Scalable Synthesis of Octafluoro-Dibromobiphenyl for Advanced Optoelectronic Applications
Scalable Synthesis of Octafluoro-Dibromobiphenyl for Advanced Optoelectronic Applications
The rapid evolution of the organic electronics sector demands intermediates that combine structural precision with economic viability. Patent CN111393256B introduces a transformative synthetic methodology for producing 2,2',3,3',5,5',6,6'-octafluoro-4,4'-dibromobiphenyl, a critical building block for high-end photoelectric materials including OLEDs and organic field-effect transistors. This innovation addresses the longstanding bottlenecks of traditional synthesis by replacing hazardous cryogenic lithiation with a robust Grignard-based coupling strategy. By leveraging inexpensive magnesium metal and versatile copper catalysis, this route offers a compelling value proposition for manufacturers seeking to optimize their supply chains for complex fluorinated aromatics. The technical breakthrough lies not only in the chemical transformation but in the holistic improvement of process safety and scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of perfluorinated biphenyl derivatives has relied heavily on halogen-lithium exchange reactions, a pathway fraught with significant operational and economic challenges. As illustrated in the prior art, this conventional approach necessitates the use of n-butyllithium, a pyrophoric reagent that poses severe safety risks during handling and storage, particularly at an industrial scale. Furthermore, the reaction requires stringent cryogenic conditions, typically maintained at -78°C, to prevent uncontrolled side reactions and ensure regioselectivity. This demand for deep cooling imposes a massive energy burden on production facilities and limits the throughput of standard reactor vessels. Additionally, the subsequent coupling step often employs titanium tetrachloride in stoichiometric excess, generating substantial amounts of hazardous waste and complicating downstream purification processes. These factors collectively render the traditional lithiation route economically inefficient and environmentally unsustainable for mass production.
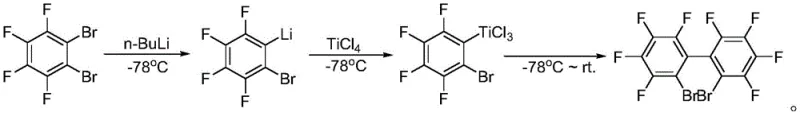
The Novel Approach
In stark contrast, the methodology disclosed in CN111393256B pioneers a streamlined two-step sequence that circumvents these critical limitations through the strategic application of Grignard chemistry. The process initiates with the formation of a tetrafluorophenyl magnesium bromide intermediate using metallic magnesium, a reagent that is abundant, cost-effective, and significantly safer to handle than organolithium compounds. This Grignard reagent then undergoes a copper-catalyzed self-coupling reaction to construct the biaryl backbone, eliminating the need for stoichiometric titanium reagents. The reaction conditions are markedly milder, operating effectively within a broad temperature range of -30°C to 100°C, which drastically reduces energy consumption and simplifies reactor engineering requirements. This novel pathway not only enhances the safety profile of the manufacturing process but also delivers superior yields exceeding 90%, establishing a new benchmark for efficiency in fluorinated intermediate synthesis.
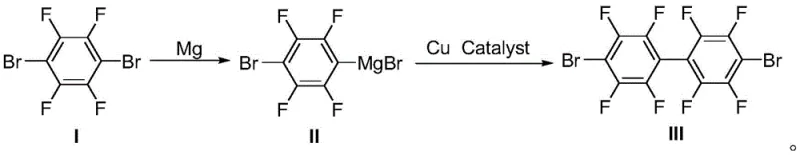
Mechanistic Insights into Copper-Catalyzed Self-Coupling
The core of this technological advancement resides in the efficient generation and utilization of the aryl Grignard species under controlled conditions. The reaction begins with the oxidative insertion of activated magnesium into the carbon-bromine bond of 2,3,5,6-tetrafluoro-1,4-dibromobenzene. The presence of multiple electron-withdrawing fluorine atoms on the aromatic ring facilitates this insertion, allowing the reaction to proceed smoothly in various ether solvents such as tetrahydrofuran or 2-methyltetrahydrofuran. Careful control of the initiation phase, potentially aided by additives like iodine or 1,2-dibromoethane, ensures complete conversion of the starting material while minimizing the formation of homocoupling byproducts or Wurtz-type side reactions. The resulting organomagnesium intermediate is stabilized by the solvent coordination, creating a reactive species poised for the subsequent cross-coupling event without the need for isolation, thereby telescoping the process for maximum efficiency.
Following the Grignard formation, the introduction of a copper catalyst triggers the self-coupling mechanism, likely proceeding through a single-electron transfer or oxidative addition-reductive elimination cycle typical of Ullmann-type reactions. The copper species, whether introduced as cuprous chloride, bromide, or oxide, mediates the coupling of two aryl groups to form the sterically crowded octafluorobiphenyl structure. A key advantage of this catalytic system is its tolerance to the highly fluorinated substrate, which can often deactivate other transition metal catalysts. The mechanism effectively suppresses the formation of poly-coupled oligomers, ensuring that the primary product is the desired dibromobiphenyl dimer. This high selectivity is crucial for downstream applications in organic electronics, where trace impurities can severely degrade the performance of the final optoelectronic device, thus underscoring the importance of this specific catalytic protocol.
How to Synthesize 2,2',3,3',5,5',6,6'-octafluoro-4,4'-dibromobiphenyl Efficiently
The practical implementation of this synthesis involves a carefully orchestrated sequence of reagent addition and temperature management to maximize yield and purity. Operators must first ensure the absolute exclusion of moisture, as the Grignard intermediate is highly sensitive to hydrolysis, which would quench the reaction and reduce overall efficiency. The process begins with the activation of magnesium metal in an anhydrous ether solvent, followed by the controlled addition of the dibromotetrafluorobenzene substrate to generate the organometallic species. Once the Grignard formation is complete, the solution is transferred to a reactor containing the copper catalyst, where the coupling reaction proceeds to completion. Detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures, are outlined in the guide below to ensure reproducible results for R&D teams scaling this technology.
- React 2,3,5,6-tetrafluoro-1,4-dibromobenzene with magnesium metal in an inert ether solvent to form the Grignard intermediate.
- Perform a copper-catalyzed self-coupling reaction of the Grignard intermediate in an inert solvent to form the biphenyl structure.
- Quench the reaction, extract the organic phase, and purify via recrystallization to achieve high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from lithiation to Grignard coupling represents a significant opportunity for cost optimization and risk mitigation. The replacement of n-butyllithium with magnesium metal eliminates the dependency on a high-cost, hazard-class 4.2 reagent that requires specialized logistics and storage infrastructure. Magnesium is a commodity chemical with a stable global supply chain, ensuring consistent availability and shielding production schedules from the volatility often associated with specialty organometallic reagents. Furthermore, the relaxation of temperature constraints from cryogenic -78°C to near-ambient or moderate heating conditions allows for the utilization of standard glass-lined reactors without the need for expensive cryogenic cooling loops. This reduction in capital expenditure and operational complexity translates directly into a more competitive cost structure for the final intermediate.
- Cost Reduction in Manufacturing: The elimination of expensive reagents like n-butyllithium and titanium tetrachloride fundamentally alters the bill of materials, driving down the variable cost per kilogram of production. By utilizing catalytic amounts of inexpensive copper salts instead of stoichiometric titanium reagents, the process generates significantly less heavy metal waste, thereby reducing the costs associated with waste treatment and disposal. The higher reaction yields reported in the patent examples, consistently hovering above 90%, mean that less raw material is wasted, further enhancing the overall material efficiency. These cumulative factors result in a substantially lower cost of goods sold, providing a distinct margin advantage in the competitive market for electronic chemicals.
- Enhanced Supply Chain Reliability: Relying on commodity-grade magnesium and widely available copper catalysts mitigates the risk of supply disruptions that can occur with niche specialty reagents. The robustness of the reaction conditions allows for greater flexibility in sourcing solvents, as the process is compatible with a wide range of ethers including THF, 2-MeTHF, and dioxane. This solvent flexibility enables manufacturers to adapt quickly to regional availability or price fluctuations without compromising reaction performance. Additionally, the improved safety profile reduces the regulatory burden and insurance costs associated with handling pyrophoric materials, ensuring smoother operations and uninterrupted supply continuity for downstream clients in the semiconductor and display industries.
- Scalability and Environmental Compliance: The simplified workflow and reduced hazard profile make this process inherently more scalable, facilitating the transition from pilot plant to multi-ton commercial production with minimal engineering hurdles. The avoidance of stoichiometric titanium waste aligns with increasingly stringent environmental regulations regarding heavy metal discharge, positioning manufacturers as responsible partners in the green chemistry initiative. The ability to operate at higher concentrations and warmer temperatures also improves the space-time yield of the reactors, allowing facilities to produce more material within the same timeframe. This scalability ensures that the supply chain can readily expand to meet the growing global demand for high-performance organic electronic materials without requiring prohibitive capital investment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process parameters and expected outcomes. Understanding these details is essential for R&D teams evaluating the feasibility of adopting this technology for their specific manufacturing lines. The answers reflect the balance between theoretical potential and practical operational realities observed in the described examples.
Q: What are the primary advantages of the Grignard route over the traditional lithiation method?
A: The Grignard route eliminates the need for cryogenic temperatures (-78°C) and expensive, hazardous n-butyllithium, significantly reducing operational costs and safety risks while maintaining high yields around 90%.
Q: Which copper catalysts are most effective for this self-coupling reaction?
A: The patent indicates that inorganic copper compounds such as cuprous chloride, cuprous bromide, and cuprous oxide are highly effective, offering flexibility in catalyst selection based on availability and cost.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method utilizes cheap raw materials like magnesium and avoids extreme low-temperature constraints, making it highly scalable and economically viable for commercial manufacturing of photoelectric intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2',3,3',5,5',6,6'-octafluoro-4,4'-dibromobiphenyl Supplier
As the demand for high-performance organic semiconductors continues to surge, securing a stable supply of critical intermediates like 2,2',3,3',5,5',6,6'-octafluoro-4,4'-dibromobiphenyl is paramount for maintaining production momentum. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent quality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for OLED and photovoltaic applications. We understand that in the fast-paced world of electronic materials, reliability is just as critical as chemical performance, and our dedicated team ensures that every batch meets the highest international standards.
We invite forward-thinking enterprises to collaborate with us to unlock the full potential of this advanced synthetic technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage potential partners to contact us directly to request specific COA data and comprehensive route feasibility assessments. Let us help you streamline your supply chain and accelerate your time-to-market with our premium grade fluorinated intermediates, ensuring your projects remain competitive and commercially viable in the global marketplace.
