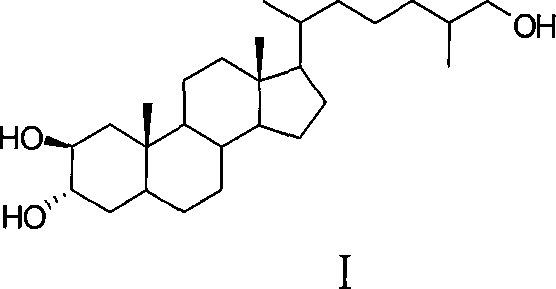
Scalable Synthesis of Bioactive Marine Steroid (25R)-5α-cholesta-2β,3α,26-triol for Pharmaceutical Applications
The pharmaceutical industry is constantly seeking novel bioactive scaffolds from marine sources, yet the supply of such compounds is often bottlenecked by their scarcity in nature. Patent CN101220073A addresses this critical challenge by disclosing a robust synthetic method for (25R)-5α-cholesta-2β,3α,26-triol, a polyhydroxy marine steroid with significant therapeutic potential. Unlike traditional extraction methods which rely on the limited biomass of marine organisms like Ophiarachna incrassata, this invention utilizes tigogenin, a cheap and abundant plant-derived sapogenin, as the starting material. The synthetic strategy involves a scientifically rational sequence of tosylation, elimination, reductive ring opening, and stereoselective functionalization to construct the unique 2β,3α,26-trihydroxy motif. This approach not only overcomes the supply limitations of natural products but also ensures consistent quality and purity essential for preclinical and clinical development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of complex marine steroids has been plagued by severe logistical and economic hurdles inherent to natural product isolation. The concentration of bioactive steroids like (25R)-5α-cholesta-2β,3α,26-triol in marine sponges and soft corals is typically trace-level, requiring the processing of tons of wet biomass to isolate mere milligrams of pure compound. This dependency on marine collection introduces massive variability in supply due to seasonal changes, environmental regulations, and ecological sustainability concerns. Furthermore, the purification of these compounds from complex marine extracts is technically demanding, often involving extensive chromatographic separation to remove structurally similar sterols and lipids, which drastically inflates the cost of goods. For pharmaceutical developers, relying on such an unstable supply chain poses a significant risk to project timelines and makes dose-ranging studies practically impossible to scale.
The Novel Approach
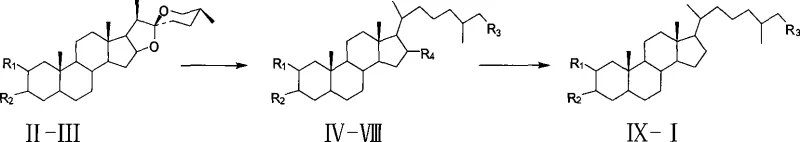
The methodology presented in the patent revolutionizes access to this scaffold by shifting the paradigm from extraction to semi-synthesis using tigogenin. This route leverages the existing steroidal backbone of tigogenin, strategically modifying the F-ring and A-ring to install the required functionality. By employing a reductive ring-opening strategy with zinc powder and ethanol, the spiroketal system is efficiently converted into the cholestane side chain with high fidelity. The subsequent steps utilize standard protecting group chemistry, such as TBDMS protection and mesylation, which are well-understood and easily controlled in a manufacturing setting. This transition to a plant-based starting material decouples production from marine ecosystems, ensuring a reliable pharmaceutical intermediates supplier can maintain continuous inventory. The route is designed for high atom economy and operational simplicity, avoiding exotic reagents that would complicate regulatory filings.
Mechanistic Insights into Reductive Ring Opening and Stereoselective Epoxidation
A critical mechanistic feature of this synthesis is the reductive cleavage of the spiroketal F-ring, which transforms the spirostane skeleton into the cholestane side chain found in the target molecule. In the presence of zinc amalgam and concentrated hydrochloric acid under reflux, the acid-catalyzed hydrolysis of the spiroketal generates an oxocarbenium ion intermediate. The zinc acts as a reducing agent, facilitating the removal of the oxygen atom at the C-22 position (originally part of the spiro center) via a radical or hydride-like mechanism, ultimately yielding the 16β-hydroxyl group. This step is crucial because it establishes the correct carbon count and oxidation state for the side chain while preserving the stereochemical integrity of the D-ring junction. The reaction conditions are optimized to prevent over-reduction of the steroid nucleus, ensuring that the A/B ring fusion remains intact as 5α.
The final installation of the 2β,3α-diol moiety relies on a highly stereoselective epoxidation followed by acid-catalyzed hydrolysis. The patent describes two oxidation systems: one utilizing m-chloroperoxybenzoic acid (mCPBA) and another using potassium permanganate with copper sulfate. In both cases, the oxidant approaches the Δ2-double bond from the less hindered α-face of the steroid nucleus, driven by the angular methyl groups at C-10 and C-13 which shield the β-face. This facial selectivity results in the formation of the 2α,3α-epoxide intermediate. Subsequent acid-catalyzed ring opening proceeds via an SN2-like mechanism where water attacks the more substituted carbon or follows the trajectory dictated by the half-chair conformation of the A-ring, leading to the trans-diaxial arrangement of the resulting hydroxyl groups. This precise control over stereochemistry is vital for maintaining the biological activity profile of the marine natural product mimic.
How to Synthesize (25R)-5α-cholesta-2β,3α,26-triol Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing reproducible conditions and straightforward workup procedures. The process begins with the activation of tigogenin via tosylation, followed by elimination to generate the requisite double bond. The core transformation involves the zinc-mediated reductive opening, which requires careful control of acid addition rates to manage exotherms. Following the establishment of the side chain, orthogonal protection strategies are employed to differentiate the C-16 and C-26 hydroxyls, allowing for the selective removal of the C-16 oxygen. The detailed standardized synthesis steps below outline the specific molar ratios, solvent choices, and reaction times required to achieve the high yields reported in the examples, ensuring that high-purity pharmaceutical intermediates can be consistently manufactured.
- Tosylation of tigogenin followed by elimination to form the spirost-2-ene intermediate.
- Reductive ring opening of the spiroketal system using zinc amalgam and hydrochloric acid to yield the cholest-2-ene-diol.
- Selective protection of the C-26 hydroxyl group with TBDMS chloride, followed by mesylation of the C-16 hydroxyl.
- Reduction of the C-16 mesylate with lithium aluminum hydride to remove the oxygen functionality at position 16.
- Epoxidation of the C2-C3 double bond using mCPBA or KMnO4/CuSO4 system to form the 2α,3α-epoxide.
- Acidic hydrolysis of the epoxide and simultaneous deprotection of the silyl ether to yield the final triol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from marine extraction to tigogenin-based semi-synthesis represents a fundamental improvement in supply security and cost structure. The primary driver of cost reduction in pharmaceutical intermediates manufacturing here is the replacement of an expensive, scarce natural resource with a commodity chemical. Tigogenin is produced in large volumes globally for the synthesis of other steroid hormones, meaning its price is stable and its availability is not subject to the vagaries of ocean harvesting. This stability allows for long-term contracting and accurate budget forecasting, eliminating the price volatility associated with niche natural products. Furthermore, the synthetic route avoids the use of precious metal catalysts or cryogenic conditions, relying instead on common industrial reagents like zinc, lithium aluminum hydride, and silyl chlorides, which simplifies the sourcing of raw materials.
- Cost Reduction in Manufacturing: The elimination of complex marine extraction and purification processes significantly lowers the operational expenditure per kilogram of product. By utilizing a linear synthetic sequence with high-yielding steps, the overall material throughput is maximized, reducing waste disposal costs. The use of standard solvents like dichloromethane, ethanol, and pyridine facilitates solvent recovery and recycling, further enhancing the economic efficiency of the process. Additionally, the avoidance of chromatographic purifications in favor of crystallization or simple extractions in several steps reduces the consumption of silica gel and eluents, contributing to substantial cost savings in consumables.
- Enhanced Supply Chain Reliability: Sourcing from a plant-based supply chain mitigates the geopolitical and environmental risks associated with marine bioprospecting. Since tigogenin is derived from terrestrial plants, the supply is less susceptible to weather events, fishing quotas, or marine protected area regulations. This reliability ensures reducing lead time for high-purity pharmaceutical intermediates because manufacturers can maintain safety stock of the starting material without fear of spoilage or scarcity. The robustness of the chemical steps also means that production schedules are predictable, allowing downstream drug developers to plan their clinical trials with confidence in material availability.
- Scalability and Environmental Compliance: The reaction conditions described, such as refluxing in ethanol or stirring at room temperature, are inherently scalable from gram to tonnage levels without requiring specialized high-pressure equipment. The waste streams generated, primarily containing zinc salts and organic solvents, are well-characterized and can be treated using standard effluent treatment protocols common in fine chemical facilities. This ease of waste management supports commercial scale-up of complex pharmaceutical intermediates while adhering to increasingly stringent environmental regulations. The process design prioritizes safety by avoiding highly unstable intermediates, making it suitable for implementation in standard multipurpose chemical plants.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this marine steroid analog. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy for R&D and procurement teams evaluating this technology. Understanding these details is crucial for assessing the feasibility of incorporating this intermediate into your drug discovery pipeline.
Q: Why is synthetic production necessary for (25R)-5α-cholesta-2β,3α,26-triol?
A: Natural extraction from marine organisms like Ophiarachna incrassata yields extremely low quantities, making it insufficient for pharmacological research or commercial development. Synthetic routes provide a sustainable and scalable alternative.
Q: What are the key biological activities associated with this steroid structure?
A: This polyhydroxy marine steroid exhibits potential antitumor, antiviral, hypolipidemic, and neuroprotective activities, making it a valuable lead compound for drug discovery programs targeting these indications.
Q: Is the starting material tigogenin readily available for large-scale production?
A: Yes, tigogenin is a commercially available sapogenin derived from plant sources, offering a significant cost advantage and supply chain stability compared to rare marine extraction sources.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (25R)-5α-cholesta-2β,3α,26-triol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of accessible marine steroid scaffolds in modern drug discovery. Our team of process chemists has extensively analyzed the route disclosed in CN101220073A and is fully prepared to execute this synthesis with the highest standards of quality and efficiency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need grams for screening or kilograms for toxicology, we can deliver. Our facilities are equipped with rigorous QC labs capable of verifying the complex stereochemistry of the product using advanced NMR and HPLC techniques, guaranteeing stringent purity specifications that meet global regulatory requirements.
We invite pharmaceutical partners to leverage our expertise to accelerate their marine drug programs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, optimizing the balance between batch size and unit cost. We encourage you to contact our technical procurement team today to request specific COA data from our pilot batches and discuss route feasibility assessments for your specific derivative needs. Let us handle the complexity of synthesis so you can focus on unlocking the therapeutic potential of this unique bioactive molecule.
