Advanced Ionic Liquid Catalysis for Commercial Scale-up of Complex Corticosteroids
The pharmaceutical industry continuously seeks robust synthetic routes for high-value corticosteroids, and patent CN101125875A presents a transformative approach for the preparation of Fluticasone Propionate. This specific intellectual property details a novel methodology that replaces traditional, hazardous reagents with a sophisticated ionic liquid catalytic system. By leveraging the unique solvation properties of imidazolium-based ionic liquids, the process achieves exceptional reaction efficiency and product purity. For R&D directors and procurement specialists, this technology represents a pivotal shift away from expensive heavy metal catalysts towards a more sustainable and economically viable manufacturing paradigm. The integration of these advanced chemical principles ensures that the production of this critical respiratory medication can be scaled with greater confidence and reduced environmental liability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for Fluticasone Propionate, such as those disclosed in US Pat 4335121, have long been plagued by significant operational inefficiencies and prohibitive costs. These legacy methods typically rely on silver fluoride for the critical fluorine-iodine exchange step, a reagent known for its extreme expense and sensitivity to light and moisture. Furthermore, the handling of silver salts generates substantial heavy metal waste, complicating disposal and increasing the overall environmental footprint of the manufacturing process. Alternative approaches, like those in World Patent WO2007/012228, attempted to mitigate some issues but introduced new risks by utilizing gaseous fluoromethane. This gaseous reagent is not only highly toxic and difficult to contain but also requires specialized high-pressure equipment, creating bottlenecks in supply chain continuity and posing severe safety risks to plant personnel.
The Novel Approach
In stark contrast, the methodology outlined in CN101125875A introduces a streamlined liquid-phase reaction system that effectively circumvents the drawbacks of prior art. By employing halogen monobromomethane in the initial alkylation step followed by a fluorination step mediated by tetraalkyl ammonium fluoride salts in an ionic liquid, the process achieves remarkable stability. The use of ionic liquids such as [Bmim][BF4] creates a highly polar environment that enhances the nucleophilicity of the fluoride ion without the need for volatile or hazardous solvents. This innovation allows the reaction to proceed under milder conditions, typically between 20°C and 100°C, significantly reducing energy consumption. The result is a synthetic pathway that is not only chemically superior but also inherently safer and more adaptable to the rigorous demands of modern Good Manufacturing Practice (GMP) facilities.
Mechanistic Insights into Ionic Liquid-Mediated Fluorination
The core of this technological advancement lies in the precise manipulation of nucleophilic substitution mechanisms within the ionic liquid medium. In the first stage, the thiocarboxylic acid precursor undergoes S-alkylation where the sulfur atom attacks the halogen monobromomethane in the presence of a mild base like potassium carbonate or sodium bicarbonate. This step is crucial for installing the necessary carbon scaffold for the subsequent fluorination. The reaction kinetics are optimized by the choice of solvent, with polar aprotic solvents like DMF or acetone facilitating the initial transformation with yields reaching as high as 99.5%. This high conversion rate minimizes the formation of unreacted starting materials, thereby simplifying the downstream purification workload and ensuring a consistent quality profile for the intermediate.
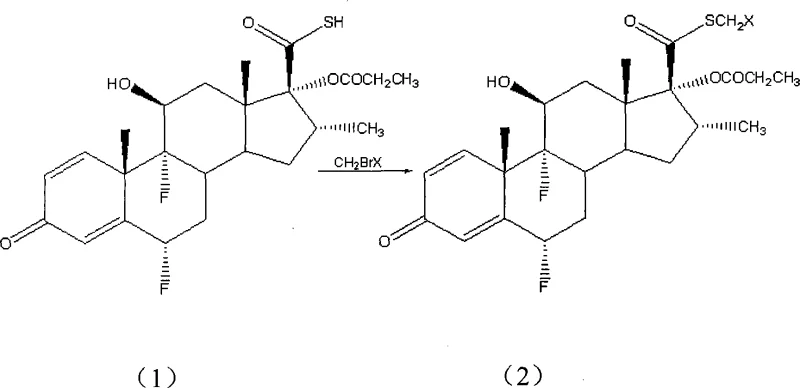
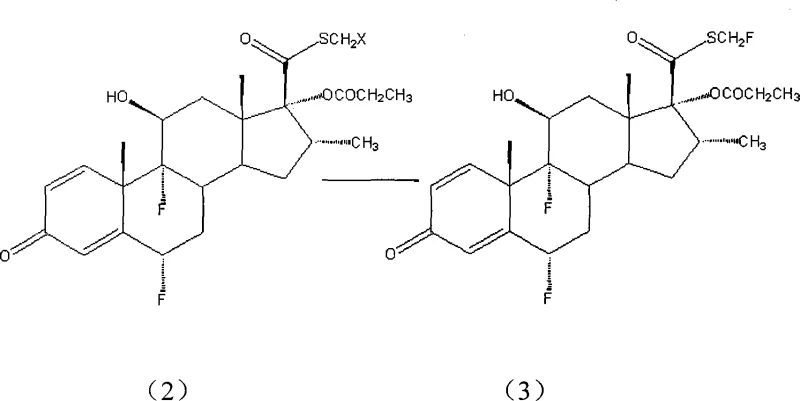
Following the alkylation, the process transitions to the critical fluorination step where the ionic liquid plays a dual role as both solvent and phase transfer catalyst. The tetraalkyl ammonium fluoride salt dissociates within the ionic liquid matrix, releasing free fluoride ions that are highly reactive yet stabilized against side reactions. This environment prevents the degradation of the sensitive steroid backbone, a common issue in traditional fluorination methods. The ionic liquid effectively solvates the transition state, lowering the activation energy required for the displacement of the leaving group. As demonstrated in the patent examples, this mechanism allows for the use of hydrated fluoride salts without compromising yield, a flexibility that is unprecedented in conventional anhydrous systems. The final product, Fluticasone Propionate, is obtained with purity levels exceeding 99%, validating the efficacy of this mechanistic approach.
How to Synthesize Fluticasone Propionate Efficiently
To implement this synthesis effectively, manufacturers must adhere to strict stoichiometric controls and temperature profiles as defined in the patent specifications. The process begins with the careful addition of the base and alkylating agent to the steroid precursor, ensuring that the exothermic nature of the reaction is managed to prevent thermal runaway. Following the isolation of the intermediate, the fluorination step requires the precise preparation of the ionic liquid mixture, ensuring that water content is controlled if anhydrous conditions are preferred, although the patent notes tolerance for hydrated salts. Detailed standard operating procedures regarding mixing speeds, addition rates, and crystallization protocols are essential for reproducibility.
- React the thiocarboxylic acid precursor with halogen monobromomethane in the presence of a base to form the S-alkylated intermediate.
- Perform nucleophilic fluorination on the intermediate using tetraalkyl ammonium fluoride salts within an ionic liquid solvent system.
- Isolate the final fluticasone propionate product through crystallization and washing, achieving purity greater than 99%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ionic liquid technology offers profound strategic benefits that extend beyond simple chemical yield. The elimination of silver fluoride removes a major cost driver from the bill of materials, as silver salts are subject to volatile market pricing and supply constraints. Additionally, the ability to recycle the ionic liquid solvent significantly reduces the volume of chemical waste requiring treatment, leading to substantial cost savings in waste disposal and environmental compliance. The use of stable, non-gaseous reagents also simplifies logistics, as there is no longer a need for specialized storage infrastructure for hazardous gases, thereby enhancing overall facility safety and reducing insurance premiums associated with chemical handling.
- Cost Reduction in Manufacturing: The replacement of expensive silver-based reagents with readily available tetraalkyl ammonium salts results in a drastic reduction in raw material costs. Furthermore, the high selectivity of the reaction minimizes the loss of valuable steroid intermediates, ensuring that every kilogram of input material contributes maximally to the final output. The recyclability of the ionic liquid medium means that solvent purchase costs are amortized over multiple batches, compounding the financial benefits over the lifecycle of the product. This economic efficiency makes the process highly competitive in the global generic pharmaceutical market where margin pressure is intense.
- Enhanced Supply Chain Reliability: By relying on stable, shelf-stable reagents rather than light-sensitive silver salts or pressurized gases, the supply chain becomes significantly more resilient. Manufacturers can maintain larger inventories of key reagents without fear of rapid degradation, ensuring continuous production even during periods of raw material scarcity. The simplified handling requirements also reduce the risk of shipping delays caused by hazardous material regulations, allowing for smoother international logistics. This reliability is critical for meeting the stringent delivery schedules demanded by large-scale pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are easily manageable in large stainless steel reactors. The reduction in heavy metal waste aligns perfectly with increasingly strict global environmental regulations, future-proofing the manufacturing site against regulatory changes. The ability to operate at moderate temperatures also lowers the energy load on the facility, contributing to a lower carbon footprint. These factors combined make the technology an ideal candidate for green chemistry initiatives and sustainable manufacturing certifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is vital for stakeholders evaluating the feasibility of adopting this technology for commercial production. The answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance.
Q: How does the ionic liquid method improve yield compared to traditional silver fluoride methods?
A: Traditional methods utilizing silver fluoride often suffer from low yields due to reagent instability and difficult purification. The ionic liquid method described in CN101125875A stabilizes the fluoride ion, enabling yields between 80% and 99% while simplifying post-reaction disposal.
Q: What are the safety advantages of avoiding gaseous fluoromethane in this synthesis?
A: Previous routes relied on gaseous fluoromethane, which poses significant toxicity and atmospheric pollution risks. This novel approach utilizes solid or liquid fluoride sources within a closed ionic liquid system, drastically reducing operational hazards and environmental impact.
Q: Is the ionic liquid solvent reusable for commercial scale production?
A: Yes, the patent highlights that the ionic liquid exhibits high stability and can be recycled. This reusability is a critical factor for reducing long-term manufacturing costs and minimizing chemical waste in large-scale pharmaceutical production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluticasone Propionate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the pharmaceutical sector. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the ionic liquid method for Fluticasone Propionate can be seamlessly transferred from the lab to the plant. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards. Our commitment to technological excellence allows us to offer clients a secure and high-quality supply of complex corticosteroids.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic advantages of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a successful and profitable collaboration.
