Advanced Triphosgene-Mediated Synthesis of 4,4'-Dinitrodiphenylurea for Commercial Scale-Up
The chemical landscape for synthesizing critical urea derivatives has evolved significantly with the introduction of safer carbonyl sources, as detailed in patent CN1286804C. This specific intellectual property outlines a robust methodology for preparing 4,4'-dinitrodiphenylurea, a pivotal intermediate utilized extensively in the manufacture of veterinary pharmaceuticals such as Nicarbazin, as well as in various dye and medicinal applications. The core innovation lies in substituting hazardous gaseous reagents with bistrichloromethyl carbonate, commonly known as triphosgene, which acts as a solid, manageable equivalent of phosgene. By leveraging ethyl acetate or butyl acetate as solvents and employing pyridine or triethylamine as acid-binding agents, this process achieves a remarkable balance between operational safety and chemical efficiency. For R&D directors and procurement specialists, understanding this shift is crucial, as it represents a transition from high-risk batch processes to streamlined, scalable operations that minimize regulatory burdens associated with toxic gas handling while maintaining rigorous purity standards required for fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 4,4'-dinitrodiphenylurea relied on several archaic and technically demanding pathways that posed significant barriers to efficient commercial manufacturing. One such method involved the direct rotary melting of free p-nitroaniline with urea, a process necessitating extreme temperatures reaching 300°C, which imposed severe stress on reactor materials and resulted in substantial energy consumption. Another conventional route utilized p-nitroaniline hydrochloride reacting with urea in high-boiling solvents like dibutyl phthalate; while this achieved reasonable yields, the post-processing was notoriously cumbersome due to the difficulty of separating the product from the high-boiling solvent matrix. Furthermore, the nitration of diphenylurea presented a lengthy multi-step workflow with low overall yields and significant challenges in controlling regioselectivity, often leading to unwanted ortho-nitrated impurities that compromised product quality. Perhaps most critically, the traditional reaction of p-nitroaniline with gaseous phosgene, while chemically effective, introduced unacceptable safety risks due to the extreme toxicity of phosgene gas, requiring specialized containment infrastructure and rigorous emergency protocols that inflated capital expenditure.
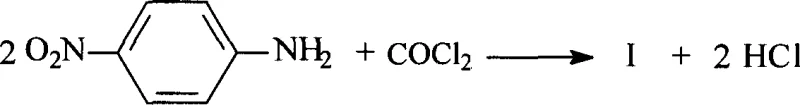
The Novel Approach
In stark contrast to these legacy techniques, the novel approach described in the patent utilizes triphosgene to effect the urea linkage formation under mild and controllable conditions. This method operates at significantly lower temperatures, typically initiating addition at 40°C and proceeding to solvent reflux, which drastically reduces energy requirements and thermal stress on the equipment. The use of common ester solvents such as ethyl acetate or butyl acetate simplifies the workup procedure, allowing for easy removal and recovery compared to the high-boiling solvents of the past. By generating the reactive carbonyl species in situ from a stable solid precursor, the process eliminates the need for complex gas dosing systems and mitigates the risk of accidental toxic release. This technological leap not only enhances the safety profile of the manufacturing site but also streamlines the operational workflow, enabling faster batch turnover and reducing the complexity of waste treatment systems, thereby offering a compelling value proposition for supply chain optimization.
Mechanistic Insights into Triphosgene-Mediated Urea Formation
The mechanistic elegance of this synthesis relies on the controlled thermal decomposition of triphosgene to generate phosgene equivalents in the presence of the nucleophilic amine. Upon heating or interaction with the amine base, one molecule of bistrichloromethyl carbonate decomposes to release three molar equivalents of phosgene, which then reacts rapidly with p-nitroaniline to form the corresponding isocyanate intermediate or directly couples to form the urea linkage. The presence of an acid-binding agent like triethylamine or pyridine is critical in this mechanism, as it scavenges the hydrogen chloride byproduct generated during the reaction, driving the equilibrium forward and preventing the protonation of the amine which would deactivate its nucleophilicity. This in situ generation ensures that the concentration of free phosgene remains low at any given moment, minimizing side reactions such as the formation of biurets or allophanates that can occur with excess reagent. The reaction pathway is highly selective for the para-position due to the pre-existing substitution pattern of the starting material, effectively bypassing the regioselectivity issues seen in direct nitration routes.
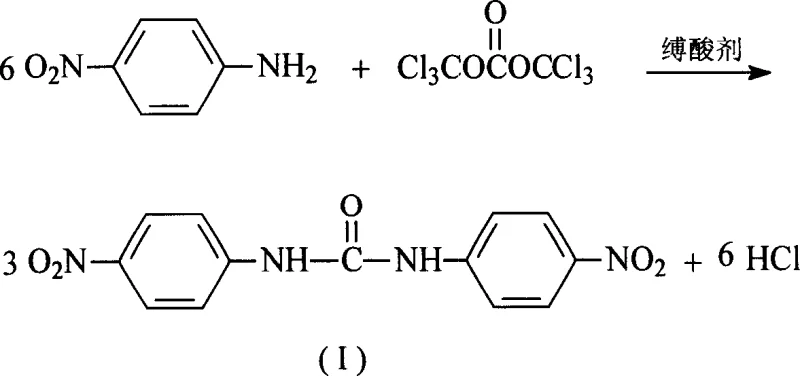
Impurity control in this system is inherently superior due to the mild reaction conditions and the specificity of the acylation mechanism. Unlike the nitration of diphenylurea, where electrophilic aromatic substitution can lead to mixtures of ortho and para isomers requiring difficult chromatographic or crystalline separations, this condensation reaction preserves the aromatic substitution pattern of the starting p-nitroaniline. The primary impurities are typically unreacted starting materials or minor hydrolysis products, both of which are easily removed during the aqueous workup and subsequent washing with ethanol. The patent data highlights melting points in the range of 304-308°C, indicative of a highly crystalline and pure lattice structure, which is essential for downstream coupling reactions in drug synthesis. This high level of chemical fidelity ensures that the intermediate meets the stringent specifications required by regulatory bodies for veterinary and pharmaceutical applications, reducing the risk of batch rejection.
How to Synthesize 4,4'-Dinitrodiphenylurea Efficiently
The practical execution of this synthesis involves a straightforward sequence of dissolution, controlled addition, and thermal maturation that can be readily adapted for pilot or plant-scale reactors. The process begins with the preparation of two distinct solutions: the carbonyl source dissolved in the ester solvent and the amine mixed with the acid binder in the same or compatible solvent medium. Maintaining the addition temperature around 40°C is vital to manage the exotherm associated with the initial reaction phases, after which the mixture is heated to reflux to ensure complete conversion of the starting materials. Following the reaction period, the addition of water facilitates the hydrolysis of any remaining acid chlorides and assists in the precipitation or phase separation of the product. Detailed standardized operating procedures regarding stoichiometry, agitation rates, and specific crystallization parameters are essential for reproducibility and are outlined in the technical guide below.
- Dissolve bistrichloromethyl carbonate in ethyl acetate or butyl acetate and prepare a separate solution of p-nitroaniline with an acid-binding agent like triethylamine.
- Add the triphosgene solution dropwise to the amine mixture at controlled temperatures around 40°C to manage exothermicity and gas evolution.
- Reflux the reaction mixture to ensure completion, followed by aqueous workup and recrystallization to isolate the high-purity yellow solid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this triphosgene-based methodology offers profound advantages in terms of cost structure and supply chain resilience. The substitution of gaseous phosgene with a solid reagent fundamentally alters the logistics profile, removing the need for specialized cylinder transport and on-site gas containment infrastructure, which translates to significant reductions in capital investment and insurance costs. Furthermore, the use of commodity solvents like ethyl acetate ensures that raw material sourcing is not bottlenecked by niche chemical availability, allowing procurement managers to leverage competitive market pricing and maintain robust inventory levels without supply disruption risks. The simplified equipment requirements, needing only standard glass-lined or stainless steel reactors capable of stirring and reflux rather than high-pressure or high-temperature vessels, further lower the barrier to entry for contract manufacturing organizations.
- Cost Reduction in Manufacturing: The elimination of toxic gas handling systems and the reduction in energy consumption due to lower operating temperatures contribute to a leaner cost base. By avoiding the complex scrubbing systems required for gaseous phosgene vents and the high-energy inputs needed for 300°C melt processes, the overall utility and maintenance overheads are substantially decreased. Additionally, the high yields reported in the patent data, consistently exceeding 84%, minimize raw material waste and maximize the throughput per batch, enhancing the economic efficiency of the production line.
- Enhanced Supply Chain Reliability: Utilizing stable solid reagents and common solvents mitigates the risk of supply interruptions caused by regulatory restrictions on hazardous gas transport or the scarcity of specialized high-boiling solvents. This stability allows for more predictable production scheduling and shorter lead times, as the raw materials are widely available from multiple global suppliers. The robustness of the process against minor variations in conditions also ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by out-of-specification results.
- Scalability and Environmental Compliance: The process generates fewer hazardous byproducts and avoids the use of persistent organic pollutants often associated with older nitration or high-boiling solvent methods. The aqueous workup and ethanol washing steps produce waste streams that are easier to treat and dispose of in compliance with modern environmental regulations. This environmental compatibility facilitates smoother permitting processes for capacity expansion and aligns with the sustainability goals of major pharmaceutical clients who prioritize green chemistry principles in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent specifications and process characteristics. Understanding these details is vital for technical teams evaluating the feasibility of integrating this intermediate into their broader manufacturing portfolios. The answers reflect the specific advantages of the triphosgene method over historical alternatives, focusing on safety, yield, and operational simplicity.
Q: Why is triphosgene preferred over gaseous phosgene for this synthesis?
A: Triphosgene is a stable solid that releases phosgene in situ, eliminating the severe safety hazards and specialized containment equipment required for handling toxic gaseous phosgene cylinders.
Q: What are the typical yields achieved with this patented process?
A: The patent data indicates consistent yields ranging from 84% to 89%, with product melting points confirming high purity suitable for downstream pharmaceutical applications.
Q: Can this process be scaled for industrial production?
A: Yes, the process utilizes common solvents like ethyl acetate and standard stirring equipment without high-pressure requirements, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Dinitrodiphenylurea Supplier
As the demand for high-quality veterinary and pharmaceutical intermediates continues to grow, partnering with a manufacturer that masters advanced synthesis technologies is paramount for ensuring product integrity and supply continuity. NINGBO INNO PHARMCHEM leverages deep expertise in fine chemical synthesis to deliver 4,4'-dinitrodiphenylurea that meets the rigorous purity standards demanded by the global market. Our facilities are equipped to handle diverse synthetic pathways, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while adhering to stringent purity specifications. Our rigorous QC labs employ state-of-the-art analytical instrumentation to verify every batch, ensuring that the impurity profiles remain well within acceptable limits for downstream drug synthesis.
We invite procurement leaders and technical directors to engage with our team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain insights into the potential economic efficiencies of switching to this safer, more sustainable manufacturing method. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a seamless integration of this critical intermediate into your production schedule.
