Advanced Synthesis Of Diiodoisophthaloyl Chloride Compounds For High Purity Contrast Agents
The pharmaceutical landscape for diagnostic imaging agents is constantly evolving, with a heightened focus on the purity and safety of non-ionic iodine contrast media. Patent CN113717067A introduces a significant advancement in the preparation and application of specific diiodoisophthaloyl chloride compounds, namely 5-amino-4-chloro-2,6-diiodoisophthaloyl chloride and 5-amino-2-chloro-4,6-diiodoisophthaloyl chloride. These compounds, historically viewed merely as troublesome impurities, are now synthesized directionally to serve as critical reference standards. This innovation addresses a long-standing challenge in the industry where traditional synthesis routes inevitably generate these chlorinated by-products, which are structurally similar to the desired 5-amino-2,4,6-triiodoisophthaloyl chloride. By establishing a reliable method to produce these specific analogues, the patent empowers manufacturers to implement rigorous quality control measures, ensuring that the final contrast agents, such as iopromide and iopamidol, are free from potentially harmful contaminants that could compromise patient safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of the key starting material, 5-amino-2,4,6-triiodoisophthaloyl chloride, begins with isophthalic acid and proceeds through nitration, reduction, iodination, and acyl chlorination. A critical bottleneck in this conventional pathway is the iodination step, which predominantly employs iodine monochloride (ICl) as the iodinating reagent. While effective for introducing iodine atoms, ICl simultaneously acts as a chlorinating agent, leading to unavoidable side reactions. This results in the formation of chlorinated impurities where a chlorine atom substitutes an iodine atom at the 2 or 4 positions of the benzene ring. These impurities, specifically 5-amino-4-chloro-2,6-diiodoisophthaloyl chloride and its isomer, possess chemical properties remarkably similar to the target molecule, rendering their separation during purification extremely difficult and costly. Consequently, these impurities often persist into downstream processes.
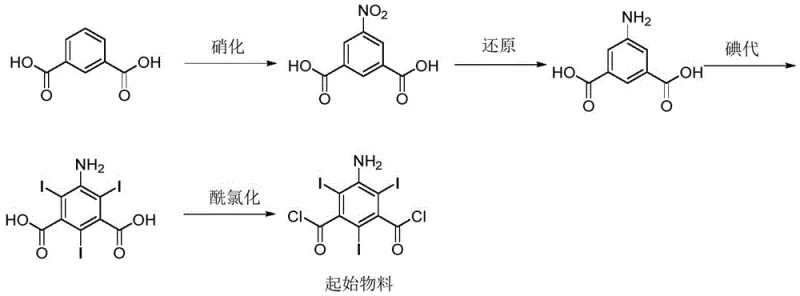
Furthermore, the persistence of these chlorinated species poses a severe risk to the quality of the final active pharmaceutical ingredient (API). As the synthesis continues into amidation reactions to form contrast agents like iopromide, these impurities react similarly to the main starting material. They incorporate into the molecular structure of the intermediates and the final drug product, creating a complex impurity profile that is hard to characterize without authentic standards. The inability to effectively detect and quantify these specific chlorinated derivatives means that batches of contrast agents could contain unknown levels of potentially toxic substances, leading to regulatory non-compliance and significant clinical safety risks for patients undergoing diagnostic procedures.
The Novel Approach
The novel approach detailed in the patent shifts the paradigm from merely trying to suppress these impurities to actively synthesizing them for quality control purposes. Instead of relying on the erratic by-product formation of the traditional route, this method provides a controlled, step-wise synthesis to generate high-purity samples of the chlorinated diiodo compounds. The process begins with the esterification of 5-aminoisophthalic acid, followed by a highly selective chlorination step using N-chlorosuccinimide (NCS). This selectivity is crucial as it allows for the precise placement of the chlorine atom before the iodination step occurs. By decoupling the chlorination and iodination events, the synthesis avoids the chaotic mixture produced by iodine monochloride, yielding specific isomers like Compound 1 and Compound 2 in high purity.
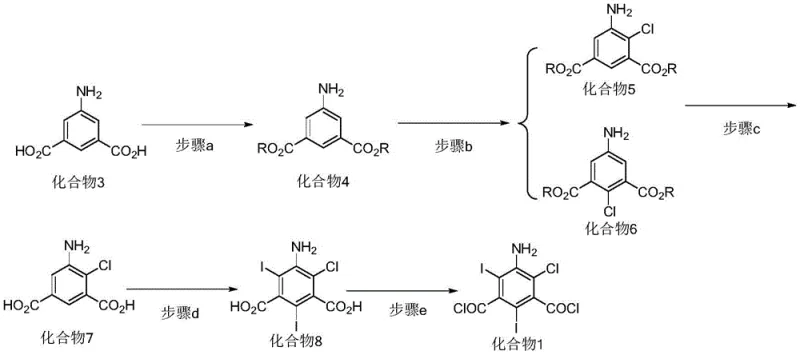
This directed synthesis strategy offers a robust solution for the pharmaceutical industry by providing the necessary tools for comprehensive impurity profiling. With access to authentic reference standards of the chlorinated impurities, quality assurance teams can develop precise HPLC methods to detect even trace amounts of these substances in the starting materials and final products. This capability is essential for meeting the stringent requirements of global regulatory bodies. Moreover, understanding the exact structure and behavior of these impurities allows chemists to optimize the main production process to minimize their formation, thereby enhancing the overall yield and purity of the valuable triiodo starting material and reducing the burden on downstream purification steps.
Mechanistic Insights into Selective Chlorination and Iodination
The core of this technological breakthrough lies in the mechanistic control of electrophilic aromatic substitution. In the novel synthesis, the use of N-chlorosuccinimide (NCS) in an organic solvent like acetonitrile allows for a mild and selective introduction of the chlorine atom onto the aromatic ring of the dimethyl 5-aminoisophthalate intermediate. Unlike the aggressive nature of iodine monochloride, NCS targets specific positions activated by the amino and ester groups, favoring the formation of the 4-chloro or 2-chloro isomers depending on the reaction conditions and steric factors. Following this, the hydrolysis step converts the esters back to carboxylic acids, preparing the molecule for the subsequent iodination. The iodination is then performed using iodine monochloride in an acidic medium, but since the chlorine position is already fixed, the iodine atoms are directed to the remaining available ortho positions relative to the amino group, resulting in the specific diiodo-chloro configuration.
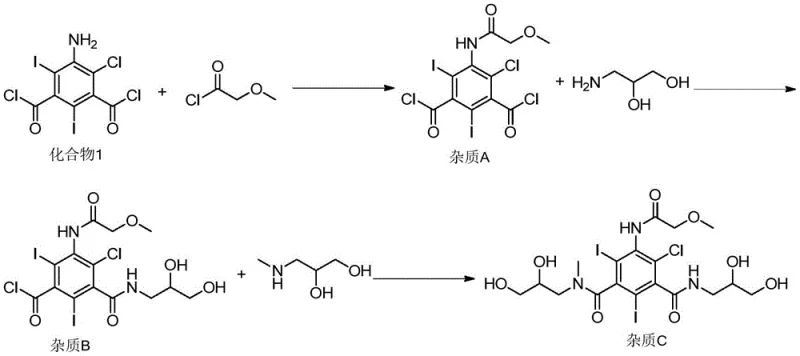
Understanding the propagation of these impurities is vital for risk assessment. The patent elucidates how these chlorinated starting materials flow through the synthesis of iopromide. For instance, Compound 1 reacts with methoxyacetyl chloride to form Impurity A, which subsequently undergoes amidation with aminopropanediol to form Impurity B, and finally reacts to form Impurity C. This cascade demonstrates that without removal at the source, the impurity burden multiplies through the synthetic tree. The ability to synthesize Compound 1 and Compound 2 independently allows researchers to spike these known impurities into process streams and validate the efficiency of purification protocols, ensuring that the final contrast agent is safe for human administration and free from genotoxic risks identified in Ames tests.
How to Synthesize 5-Amino-4-Chloro-2,6-Diiodoisophthaloyl Chloride Efficiently
The efficient synthesis of these critical reference standards relies on a optimized five-step sequence that balances yield with operational simplicity. The process initiates with the esterification of 5-aminoisophthalic acid using thionyl chloride in methanol, a reaction that proceeds smoothly under reflux conditions to protect the carboxylic acid groups. This is followed by the pivotal chlorination step where N-chlorosuccinimide is added in batches to control exotherms and ensure selectivity. The subsequent hydrolysis restores the acid functionality, setting the stage for the introduction of the heavy iodine atoms. The final acyl chlorination converts the diacid into the reactive diacyl chloride, ready for use in amidation reactions or as a stable reference material. Detailed standardized synthetic steps for this process are provided in the guide below.
- Perform esterification of 5-aminoisophthalic acid in an alcohol solvent with an acid catalyst to form the diester intermediate.
- Execute selective chlorination using N-chlorosuccinimide (NCS) in an organic solvent to introduce the chlorine atom at the desired position.
- Hydrolyze the ester under alkaline conditions, followed by iodination with iodine monochloride and final acyl chlorination with thionyl chloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the implementation of this patented technology translates into tangible strategic benefits beyond mere technical compliance. The primary advantage lies in the stabilization of the supply chain for high-value contrast agent intermediates. By adopting a quality control regime that utilizes these specific reference standards, manufacturers can significantly reduce the rate of batch failures caused by undefined impurities. In the past, batches of starting materials might have been rejected due to unknown peaks in chromatograms that could not be identified. With the ability to positively identify and quantify chlorinated impurities, companies can make informed decisions about blending or reprocessing, rather than scrapping expensive material, leading to substantial cost savings in raw material utilization.
- Cost Reduction in Manufacturing: The ability to precisely monitor impurity levels allows for the optimization of the main synthesis process. By identifying exactly when and how chlorinated by-products form, process engineers can tweak reaction parameters to suppress their generation at the source. This reduces the load on purification units such as crystallization or chromatography columns, lowering solvent consumption and energy usage. Furthermore, avoiding the release of off-specification batches prevents the massive financial losses associated with product recalls or regulatory penalties, ensuring a more predictable and efficient manufacturing cost structure.
- Enhanced Supply Chain Reliability: Sourcing high-purity intermediates is often a bottleneck in the production of diagnostic agents. This technology enhances reliability by enabling suppliers to certify their products against well-characterized standards. When a supplier can guarantee that chlorinated impurities are below a specific threshold backed by authentic reference data, it builds trust and reduces the need for extensive incoming quality testing by the buyer. This streamlines the procurement process, shortens lead times for material release, and ensures a continuous flow of compliant starting materials to the production line, mitigating the risk of stockouts.
- Scalability and Environmental Compliance: The synthetic route described utilizes common, scalable reagents like thionyl chloride and NCS, which are readily available in the global chemical market. This ensures that the production of these reference standards can be easily scaled from laboratory grams to commercial kilograms without requiring exotic catalysts or specialized equipment. Additionally, by improving the overall efficiency of the contrast agent synthesis and reducing waste from failed batches, the process contributes to a lower environmental footprint. This aligns with modern green chemistry initiatives and helps companies meet increasingly strict environmental regulations regarding chemical waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these diiodoisophthaloyl chloride compounds. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on the practical implementation of this quality control strategy.
Q: Why are chlorinated impurities critical in contrast agent synthesis?
A: Chlorinated impurities like Compound 1 and Compound 2 share similar structures with the target triiodo compound, making them difficult to remove. They can propagate through subsequent amidation steps, ending up in the final drug product and posing potential safety risks.
Q: How does this patent improve quality control?
A: By providing a directed synthesis method for these specific impurities, the patent enables manufacturers to create authentic reference standards. This allows for precise qualitative and quantitative analysis via HPLC, ensuring the starting material meets stringent purity specifications.
Q: What are the key reagents used in the novel synthesis route?
A: The process utilizes common and scalable reagents including thionyl chloride for esterification and acyl chlorination, N-chlorosuccinimide (NCS) for selective chlorination, and iodine monochloride for the iodination step, facilitating easy commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diiodoisophthaloyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the safety and efficacy of diagnostic imaging agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of global pharmaceutical partners. We are committed to delivering materials that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation. By leveraging the insights from patents like CN113717067A, we continuously refine our manufacturing processes to minimize impurities and maximize yield, providing our clients with a competitive edge in the marketplace.
We invite you to collaborate with us to optimize your supply chain for contrast agent intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical synthesis can support your journey towards safer and more effective diagnostic solutions.
