Scalable Synthesis of 5-(Trifluoromethoxy)benzimidazole-2-carboxylic Acid for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable pathways for constructing complex heterocyclic scaffolds, particularly benzimidazole derivatives which serve as critical building blocks for a wide array of therapeutic agents. Patent CN111995581A discloses a highly efficient synthetic methodology for preparing 5-(trifluoromethoxy)-1H-benzo[d]imidazole-2-carboxylic acid, a valuable intermediate with potential applications in anticancer and antifungal drug development. This novel approach addresses the longstanding challenges associated with traditional benzimidazole synthesis by utilizing a streamlined three-step sequence that begins with a readily available nitro-aniline precursor. The process leverages inexpensive reagents such as iron powder and potassium permanganate, circumventing the need for costly transition metal catalysts often required in similar transformations. By optimizing reaction parameters including temperature, solvent ratios, and reagent stoichiometry, the inventors have achieved a route that balances high chemical yield with operational simplicity. For R&D directors and process chemists, this patent represents a significant advancement in the accessible chemistry of fluorinated benzimidazoles, offering a reliable blueprint for generating high-purity materials essential for preclinical and clinical supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functionalized benzimidazole-2-carboxylic acids has often relied on methodologies that present substantial hurdles for large-scale manufacturing. Conventional routes frequently employ catalytic hydrogenation using palladium or platinum on carbon, which necessitates specialized high-pressure reactor infrastructure and rigorous safety protocols to manage hydrogen gas hazards. Furthermore, these precious metal catalysts are not only expensive but also pose significant challenges regarding residual metal removal, a critical quality attribute for pharmaceutical intermediates intended for human use. Alternative oxidative methods might utilize harsh reagents that lack chemoselectivity, potentially leading to the degradation of sensitive functional groups such as the trifluoromethoxy moiety or the formation of difficult-to-remove byproducts. The cumulative effect of these limitations is a manufacturing process characterized by high capital expenditure, complex purification workflows, and variable batch-to-batch consistency. Such inefficiencies directly impact the cost of goods sold (COGS) and can introduce bottlenecks in the supply chain, delaying the availability of key intermediates for downstream drug substance production.
The Novel Approach
In stark contrast to these traditional constraints, the method disclosed in CN111995581A introduces a pragmatic and economically superior strategy centered on earth-abundant reagents and mild reaction conditions. The core innovation lies in the substitution of noble metal catalysts with iron powder for the critical reduction step, coupled with a straightforward condensation and oxidation sequence. This approach eliminates the requirement for high-pressure hydrogenation equipment, thereby drastically reducing the safety risks and capital investment associated with the manufacturing facility. The use of glycolic acid as a cyclization partner allows for the direct construction of the imidazole ring with a handle for subsequent oxidation, streamlining the overall synthetic logic. Moreover, the process demonstrates excellent tolerance for the trifluoromethoxy group, ensuring that the valuable fluorine content is retained throughout the synthesis without defluorination side reactions. By prioritizing reagent availability and operational ease, this novel route provides a compelling solution for cost reduction in pharmaceutical intermediate manufacturing, enabling producers to deliver high-quality materials with greater speed and reliability.
Mechanistic Insights into Iron-Mediated Reduction and Permanganate Oxidation
The first stage of this synthesis involves the chemoselective reduction of the nitro group in Compound A to an amine, yielding the diamine intermediate Compound B. This transformation is mediated by iron powder in the presence of ammonium chloride, acting as a proton source and electrolyte in a mixed solvent system of methanol, tetrahydrofuran, and water. The mechanism proceeds through a series of electron transfers from the metallic iron surface to the nitro group, sequentially reducing it through nitroso and hydroxylamine intermediates before arriving at the final amine. The specific solvent ratio of 1:2:1 (MeOH:THF:Water) is crucial for solubilizing both the organic substrate and the inorganic salts while maintaining the reactivity of the iron surface. Operating at a moderate temperature range of 60-65°C ensures that the reaction kinetics are favorable without promoting thermal decomposition or over-reduction of other sensitive sites. This step is pivotal as it generates the requisite 1,2-diamine motif needed for the subsequent ring closure, setting the foundation for the benzimidazole core structure with high fidelity.

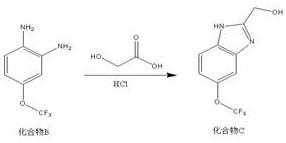
Following the reduction, the diamine Compound B undergoes a condensation reaction with glycolic acid in the presence of hydrochloric acid to form the benzimidazole ring, resulting in Compound C. This cyclization is driven by the nucleophilic attack of the amine nitrogens on the carbonyl and hydroxyl functionalities of the glycolic acid, followed by dehydration to establish the aromatic imidazole system. The acidic environment provided by HCl serves to activate the carboxylic acid of the glycolic acid and protonate the intermediates, facilitating the elimination of water and driving the equilibrium towards the product. The reaction is conducted at elevated temperatures between 98-102°C, which provides the necessary activation energy to overcome the barrier for ring closure and aromatization. The result is the formation of a hydroxymethyl-substituted benzimidazole, a strategic intermediate that positions a primary alcohol at the 2-position of the heterocycle, perfectly poised for the final oxidative conversion to the target carboxylic acid.
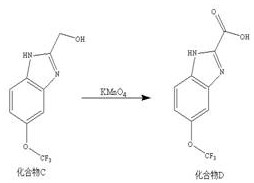
The final transformation involves the oxidation of the hydroxymethyl group on Compound C to the corresponding carboxylic acid, yielding the target Compound D. This step utilizes potassium permanganate (KMnO4) in an alkaline medium provided by sodium hydroxide solution. The mechanism entails the initial oxidation of the primary alcohol to an aldehyde, which is rapidly further oxidized to the carboxylate salt under the strongly oxidizing conditions. The use of a dilute sodium hydroxide solution (1.2% mass fraction) helps to solubilize the intermediate species and prevents the precipitation of manganese dioxide from interfering excessively with the reaction mixture, although filtration is still required post-reaction. Heating the mixture to 100-103°C ensures complete conversion of the alcohol to the acid, minimizing the presence of partially oxidized aldehyde impurities. Subsequent acidification of the reaction mixture precipitates the final product as a white solid, allowing for easy isolation and purification. This robust oxidation protocol ensures that the final API intermediate meets stringent purity specifications required for pharmaceutical applications.
How to Synthesize 5-(Trifluoromethoxy)benzimidazole-2-carboxylic Acid Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and thermal profiles as outlined in the patent examples. The process begins with the careful preparation of the reduction mixture, ensuring that the iron powder is added in batches to control the exotherm and maintain consistent reaction rates. Following the isolation of the diamine, the cyclization step requires strict temperature control near the boiling point of the aqueous acid mixture to drive the condensation to completion. Finally, the oxidation step demands thorough mixing and hot filtration techniques to manage the manganese byproducts effectively. While the general chemistry is straightforward, attention to detail in the workup procedures—such as pH adjustments and washing steps—is essential to maximize yield and purity. For a comprehensive guide on the specific operational parameters and safety considerations, please refer to the detailed synthesis instructions below.
- Reduction of the nitro precursor (Compound A) using iron powder and ammonium chloride in a methanol/THF/water solvent system at 60-65°C to yield the diamine intermediate (Compound B).
- Cyclization of the diamine intermediate with glycolic acid in hydrochloric acid at 98-102°C to form the benzimidazole alcohol (Compound C).
- Oxidation of the hydroxymethyl group on Compound C using potassium permanganate in sodium hydroxide solution at 100-103°C to afford the final carboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers transformative benefits that extend far beyond simple chemical yield improvements. The strategic selection of reagents fundamentally alters the cost structure of the manufacturing process, shifting away from reliance on volatile precious metal markets towards stable, commodity-grade chemicals. This shift not only stabilizes raw material costs but also mitigates the risk of supply disruptions that can occur with specialized catalysts. Furthermore, the simplified equipment requirements mean that the process can be executed in a wider range of manufacturing facilities, increasing the pool of potential contract manufacturing organizations (CMOs) and enhancing supply chain resilience. The ability to produce high-purity intermediates with fewer purification steps translates directly into reduced processing time and lower utility consumption, contributing to a more sustainable and cost-effective production model.
- Cost Reduction in Manufacturing: The replacement of expensive palladium or platinum catalysts with iron powder represents a direct and substantial reduction in raw material costs. Iron is one of the most abundant and inexpensive metals available, and its use eliminates the need for complex catalyst recovery systems or extensive testing for heavy metal residues. Additionally, the avoidance of high-pressure hydrogenation equipment reduces both the capital expenditure for new plants and the maintenance costs for existing infrastructure. The overall process efficiency, characterized by high yields in each step, minimizes the loss of valuable starting materials, further driving down the effective cost per kilogram of the final product. These factors combine to create a highly competitive cost profile for the manufacture of this benzimidazole derivative.
- Enhanced Supply Chain Reliability: The reliance on globally available commodity chemicals such as glycolic acid, hydrochloric acid, and potassium permanganate ensures a robust and uninterrupted supply of reagents. Unlike specialized ligands or catalysts that may have single-source suppliers or long lead times, these materials can be sourced from multiple vendors worldwide, reducing the risk of bottlenecks. The moderate reaction conditions also imply that the process is less susceptible to delays caused by equipment availability or specialized safety certifications. This reliability allows supply chain managers to forecast production timelines with greater confidence, ensuring that downstream drug development programs remain on schedule without the threat of intermediate shortages.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard unit operations like stirring, heating, filtration, and crystallization that are easily transferred from the laboratory to pilot and commercial scales. The waste streams generated, primarily consisting of iron salts and manganese dioxide, are well-understood and can be managed through established waste treatment protocols, facilitating environmental compliance. The absence of toxic organic solvents in large quantities or hazardous gases simplifies the permitting process for manufacturing sites. This ease of scale-up means that production volumes can be rapidly increased to meet market demand, from 100 kgs to 100 MT/annual commercial production, without the need for significant process re-engineering or regulatory re-validation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 5-(trifluoromethoxy)-1H-benzo[d]imidazole-2-carboxylic acid. These answers are derived directly from the technical disclosures and experimental data provided in the patent literature, offering clarity on the feasibility and advantages of this specific manufacturing route. Understanding these details is crucial for stakeholders evaluating the potential integration of this intermediate into their broader supply chains or drug discovery pipelines.
Q: What are the key advantages of using iron powder for the reduction step in this synthesis?
A: The use of iron powder and ammonium chloride offers a cost-effective and chemoselective alternative to catalytic hydrogenation. It operates under mild thermal conditions (60-65°C) and avoids the need for high-pressure equipment or expensive precious metal catalysts, significantly simplifying the process safety profile and reducing capital expenditure for manufacturing.
Q: How does this method ensure high purity of the final benzimidazole derivative?
A: The process utilizes distinct workup procedures for each step, including diatomite filtration to remove metal residues and precise pH adjustments to isolate products. Example 1 in the patent demonstrates that these controls allow for the isolation of the final acid with a purity of 99.2%, minimizing the burden on downstream purification processes.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the route is specifically designed for industrial scalability. It employs readily available commodity chemicals like iron powder, glycolic acid, and potassium permanganate. The reaction conditions are moderate, and the workups involve standard unit operations like filtration and crystallization, making it highly adaptable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(Trifluoromethoxy)benzimidazole-2-carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of novel therapeutics. Our team of expert process chemists has thoroughly analyzed the synthetic route disclosed in CN111995581A and is fully prepared to execute this chemistry with precision and efficiency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-(trifluoromethoxy)-1H-benzo[d]imidazole-2-carboxylic acid we deliver adheres to the highest industry standards. We are committed to being a transparent and responsive partner in your supply chain.
We invite you to engage with our technical procurement team to discuss how we can support your specific requirements. Whether you need a Customized Cost-Saving Analysis for your current sourcing strategy or require specific COA data and route feasibility assessments for this compound, we are ready to assist. By leveraging our manufacturing capabilities and technical expertise, we can help you secure a stable supply of this vital intermediate, allowing you to focus on advancing your drug development programs with confidence.
