs. By collaborating with our technical team, you can gain access to a Customized Cost-Saving Analysis that quantifies the economic advantages of switching to this route. We encourage you to contact our technical procurement team today to request specific COA data and discuss route feasibility assessments tailored to your supply chain requirements, ensuring a secure and cost-effective source for this vital dermatological API.
