Advanced Manufacturing of Arylsulfonic Anhydrides: A Scalable Commercial Solution
Advanced Manufacturing of Arylsulfonic Anhydrides: A Scalable Commercial Solution
The chemical industry continuously seeks robust methodologies for synthesizing critical intermediates that balance high purity with operational safety and economic viability. Patent CN1847221A introduces a transformative process for the preparation of optionally substituted arylsulfonic anhydrides, specifically addressing the longstanding challenges associated with traditional synthetic routes. This technology leverages the reaction of arylsulfonic acids with thionyl chloride in aliphatic hydrocarbon solvents, a strategy that remarkably overcomes solubility issues while delivering yields sufficient for industrial application. For R&D directors and procurement specialists alike, this represents a pivotal shift away from hazardous reagents toward a more sustainable and cost-effective manufacturing paradigm. The ability to produce high-purity p-toluenesulfonic anhydride without relying on carcinogenic solvents like benzene or expensive metal salts positions this method as a cornerstone for modern fine chemical production. As we delve into the technical specifics, it becomes clear that this innovation not only enhances product quality but also streamlines the supply chain for downstream applications in pharmaceuticals and agrochemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of arylsulfonic anhydrides has been plagued by significant technical and safety hurdles that hindered large-scale adoption. Prior art methods frequently relied on highly toxic and carcinogenic solvents such as benzene, posing severe health risks to operators and creating complex waste disposal challenges for manufacturing facilities. Furthermore, many established routes necessitated the use of prohibitively expensive reagents, including silver p-toluenesulfonate, phenyl cyanate, or mercury oxide, which drastically inflated production costs and introduced supply chain vulnerabilities. For instance, reactions involving di-p-tolyldisulfide with dinitrogen tetroxide or the use of phosphorus pentoxide often resulted in complicated work-up procedures and the formation of difficult-to-remove polyphosphate ester byproducts. These legacy processes were not only environmentally unsustainable but also suffered from inconsistent yields and lengthy reaction times, sometimes extending over several days, which severely impacted production throughput. The reliance on moisture-sensitive or unstable intermediates further compounded these issues, making reproducible commercial scale-up nearly impossible for many organizations seeking a reliable arylsulfonic anhydride supplier.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel approach detailed in the patent utilizes a straightforward condensation reaction between arylsulfonic acids and thionyl chloride within a safe aliphatic hydrocarbon medium. This method surprisingly achieves high conversion rates despite the typically poor solubility of arylsulfonic acids in non-polar solvents, a phenomenon that defies conventional solubility expectations. By employing solvents such as cyclohexane, methylcyclohexane, or petroleum ether, the process eliminates the need for hazardous aromatic solvents while facilitating easy product isolation through precipitation upon cooling. The reaction conditions are mild yet effective, typically operating between 40°C and 80°C, which reduces energy consumption compared to high-temperature alternatives. Moreover, the use of thionyl chloride serves a dual purpose as both a dehydrating agent and a reactant, with the excess reagent easily recoverable via distillation for recycling, thereby enhancing the overall atom economy. This streamlined workflow significantly simplifies the purification process, allowing for the direct isolation of high-purity products suitable for sensitive applications in API synthesis without the need for extensive chromatographic separation.
Mechanistic Insights into Thionyl Chloride-Mediated Dehydration
The core of this innovative synthesis lies in the efficient activation of the sulfonic acid moiety by thionyl chloride, leading to the formation of the anhydride linkage through a dehydration mechanism. Initially, the arylsulfonic acid reacts with thionyl chloride to form a reactive sulfonyl chloride intermediate in situ, accompanied by the evolution of sulfur dioxide and hydrogen chloride gases. In the presence of a second equivalent of the sulfonic acid or its salt form, this activated species undergoes nucleophilic attack, resulting in the elimination of another molecule of HCl and the formation of the symmetric arylsulfonic anhydride bond. The choice of aliphatic hydrocarbon solvents plays a critical mechanistic role; while they do not solubilize the starting acid well at room temperature, heating the suspension creates a dynamic heterogeneous system where the reaction occurs at the interface or in the limited soluble fraction, driving the equilibrium forward as the product precipitates or remains suspended. This precipitation effect effectively pulls the reaction equilibrium towards the product side, preventing reverse hydrolysis and ensuring high conversion rates even with simple stoichiometry.
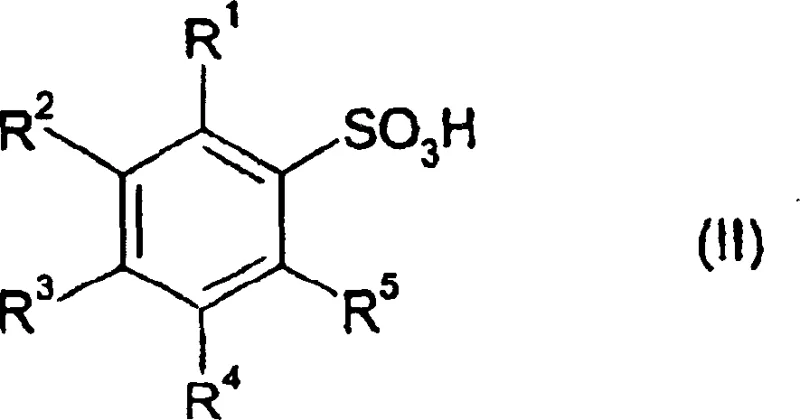
Controlling impurities in this system is inherently managed by the volatility of the byproducts and the solubility differences between the reactants and the final anhydride. Since the primary byproducts are gaseous (SO2 and HCl) or volatile (excess SOCl2), they are readily removed during the distillation and stirring phases, leaving behind a chemically clean solid residue. The patent specifies that substituents on the aromatic ring, such as alkyl, alkoxy, or halogen groups, are well-tolerated under these conditions, indicating a broad substrate scope that is vital for generating diverse libraries of fine chemical intermediates. The absence of transition metal catalysts means there is no risk of heavy metal contamination, a critical quality attribute for pharmaceutical intermediates that must meet stringent regulatory limits. This mechanistic simplicity translates directly into operational robustness, allowing for precise control over the reaction endpoint and minimizing the formation of side products that often plague more complex catalytic cycles.
How to Synthesize p-Toluenesulfonic Anhydride Efficiently
Implementing this synthesis route requires careful attention to thermal management and addition rates to ensure safe gas evolution and optimal crystal formation. The process begins by suspending the arylsulfonic acid, such as p-toluenesulfonic acid monohydrate, in a selected aliphatic solvent like methylcyclohexane, followed by heating to initiate the reaction environment. Thionyl chloride is then introduced, typically in a molar excess of 2 to 3 equivalents, to drive the dehydration to completion while compensating for any losses due to volatility. The detailed standardized synthesis steps below outline the precise parameters for temperature control, distillation, and isolation that guarantee reproducible results on a commercial scale.
- Suspend the arylsulfonic acid starting material in an aliphatic hydrocarbon solvent such as methylcyclohexane or cyclohexane and heat the mixture to between 40°C and 80°C.
- Add thionyl chloride dropwise in excess (2 to 3 equivalents) while maintaining the temperature, observing slow gas evolution.
- Distill off excess thionyl chloride under reduced or ambient pressure, then cool the residue to precipitate the product for filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers profound strategic advantages that extend far beyond simple chemical transformation. By shifting away from precious metal catalysts and carcinogenic solvents, manufacturers can achieve substantial cost savings in pharmaceutical intermediates manufacturing through reduced raw material expenditure and simplified waste treatment protocols. The elimination of expensive reagents like silver salts or phenyl cyanate removes a significant variable from the cost structure, stabilizing pricing models and protecting margins against volatile commodity markets. Furthermore, the use of common, bulk-available solvents such as cyclohexane ensures a resilient supply chain that is less susceptible to geopolitical disruptions or specialized vendor shortages. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global multinational clients.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of high-cost reagents with commodity chemicals and the recyclability of the thionyl chloride. Since the excess thionyl chloride can be distilled off and reused in subsequent batches, the effective consumption of this reagent is minimized, leading to a drastic reduction in material costs per kilogram of product. Additionally, the avoidance of complex purification steps like column chromatography or extensive recrystallization from hazardous solvent mixtures reduces labor hours and utility consumption. The simplified downstream processing also lowers the capital expenditure required for specialized containment equipment needed for toxic substances, further enhancing the overall return on investment for production facilities.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is significantly more straightforward compared to legacy methods, as aliphatic hydrocarbons and thionyl chloride are produced at massive scales globally. This abundance ensures that production is never halted due to a lack of niche reagents, providing a stable foundation for long-term supply agreements. The robustness of the chemistry also means that batch-to-batch variability is minimized, reducing the risk of out-of-specification results that could delay shipments. For supply chain planners, this predictability allows for more accurate inventory management and the ability to respond rapidly to fluctuations in market demand without compromising on quality or lead times.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns perfectly with modern green chemistry principles by eliminating benzene and heavy metals from the workflow. The reduction in hazardous waste generation simplifies compliance with increasingly strict environmental regulations, lowering the administrative burden and potential liability for the manufacturer. The scalability is proven by the ability to handle the exothermic nature of the reaction through controlled addition and the ease of isolating the solid product via filtration, which is a unit operation that scales linearly from pilot plant to multi-ton production. This makes the commercial scale-up of complex fine chemical intermediates not only feasible but also economically attractive for large-volume contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this arylsulfonic anhydride synthesis technology. These insights are derived directly from the patent specifications and are designed to clarify the operational benefits and safety profile for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: Why are aliphatic hydrocarbons preferred over benzene for this synthesis?
A: Aliphatic hydrocarbons like cyclohexane eliminate the use of carcinogenic benzene, significantly improving operator safety and environmental compliance while maintaining effective solubility profiles for the reaction.
Q: How does this process improve supply chain reliability compared to traditional methods?
A: By avoiding expensive and scarce reagents like silver salts or phenyl cyanate, and utilizing common industrial solvents, the process ensures consistent raw material availability and reduces procurement risks.
Q: Is the excess thionyl chloride recoverable in this process?
A: Yes, the process includes a distillation step where excess thionyl chloride can be recovered and recycled back into the reaction, minimizing waste and lowering overall material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Toluenesulfonic Anhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer and more efficient synthetic routes is critical for maintaining competitiveness in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN1847221A are fully realized in practical, high-volume manufacturing. We are committed to delivering high-purity p-toluenesulfonic anhydride that meets stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our infrastructure is designed to handle the specific requirements of this chemistry, including solvent recovery systems and corrosion-resistant reactors, guaranteeing a consistent and reliable supply for your critical applications.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic impact of switching to this greener synthesis route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring that your supply chain is optimized for both performance and profitability.
