Advanced Catalytic Synthesis of Anacetrapib Intermediate for Scalable Pharmaceutical Production
Advanced Catalytic Synthesis of Anacetrapib Intermediate for Scalable Pharmaceutical Production
The pharmaceutical industry continuously seeks robust synthetic routes for complex small molecule inhibitors, particularly those targeting cardiovascular diseases. Patent CN109384807B introduces a significant breakthrough in the preparation of a critical intermediate for Anacetrapib, a potent Cholesteryl Ester Transfer Protein (CETP) inhibitor used to treat atherosclerosis and coronary heart disease. This novel methodology focuses on the efficient synthesis of (4-fluoro-5-isopropyl-2-methoxyphenyl)boronic acid, a key building block that dictates the quality and cost-efficiency of the final Active Pharmaceutical Ingredient (API). By leveraging advanced magnesium-lithium catalytic systems, this technology overcomes the severe limitations of traditional cryogenic processes, offering a pathway that is not only chemically superior but also commercially viable for large-scale manufacturing. The structural complexity of Anacetrapib, as illustrated below, demands precise intermediate synthesis to maintain stereochemical integrity and minimize impurity profiles.
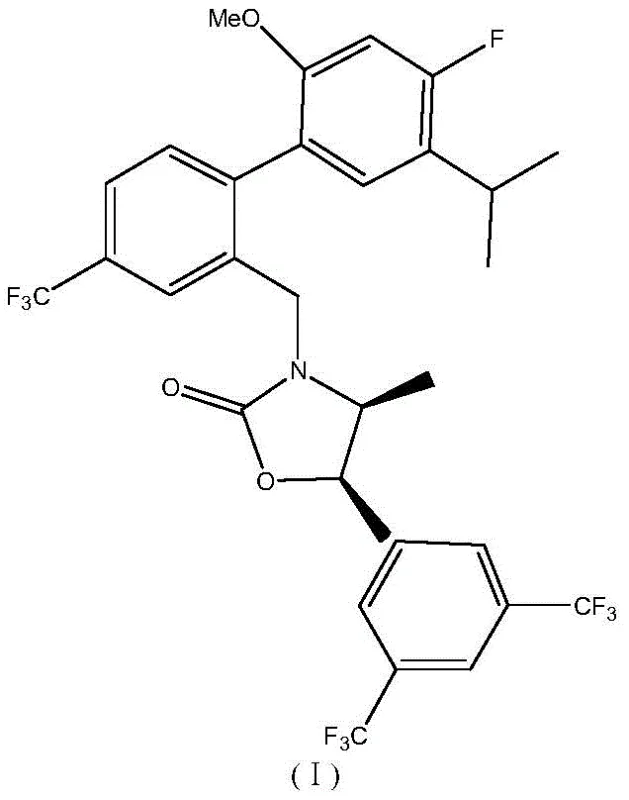
Anacetrapib represents a sophisticated therapeutic agent with the chemical name (4S, 5R)-5-[3, 5-bis(trifluoromethyl)phenyl]-3-[[2-(4-fluoro-2-methyloxy-5-prop-2-ylphenyl)-5-(trifluoromethyl)phenyl]methyl]-4-methyl-1, 3-oxazolidin-2-one. The synthesis of such a complex molecule relies heavily on the availability of high-purity precursors. The intermediate highlighted in this patent, specifically the boronic acid derivative shown as Formula II, serves as a pivotal coupling partner in the construction of the biaryl framework essential for the drug's biological activity. As a reliable pharmaceutical intermediate supplier, understanding the nuances of generating this specific boronic acid moiety is crucial for ensuring supply chain continuity for global drug developers. The transition from laboratory-scale curiosity to industrial reality requires a process that balances reaction kinetics with operational safety, a balance that this patent successfully achieves through innovative catalysis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl boronic acids from their corresponding bromo-precursors has been plagued by harsh reaction conditions and safety hazards. Prior art, such as that described in patent WO2007/005572, relied heavily on n-butyllithium as the metalating agent. This approach necessitates extremely low reaction temperatures, typically ranging from -80°C to -100°C, to control the exothermic nature of the lithiation and prevent decomposition. Such cryogenic conditions impose a massive burden on manufacturing infrastructure, requiring specialized cooling equipment and significant energy consumption. Furthermore, n-butyllithium is notoriously pyrophoric, posing severe safety risks regarding spontaneous combustion upon exposure to air or moisture. Alternative methods utilizing isopropyl magnesium chloride lithium chloride have been explored, as noted in US2014/0303380A1, but these often suffer from low reaction activity, resulting in sluggish kinetics and suboptimal yields that are economically unfeasible for commercial production.
The Novel Approach
The technology disclosed in CN109384807B revolutionizes this transformation by introducing a suite of highly active magnesium-lithium catalysts that operate under remarkably mild conditions. Instead of relying on dangerous organolithium reagents at cryogenic temperatures, this method utilizes catalysts such as lithium diisopropylmagnesium chloride (i-Pr2Mg·LiCl), TMPMgCl·LiCl, or MesMgBr·LiCl. These catalysts facilitate the halogen-metal exchange and subsequent boronation at temperatures between 0°C and 10°C, a dramatic improvement that eliminates the need for expensive cryogenic cooling. The reaction scheme below illustrates the direct conversion of the bromo-precursor (Formula III) into the desired boronic acid (Formula II) using triisopropyl borate. This streamlined approach not only accelerates the reaction rate, completing the transformation in just 1 to 4 hours, but also ensures a single-product profile that simplifies downstream purification.
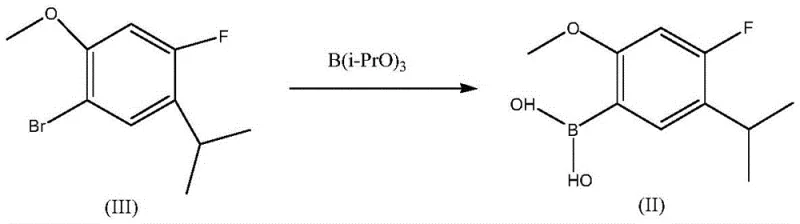
The implementation of this novel catalytic system translates directly into tangible benefits for cost reduction in API manufacturing. By shifting the operating window from -80°C to near-ambient temperatures, manufacturers can utilize standard reactor vessels without specialized low-temperature jackets, significantly lowering capital expenditure. Moreover, the enhanced reactivity of the magnesium-lithium complexes ensures that the conversion of the starting material, 4-fluoro-1-bromo-2-methoxy-5-isopropylbenzene, proceeds to completion with minimal formation of side products. This high selectivity is critical for maintaining the purity specifications required for pharmaceutical intermediates, reducing the need for resource-intensive chromatographic separations. The ability to use common solvents like toluene, acetone, or ethyl acetate further enhances the economic attractiveness of this route, making it an ideal candidate for scale-up in multi-ton production campaigns.
Mechanistic Insights into Magnesium-Lithium Catalyzed Boronation
The core innovation of this patent lies in the specific coordination chemistry of the magnesium-lithium catalysts employed. Unlike simple Grignard reagents which can be sluggish or prone to Wurtz-type coupling side reactions, the heterobimetallic species formed by i-Pr2Mg·LiCl or TMPMgCl·LiCl exhibit superior nucleophilicity and stability. The mechanism likely involves a rapid halogen-magnesium exchange where the bulky, electron-rich magnesium species displaces the bromine atom on the aromatic ring to form an aryl-magnesium intermediate. The presence of lithium chloride plays a crucial role in stabilizing this organometallic species through complexation, preventing aggregation and maintaining high reactivity even at elevated temperatures relative to lithiation. Once the aryl-magnesium species is generated, it immediately attacks the electrophilic boron center of triisopropyl borate, forming a tetra-coordinate borate complex. Subsequent acidic hydrolysis during the workup phase liberates the free boronic acid functionality.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over traditional lithiation. The milder basicity of the magnesium reagents compared to n-butyllithium reduces the risk of deprotonating sensitive functional groups on the aromatic ring, such as the methoxy or fluoro substituents, which could lead to defluorination or demethylation byproducts. The patent data indicates that the reaction yields a single major product, suggesting that the catalyst system effectively suppresses competing homocoupling reactions (Biaryl formation) that often plague metal-halogen exchange processes. This high level of chemoselectivity is paramount for R&D directors focused on impurity profiling, as it minimizes the burden on analytical teams to identify and quantify trace genotoxic impurities. The result is a robust process capable of delivering material with purity levels exceeding 99%, as demonstrated in the experimental examples where yields consistently hovered around 95-96%.
How to Synthesize (4-fluoro-5-isopropyl-2-methoxyphenyl)boronic acid Efficiently
Executing this synthesis requires careful attention to stoichiometry and temperature control to maximize the benefits of the catalytic system. The general procedure involves dissolving the bromo-substrate and triisopropyl borate in a suitable aprotic solvent, followed by the controlled addition of the magnesium-lithium catalyst. The reaction is exothermic, so maintaining the temperature within the 0-10°C range is essential to prevent thermal runaway while ensuring complete conversion. Following the reaction period, a standard aqueous workup involving phase separation and pH adjustment allows for the isolation of the product. For detailed operational parameters and specific molar ratios optimized for different solvent systems, please refer to the standardized synthesis guide below.
- React 4-fluoro-1-bromo-2-methoxy-5-isopropylbenzene with triisopropyl borate in a solvent like acetone or toluene.
- Add the catalyst lithium diisopropylmagnesium chloride at a temperature between 0-10°C and stir for 1-4 hours.
- Perform aqueous workup involving phase separation, NaOH extraction, and acidification to isolate the pure boronic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers a strategic advantage in securing a stable and cost-effective supply of critical intermediates. The shift away from hazardous reagents like n-butyllithium significantly mitigates supply chain risks associated with the storage and handling of pyrophoric materials, which often require specialized logistics and insurance. Furthermore, the simplified process flow, characterized by shorter reaction times and ambient temperature operation, enhances the overall throughput of manufacturing facilities. This efficiency gain means that production slots can be turned over more rapidly, allowing suppliers to respond more agilely to fluctuating market demands for Anacetrapib precursors. The use of commodity chemicals and solvents also insulates the supply chain from volatility in the pricing of exotic reagents.
- Cost Reduction in Manufacturing: The elimination of cryogenic cooling requirements represents a substantial reduction in utility costs, as maintaining reactors at -80°C is energy-intensive and expensive. Additionally, the high molar yields (consistently above 93%) mean that less raw material is wasted, directly improving the cost of goods sold (COGS). The simplified purification process, which relies on acid-base extraction rather than complex chromatography, further reduces solvent consumption and waste disposal costs, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: By utilizing catalysts that are less sensitive to moisture and air compared to n-butyllithium, the process becomes more robust against minor variations in operational conditions. This robustness translates to fewer batch failures and a more predictable delivery schedule for downstream API manufacturers. The availability of multiple effective catalyst options (i-Pr2Mg·LiCl, TMPMgCl·LiCl, MesMgBr·LiCl) provides flexibility in sourcing, ensuring that production is not bottlenecked by the shortage of a single specific reagent.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of greener solvents like acetone or ethyl acetate align well with modern environmental, health, and safety (EHS) standards. Scaling this process from pilot plant to commercial tonnage does not require exponential increases in safety infrastructure, making it easier to validate and approve for GMP production. The reduction in hazardous waste generation due to higher selectivity and yield also simplifies regulatory compliance and lowers the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic boronation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, aiming to clarify the operational benefits for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production lines.
Q: What are the advantages of the new magnesium-lithium catalyst over n-butyllithium?
A: The new catalyst system operates at mild temperatures (0-10°C) compared to the cryogenic conditions (-80°C to -100°C) required for n-butyllithium, significantly reducing energy costs and safety risks associated with pyrophoric reagents.
Q: What purity levels can be achieved with this synthesis method?
A: The patented process consistently achieves purity levels exceeding 99%, with molar yields ranging from 93% to 96%, ensuring high-quality material for downstream API synthesis.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes simple solvents like toluene or acetone and avoids hazardous cryogenic operations, making it highly scalable and compliant with industrial safety standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (4-fluoro-5-isopropyl-2-methoxyphenyl)boronic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals like Anacetrapib depends on the reliability of the supply chain for key intermediates. Our technical team has thoroughly analyzed the catalytic mechanisms described in CN109384807B and is fully equipped to translate this laboratory innovation into industrial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project never faces supply bottlenecks. Our state-of-the-art facilities are designed to handle sensitive organometallic chemistry safely, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for API synthesis.
We invite you to collaborate with us to optimize your supply chain for cardiovascular therapeutics. By leveraging our expertise in magnesium-lithium catalysis, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced manufacturing capabilities can drive efficiency and quality in your Anacetrapib production program.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
