Advanced Synthesis of Boc-Taurine: A Scalable Route for High-Purity Pharmaceutical Intermediates
Advanced Synthesis of Boc-Taurine: A Scalable Route for High-Purity Pharmaceutical Intermediates
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more efficient, scalable, and cost-effective synthetic routes for critical building blocks. A significant advancement in this domain is detailed in Chinese Patent CN109824553B, which discloses a robust preparation method for 2-((tert-butoxycarbonyl)amino)ethyl-1-sulfonic acid, commonly known as Boc-Taurine. This compound serves as a pivotal intermediate in the synthesis of various bioactive molecules, particularly where the unique physicochemical properties of taurine must be preserved while rendering the amino group inert for subsequent coupling reactions. The patent highlights a novel approach that utilizes readily available reagents such as sodium bicarbonate and di-tert-butyl dicarbonate (Boc anhydride) in a mixed solvent system of tetrahydrofuran and methanol. This methodology addresses long-standing challenges associated with the solubility and reactivity of taurine, offering a pathway that is not only chemically elegant but also commercially viable for large-scale production.
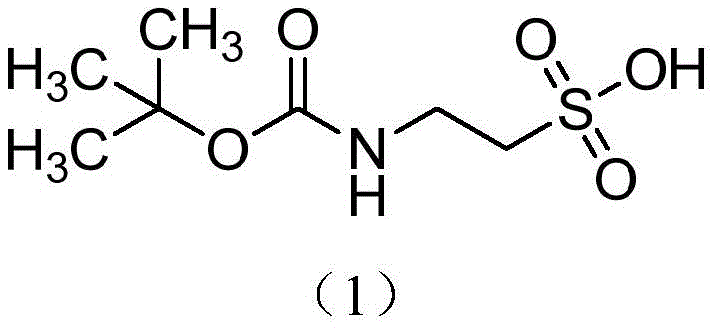
For R&D directors and process chemists, the structural integrity and purity of such intermediates are paramount. The molecule features a stable carbamate linkage that effectively masks the nucleophilic character of the primary amine, preventing unwanted side reactions during downstream synthesis. Furthermore, the presence of the sulfonic acid moiety introduces significant polarity, which traditionally complicates purification. However, the disclosed method ensures that the final product maintains a stable structure, effectively protecting the amino group in taurine. This stability is crucial for facilitating the participation of taurine derivatives in complex multi-step syntheses, such as the production of drugs designed to improve immunity or metabolic regulation. By establishing a reliable protocol for this transformation, the patent provides a foundation for consistent quality in the supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the protection of amino acids containing highly polar functional groups like sulfonic acids has presented substantial technical hurdles for process engineers. Conventional methods often rely on strong organic bases or expensive phase-transfer catalysts to solubilize the zwitterionic starting materials in organic solvents. These traditional approaches frequently suffer from poor atom economy and generate significant amounts of hazardous waste, complicating the environmental compliance profile of the manufacturing process. Moreover, the use of harsh reaction conditions can lead to the degradation of the sensitive sulfonic acid group or result in incomplete protection, necessitating rigorous and costly purification steps such as preparative HPLC. The difficulty in separating the protected product from unreacted starting materials and byproducts often leads to reduced overall yields and extended production cycles. For procurement managers, these inefficiencies translate into higher raw material costs and unpredictable lead times, creating bottlenecks in the supply chain for critical API precursors.
The Novel Approach
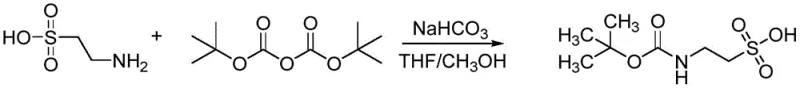
In stark contrast, the methodology described in CN109824553B offers a streamlined solution that circumvents these traditional pitfalls through the strategic selection of mild reagents and optimized solvent systems. By employing sodium bicarbonate as a catalyst and base, the process operates under significantly milder conditions, eliminating the need for aggressive alkalis that could compromise product integrity. The use of a 1:1 volume ratio of methanol and tetrahydrofuran creates a homogeneous reaction medium that effectively dissolves both the polar taurine salt and the organic Boc anhydride, ensuring efficient mass transfer and reaction kinetics. This novel approach not only simplifies the operational procedure but also enhances the ease of product isolation, as the target molecule can be obtained through straightforward rotary evaporation and column chromatography. The result is a process that delivers high yields of the target product with minimal impurity formation, representing a significant leap forward in cost reduction in pharma manufacturing.
Mechanistic Insights into Bicarbonate-Mediated Boc-Protection
The success of this synthesis lies in the delicate balance of nucleophilicity and solubility managed by the reaction conditions. Mechanistically, the reaction proceeds via a nucleophilic attack of the primary amino group of taurine on one of the carbonyl carbons of the di-tert-butyl dicarbonate (Boc anhydride). In the absence of a suitable base, the proton released during this attack would protonate the remaining amine, halting the reaction. Sodium bicarbonate acts as a proton scavenger, neutralizing the generated acid and driving the equilibrium towards the formation of the carbamate product. Crucially, the choice of sodium bicarbonate over stronger bases like sodium hydroxide prevents the hydrolysis of the Boc group, which is susceptible to base-catalyzed cleavage. The mixed solvent system plays an equally vital role; methanol aids in solvating the ionic taurine species, while tetrahydrofuran ensures the Boc anhydride remains in solution, facilitating the collision frequency required for the reaction to proceed efficiently at low temperatures.
From an impurity control perspective, the mild temperature range of -20°C to 20°C specified in the patent is instrumental in suppressing side reactions. At elevated temperatures, Boc anhydride can undergo thermal decomposition or react with the solvent, leading to complex impurity profiles that are difficult to remove. By maintaining the reaction within this controlled thermal window, typically around -4°C to 2°C for optimal results, the process minimizes the formation of di-Boc protected species or urea byproducts. The stoichiometry is also carefully managed, with a mass ratio of taurine to Boc anhydride ranging from 1:1 to 1:10, allowing for flexibility depending on the desired conversion rate. This precise control over reaction parameters ensures that the final crude product is of high purity, reducing the burden on downstream purification units and aligning with the stringent purity specifications required for GMP manufacturing environments.
How to Synthesize 2-((tert-butoxycarbonyl)amino)ethyl-1-sulfonic acid Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the procedural details outlined in the patent to maximize yield and reproducibility. The process begins with the preparation of the solvent system, followed by the sequential addition of reagents under controlled thermal conditions. The key to success lies in the slow addition of the Boc anhydride solution to prevent local exotherms that could degrade the reagents. While the general workflow is straightforward, specific attention must be paid to the stirring efficiency and temperature maintenance throughout the 12 to 36-hour reaction period. For those seeking to replicate this high-efficiency protocol, the detailed standardized synthesis steps are provided in the guide below, ensuring that technical teams can achieve the reported 90-96% yields consistently.
- Prepare a mixed solvent system of methanol and tetrahydrofuran (1: 1 volume ratio) and cool in an ice-water bath.
- Add taurine and sodium bicarbonate to the cooled solvent mixture and stir thoroughly to form a suspension.
- Slowly add a solution of di-tert-butyl dicarbonate (Boc anhydride) in THF dropwise while maintaining temperature between -20°C and 20°C for 12-36 hours.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply chain resilience, the adoption of this synthetic route offers compelling economic and logistical benefits. The shift away from exotic catalysts and harsh reagents towards commodity chemicals fundamentally alters the cost structure of producing this valuable intermediate. By leveraging widely available raw materials, manufacturers can mitigate the risks associated with supply disruptions of specialized reagents, thereby enhancing supply chain reliability. Furthermore, the simplicity of the workup procedure reduces the consumption of energy and solvents, contributing to a more sustainable manufacturing footprint. These factors combined create a robust value proposition for companies looking to optimize their procurement strategies for amino acid derivatives.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of sodium bicarbonate, an inexpensive and abundant base, drastically lowers the raw material costs per kilogram of product. Additionally, the high yield reported in the patent embodiments (ranging from 90% to 96%) means that less starting material is wasted, directly improving the overall process economics. The simplified purification process further reduces operational expenditures by minimizing the need for complex chromatographic separations or extensive recrystallization steps. This cumulative effect results in substantial cost savings that can be passed down the supply chain, making the final API more competitive in the global market.
- Enhanced Supply Chain Reliability: Sourcing taurine and Boc anhydride is straightforward as they are produced on a massive industrial scale for various applications, ensuring a steady and reliable supply of key inputs. Unlike processes dependent on custom-synthesized ligands or rare earth metals, this method is insulated from the volatility of niche chemical markets. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the rejection rate of incoming batches. This stability allows procurement managers to negotiate better long-term contracts and maintain lower safety stock levels, optimizing working capital.
- Scalability and Environmental Compliance: The use of THF and methanol, while requiring careful handling, is well-established in the industry with mature recovery and recycling protocols, facilitating easier scale-up from gram to ton quantities. The absence of heavy metals in the process simplifies the waste treatment stream, as there is no need for expensive heavy metal scavenging or disposal procedures. This aligns perfectly with increasingly stringent environmental regulations, reducing the regulatory burden on the manufacturing site. The ability to run the reaction at near-ambient or mildly cooled temperatures also reduces the energy load on HVAC and chilling systems, contributing to a greener and more scalable operation.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the synthesis and application of Boc-Taurine. These insights are derived directly from the experimental data and technical disclosures within the patent literature, providing a factual basis for decision-making. Understanding the nuances of reaction conditions and purification strategies is essential for successful technology transfer and process validation.
Q: Why is sodium bicarbonate preferred over stronger bases for this Boc-protection?
A: Sodium bicarbonate provides a mild basic environment that effectively neutralizes the tert-butanol byproduct without risking the hydrolysis of the sensitive sulfonic acid group or causing side reactions common with stronger alkalis.
Q: What is the typical yield range for this synthesis method?
A: According to patent data, the process consistently achieves high yields ranging from 90% to 96%, demonstrating excellent reproducibility across different scales and slight variations in temperature.
Q: How does this method improve supply chain reliability for taurine derivatives?
A: By utilizing commodity chemicals like sodium bicarbonate and standard solvents (THF/Methanol) instead of exotic catalysts, the method reduces dependency on specialized supply chains and simplifies procurement logistics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-((tert-butoxycarbonyl)amino)ethyl-1-sulfonic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation therapeutics. Our team of expert chemists has extensively analyzed the patented route for 2-((tert-butoxycarbonyl)amino)ethyl-1-sulfonic acid and possesses the technical capability to execute this synthesis with precision and efficiency. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met whether you are in the early discovery phase or full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Boc-Taurine we deliver meets the highest standards of quality and consistency required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing you to make informed decisions with confidence. By choosing NINGBO INNO PHARMCHEM, you are securing a partner dedicated to innovation, reliability, and mutual success in the complex landscape of fine chemical manufacturing.
