Optimizing Corticosteroid Production: A Novel Bromination Route for Betamethasone Acetate
Optimizing Corticosteroid Production: A Novel Bromination Route for Betamethasone Acetate
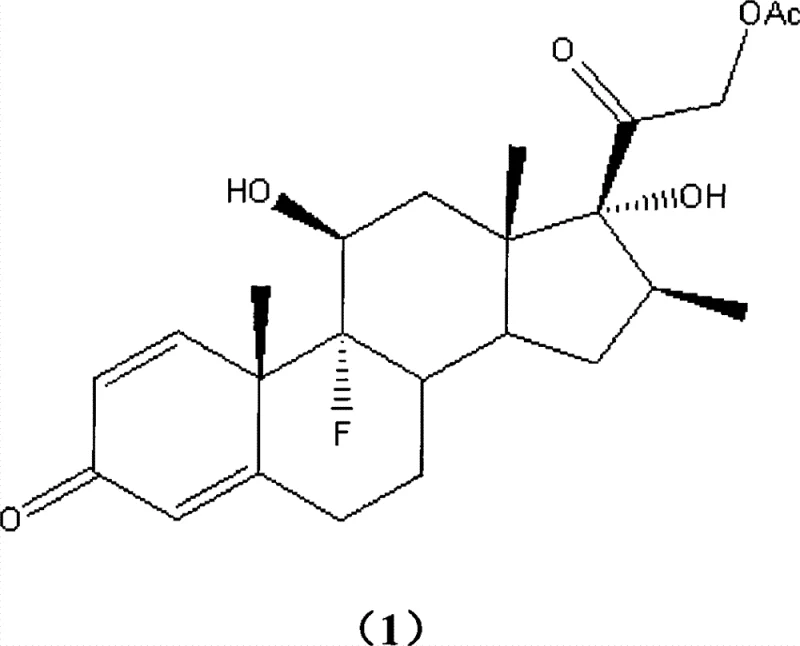
The pharmaceutical industry continuously seeks robust synthetic pathways for critical corticosteroids like Betamethasone acetate, a potent anti-inflammatory agent widely used in treating rheumatic arthritis and severe dermatitis. Patent CN101417912B introduces a transformative methodology that fundamentally alters the manufacturing landscape for this high-value active pharmaceutical ingredient (API). By shifting away from traditional iodination protocols toward a novel ammonium salt-catalyzed bromination strategy, this technology addresses long-standing inefficiencies regarding reagent cost, intermediate stability, and environmental impact. The process achieves exceptional purity levels exceeding 99% while utilizing readily available reagents, positioning it as a cornerstone for modern, scalable steroid synthesis. This report analyzes the technical merits and commercial implications of this innovation for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Betamethasone acetate has relied heavily on iodination chemistry, specifically involving the reaction of 17α-hydroxy-9,11β-epoxy-16β-methylpregna-1,4-diene-3,20-dione with elemental iodine. This conventional pathway suffers from significant economic and logistical drawbacks that hinder efficient large-scale production. The primary issue lies in the high cost and poor atom economy of elemental iodine, which necessitates the use of stoichiometric amounts to drive the reaction forward. Furthermore, the resulting diiodo intermediate is chemically unstable, requiring stringent storage conditions such as nitrogen protection and light exclusion to prevent decomposition. This instability creates bottlenecks in inventory management and increases the risk of batch failures during manufacturing.
The Novel Approach
The methodology disclosed in CN101417912B offers a decisive break from these constraints by employing brominating reagents such as N-bromosuccinimide (NBS) or 1,3-dibromo-5,5-dimethylhydantoin. This substitution not only lowers raw material costs but also generates a 21-bromo intermediate that exhibits superior stability compared to its iodo counterpart. The process eliminates the need for complex preservation measures, allowing the intermediate to be stored for extended periods without degradation. Additionally, the reaction conditions are remarkably mild, operating effectively between 20°C and 100°C, which reduces energy consumption and simplifies reactor requirements. This streamlined approach facilitates a more continuous and reliable manufacturing workflow.
Mechanistic Insights into Ammonium Salt-Catalyzed Bromination
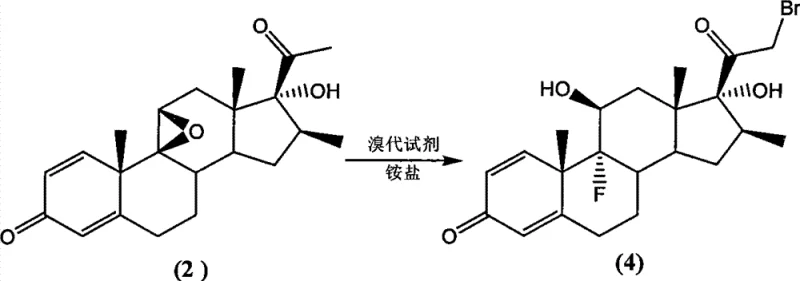
The core innovation of this synthesis lies in the strategic use of ammonium salts as catalysts during the bromination step. Mechanistically, the ammonium salt—selected from options like ammonium acetate or ammonium formiate—activates the reaction environment, enabling the brominating reagent to selectively target the C-21 position of the steroid backbone. Without this catalytic presence, the reaction between the starting ketone and the brominating agent would either fail to initiate or produce a complex mixture of undesirable by-products. The catalyst ensures high regioselectivity, driving the conversion of the starting material (Compound 2) into the 21-bromo intermediate (Compound 4) with yields ranging from 80% to 99.5%. This high efficiency minimizes waste generation and maximizes the throughput of valuable steroid intermediates.
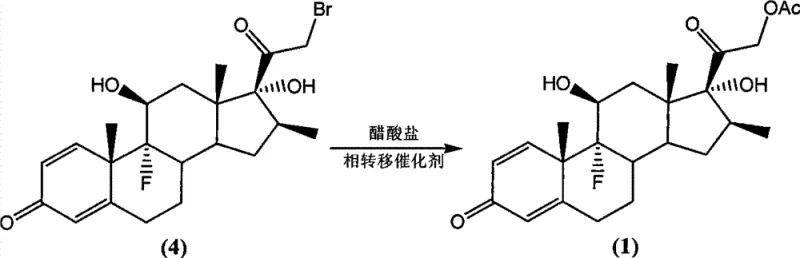
Following the bromination, the process utilizes phase transfer catalysis (PTC) for the subsequent acetylation step. In this stage, the 21-bromo intermediate reacts with acetate salts in the presence of quaternary ammonium salts, such as tetrabutylammonium bromide. The phase transfer catalyst facilitates the movement of the acetate anion into the organic phase, where it displaces the bromine atom to form the final acetate ester. This mechanism allows the reaction to proceed rapidly at moderate temperatures (50°C to 120°C) with yields reaching up to 99%. The combination of ammonium salt catalysis in the first step and PTC in the second creates a synergistic effect that ensures high purity (>99%) and minimizes the formation of difficult-to-remove impurities, which is critical for meeting stringent pharmacopeial standards.
How to Synthesize Betamethasone Acetate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing high-quality Betamethasone acetate with minimal purification burden. The process begins with the dissolution of the epoxy-ketone starting material in a suitable solvent like ether or toluene, followed by the addition of the brominating agent and the catalytic ammonium salt. After stirring for a defined period, the reaction mixture is filtered to remove solids, and the solvent is evaporated to yield the crude bromo-intermediate, which can be used directly in the next step without further purification. This telescoping capability significantly reduces processing time and solvent usage. For the detailed standardized synthesis steps including specific molar ratios and workup procedures, please refer to the guide below.
- React 17α-hydroxy-9,11β-epoxy-16β-methylpregna-1,4-diene-3,20-dione with a brominating reagent (e.g., NBS) and catalytic ammonium salt in solvent at 20-100°C to obtain the 21-bromo intermediate.
- Separate the liquid phase, remove solvent, and collect the intermediate compound without purification for the next step.
- React the intermediate with an acetate salt and a phase transfer catalyst in solvent at 50-120°C to yield high-purity Betamethasone acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this bromination-based route offers substantial strategic benefits that extend beyond simple chemical yield. The elimination of elemental iodine removes a volatile cost factor from the bill of materials, as iodine prices can fluctuate significantly based on global mining outputs. Furthermore, the stability of the bromo-intermediate allows manufacturers to build strategic stockpiles without the fear of rapid degradation, thereby enhancing supply continuity and reducing the risk of production stoppages due to raw material shortages. The ability to recycle solvents such as methylene chloride and toluene further contributes to a leaner cost structure by reducing waste disposal fees and raw solvent procurement needs.
- Cost Reduction in Manufacturing: The replacement of expensive iodine with cost-effective brominating reagents like NBS leads to a direct reduction in raw material expenses. Additionally, the high atom economy of the bromination reaction means less reagent is wasted, optimizing the overall material balance. The process also avoids the need for specialized equipment required to handle corrosive iodine vapors, lowering capital expenditure and maintenance costs associated with reactor linings and ventilation systems.
- Enhanced Supply Chain Reliability: The chemical stability of the 21-bromo intermediate is a game-changer for logistics. Unlike the light-sensitive and unstable diiodo compounds used in traditional methods, the bromo-intermediate can be stored under standard conditions for extended periods. This stability allows for flexible production scheduling and the decoupling of the bromination and acetylation steps, enabling manufacturers to respond more agilely to market demand fluctuations without compromising product integrity.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of common organic solvents make this process highly scalable from pilot plant to commercial tonnage. The significant reduction in "three wastes" (waste water, gas, and solid) discharge aligns with increasingly strict environmental regulations. By minimizing toxic by-products and enabling solvent recovery, this method supports sustainable manufacturing practices, reducing the environmental footprint of API production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on reaction parameters and product quality. Understanding these details is essential for R&D teams evaluating the feasibility of technology transfer.
Q: Why is the bromination method superior to the traditional iodination route?
A: The bromination method replaces expensive elemental iodine with low-cost brominating reagents like NBS. It avoids the formation of unstable diiodo intermediates that require nitrogen protection and dark storage, significantly improving process stability and atom economy.
Q: What is the role of the ammonium salt in the first reaction step?
A: The ammonium salt acts as a crucial catalyst. Without it, the reaction between the starting steroid and the brominating reagent does not proceed or yields excessive by-products. Its addition drastically improves reaction selectivity and yield.
Q: Can the solvents used in this process be recycled?
A: Yes, the patent explicitly states that the solvents used in both the bromination and acetylation steps can be recovered and reused, which contributes to reduced waste discharge and lower operational costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Betamethasone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant synthesis routes for high-value corticosteroids. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering Betamethasone acetate that meets stringent purity specifications through our rigorous QC labs, guaranteeing consistency for your downstream formulation processes. Our facility is equipped to handle the specific solvent recovery and waste treatment requirements of this bromination process, ensuring full environmental compliance.
We invite global partners to leverage our expertise for their API manufacturing needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can optimize your supply chain and reduce total landed costs.
