Revolutionizing Alkyl Phenylboronic Acid Production with One-Pot Grignard Technology
The landscape of organic boronic compound manufacturing is undergoing a significant transformation driven by the need for safer, more cost-effective synthetic routes. Patent CN101775029A introduces a groundbreaking convenient preparation technique for alkyl substituted phenyloboricacid, a critical building block widely utilized in the fields of medicine, agrochemicals, and advanced materials. This innovation addresses the longstanding inefficiencies of traditional methods by integrating Grignard reagent preparation and low-temperature nucleophilic substitution into a seamless one-pot operation. By shifting the reaction paradigm from cryogenic conditions to mild ambient temperatures, this technology not only enhances product yield but also drastically simplifies the production infrastructure required for large-scale manufacturing. For industry leaders seeking a reliable pharmaceutical intermediates supplier, understanding this technological leap is essential for optimizing supply chains and reducing the overall cost of goods sold in complex drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkyl substituted benzene boric acid has been plagued by significant operational hurdles and safety concerns inherent to legacy chemical processes. The organolithium reagent method, while effective for certain substrates, necessitates extreme cryogenic conditions ranging from -78°C to -40°C, demanding specialized refrigeration equipment and consuming vast amounts of energy. Furthermore, organolithium reagents are notoriously hazardous, requiring rigorous exclusion of moisture and oxygen, which complicates handling and increases the risk of industrial accidents. Alternatively, Palladium-catalyzed oxygen borylation offers milder conditions but introduces the prohibitive cost of precious metal catalysts and the persistent challenge of heavy metal contamination, which is unacceptable for high-purity API intermediates. These conventional pathways often result in lower atom economy, higher solvent consumption, and complex workup procedures that erode profit margins and extend lead times for critical chemical supplies.
The Novel Approach
The novel approach detailed in the patent data revolutionizes this landscape by employing a streamlined Grignard reagent method that operates under remarkably mild conditions. By combining the formation of the Grignard reagent and the subsequent nucleophilic substitution with trialkyl borate into a single vessel, the process eliminates the need for intermediate isolation and the associated losses. The reaction proceeds efficiently at temperatures between 25°C and 40°C, removing the energy burden of cryogenic cooling and allowing for the use of standard stainless steel reactors. This one-pot strategy significantly reduces the volume of solvent required, often by half compared to traditional multi-step protocols, thereby minimizing waste generation and lowering disposal costs. The result is a robust, scalable methodology that delivers high-purity products with simplified post-treatment steps, positioning it as a superior choice for cost reduction in fine chemical manufacturing.
Mechanistic Insights into One-Pot Grignard Borylation
The core of this technological advancement lies in the precise orchestration of the Grignard reaction mechanism within a unified reaction environment. The process initiates with the oxidative insertion of magnesium into the carbon-halogen bond of the alkyl bromobenzene substrate in the presence of tetrahydrofuran (THF), generating the reactive organomagnesium species in situ. Unlike traditional methods where this intermediate might be isolated or transferred under strict inert atmospheres, here it immediately engages with the trialkyl borate electrophile. The use of tri-n-butyl borate is particularly advantageous due to its stability and reactivity profile, facilitating a smooth nucleophilic attack by the Grignard carbon on the boron center. This concerted mechanism ensures that the highly reactive organometallic intermediate is consumed as soon as it is formed, minimizing side reactions such as homocoupling or decomposition that often plague batch processes.
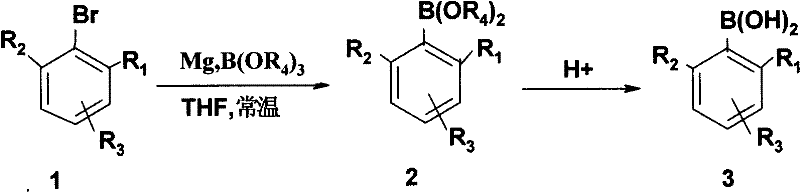
Following the formation of the alkylbenzene boric acid dialkyl intermediate, the system undergoes a controlled acidic hydrolysis to liberate the free boronic acid. The mechanistic elegance of this route is further evidenced by its tolerance to various substituents on the phenyl ring, provided they are compatible with Grignard chemistry. The subsequent workup involves a clever pH-swing strategy where the crude product is treated with dilute alkali to form a water-soluble boronate salt, allowing for the removal of organic impurities via steam distillation. Final acidification precipitates the pure alkyl substituted phenyloboricacid, effectively separating it from inorganic salts and residual organic byproducts. This sophisticated yet operationally simple sequence ensures that the final杂质 profile is minimized, meeting the stringent purity specifications required for downstream pharmaceutical applications without the need for chromatographic purification.
How to Synthesize Alkyl Substituted Phenyloboricacid Efficiently
Implementing this synthesis route requires careful attention to the addition rates and temperature control during the initial Grignard formation phase to prevent thermal runaway. The patent outlines a specific protocol where a solution of alkyl bromobenzene and trialkyl borate in THF is added dropwise to magnesium chips, maintaining the exotherm within the 25-40°C window to ensure optimal conversion. Detailed standardized operating procedures regarding stoichiometry, agitation speeds, and hydrolysis conditions are critical for reproducing the high yields reported in the experimental data. For process chemists looking to adopt this technology, the following guide summarizes the critical unit operations required to transition from laboratory benchtop to pilot plant scale effectively.
- Add a tetrahydrofuran solution of alkyl bromobenzene and trialkyl borate dropwise into a reactor containing magnesium chips, maintaining a temperature of 25-40°C for approximately 3 hours.
- Pour the resulting reaction mixture into cold diluted mineral acid for hydrolysis, followed by extraction and distillation to reclaim the organic solvent.
- Add dilute alkaline solution to the concentrated residue, remove organic solvents via aqueous vapor distillation, filter, acidify to precipitate crystals, and dry to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot Grignard technology translates directly into tangible strategic advantages regarding cost stability and supply continuity. By eliminating the dependency on cryogenic infrastructure and expensive palladium catalysts, the capital expenditure (CAPEX) required for production facilities is significantly lowered, while operational expenditures (OPEX) are reduced through decreased energy consumption and solvent usage. The simplification of the workflow from multiple steps to a single pot reduces the manpower hours required per batch and minimizes the potential for human error during transfers, thereby enhancing overall plant throughput. Furthermore, the use of readily available raw materials like magnesium and tri-n-butyl borate mitigates the risk of supply chain disruptions often associated with specialized organometallic reagents, ensuring a more resilient sourcing strategy for critical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the complete avoidance of precious metal catalysts which represent a major cost driver in alternative borylation methods. Additionally, the reduction in solvent volume by approximately half leads to substantial savings in both raw material procurement and waste treatment costs, as less hazardous waste requires disposal. The energy efficiency gained by operating at ambient temperature rather than cryogenic conditions further compounds these savings, resulting in a significantly lower cost of goods sold that can be passed down the value chain or retained as improved margin.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this synthetic route ensures consistent delivery schedules by reducing the complexity of the manufacturing process. The use of stable, commodity-grade reagents means that production is less susceptible to the volatility of the specialty chemical market, providing a secure foundation for long-term supply agreements. Moreover, the simplified equipment requirements allow for greater flexibility in manufacturing sites, enabling production to be scaled or shifted more easily in response to global demand fluctuations without compromising product quality or availability.
- Scalability and Environmental Compliance: The environmental footprint of this manufacturing process is markedly smaller, aligning with increasingly stringent global regulations on industrial emissions and waste. The reduction in solvent usage and the elimination of heavy metal contaminants simplify the effluent treatment process, reducing the burden on environmental compliance teams. This green chemistry advantage not only lowers regulatory risk but also enhances the brand value of the final pharmaceutical or agrochemical products by supporting sustainability goals, making it an attractive option for eco-conscious multinational corporations seeking responsible partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method for alkyl substituted phenyloboricacid. These insights are derived directly from the patent specifications and are designed to clarify the operational benefits and feasibility for industrial adoption. Understanding these details is crucial for technical teams evaluating the integration of this process into existing manufacturing portfolios to ensure alignment with quality and efficiency targets.
Q: How does this one-pot Grignard method compare to traditional organolithium synthesis?
A: Unlike organolithium methods which require hazardous low temperatures (-78°C to -40°C) and expensive reagents, this novel Grignard approach operates at mild room temperatures (25-40°C), significantly improving operational safety and reducing energy costs while maintaining high yields.
Q: What are the cost benefits of avoiding Palladium catalysts in boronic acid synthesis?
A: By utilizing a Grignard-based route instead of Palladium-catalyzed borylation, manufacturers eliminate the need for expensive precious metal catalysts and the complex downstream purification steps required to remove heavy metal residues, leading to substantial reductions in raw material and processing costs.
Q: Is this synthesis method scalable for industrial production of API intermediates?
A: Yes, the process simplifies production equipment by combining two reaction steps into one pot and reduces solvent usage by half, making it highly suitable for commercial scale-up with simplified post-treatment procedures like steam distillation and crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Substituted Phenyloboricacid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this one-pot Grignard technology in delivering high-value intermediates for the global pharmaceutical and agrochemical industries. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical, large-volume manufacturing. Our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of Grignard chemistry, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards of our international clientele.
We invite you to collaborate with us to leverage this efficient synthesis route for your next project, unlocking new levels of cost efficiency and supply security. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can accelerate your development timelines and optimize your supply chain performance.
