Advanced Synthesis of Epoxy Pregnenolone: A High-Efficiency Route for Steroid Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways for synthesizing critical steroid intermediates, and patent CN102219825B presents a groundbreaking methodology for producing 3β-hydroxy-16α,17α-epoxy-5-pregnene-20-one. This specific compound, often referred to as the oxygen bridge or epoxy pregnenolone, serves as a pivotal precursor in the manufacture of corticosteroids and progestogens, directly influencing the quality of downstream products like Woshi oxide. The disclosed innovation addresses long-standing inefficiencies in traditional synthesis by optimizing reaction kinetics and eliminating cumbersome purification steps. By leveraging a precise temperature-controlled epoxidation in a methanol medium, this technology achieves reaction completion in merely 3 to 4 hours, a drastic improvement over conventional methods that often require upwards of 18 hours. For R&D directors and procurement specialists alike, this represents a significant opportunity to enhance throughput while maintaining exceptional product integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3β-hydroxy-16α,17α-epoxy-5-pregnene-20-one has been plagued by prolonged reaction times and suboptimal yields that hinder large-scale economic viability. Traditional protocols typically necessitate reaction durations of approximately 18 hours, which not only ties up reactor capacity but also increases energy consumption and operational costs significantly. Furthermore, earlier attempts to improve purity, such as those utilizing sodium silicate complexing agents described in patent CN101717426A, introduced new complications; while they marginally improved content to 99.0%, the presence of silicates created severe downstream processing challenges, requiring additional filtration and washing steps that are ill-suited for continuous factory production. Other methods, such as those employing aqueous methanol solvents at low temperatures (5-15°C) as seen in European Patent PCT No. 2004021975, suffered from extended reaction times of up to 13 hours and increased solvent handling costs, rendering them less competitive for high-volume manufacturing environments where speed and simplicity are paramount.
The Novel Approach
The methodology outlined in CN102219825B fundamentally reengineers the epoxidation process by optimizing reagent ratios and thermal profiles to achieve near-quantitative conversion with minimal waste. Instead of relying on complexing agents or cryogenic conditions, this novel approach utilizes a specific molar ratio of dienolone acetate to 30% hydrogen peroxide and 10% sodium hydroxide (1:3:1.5) in a pure methanol solvent system. The process initiates by dissolving the diene at 40°C before carefully cooling to 26-27°C, followed by the controlled addition of oxidants while maintaining a tight temperature window of 28-30°C. Subsequently, the reaction mixture is gently heated to 34-35°C and held for only 3 to 4 hours, ensuring complete conversion without degrading the sensitive steroid backbone. This streamlined workflow eliminates the need for silicate removal, drastically simplifies the workup procedure to simple distillation and washing, and consistently delivers a white solid product with a yield between 98.5% and 99.1%, setting a new benchmark for efficiency in steroid intermediate manufacturing.
Mechanistic Insights into Alkaline Epoxidation of Dienolone Acetate
The core chemical transformation in this process is the nucleophilic epoxidation of the Δ16 double bond in the presence of alkaline hydrogen peroxide, a reaction that requires precise control to prevent over-oxidation or hydrolysis of the acetoxy group. The mechanism involves the generation of the hydroperoxide anion (HOO-) from hydrogen peroxide under basic conditions, which then attacks the electron-deficient double bond at the C16-C17 position of the steroid nucleus. The specific temperature ramping strategy—starting low to control the exotherm during reagent addition and then raising to 34-35°C—is critical for driving the reaction to completion while minimizing side reactions such as the formation of glycols or degradation of the 3β-acetoxy moiety. By avoiding the use of water-methanol mixtures found in older patents, the reaction environment remains sufficiently anhydrous to protect the ester functionality, thereby preserving the structural integrity required for subsequent synthetic steps in the hormone production chain.
Impurity control is inherently built into this mechanistic design through the exclusion of extraneous metal ions and complexing agents that often act as catalysts for decomposition. In traditional silicate-based methods, residual silicon species can trap the product or catalyze unwanted rearrangements during the drying phase, leading to unstable melting points and variable assay results. In contrast, the clean reaction profile of this methanol-based system ensures that the only major byproducts are water and sodium salts, which are easily removed during the aqueous washing stage. This results in a product with a sharp, stable melting point range of 189.5-191.2°C and a purity of 99.3%, indicating a highly selective transformation that minimizes the formation of regioisomers or oxidative degradation products that could complicate downstream crystallization processes.
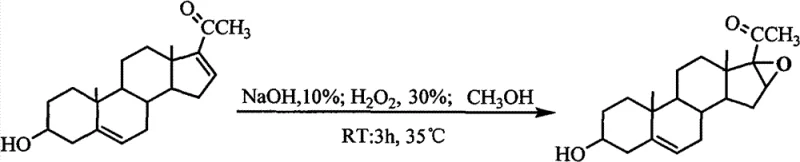
How to Synthesize 3β-hydroxy-16α,17α-epoxy-5-pregnene-20-one Efficiently
Implementing this synthesis route in a pilot or production plant requires strict adherence to the thermal profile and reagent addition rates specified in the patent to ensure safety and reproducibility. The process begins with the dissolution of the starting diene in methanol, followed by a controlled cooling phase to prepare the system for the exothermic addition of hydrogen peroxide and sodium hydroxide. Operators must monitor the temperature closely during the addition phase to prevent runaway reactions, keeping the bulk liquid between 28°C and 30°C before initiating the final heating stage. The detailed standardized operating procedures, including specific flow rates and agitation requirements for scaling this reaction from laboratory to commercial volumes, are outlined in the technical guide below.
- Dissolve dienolone acetate in methanol at 40°C, then cool the solution to 26-27°C.
- Slowly add 30% H2O2 and 10% NaOH sequentially while maintaining the temperature between 28-30°C.
- Raise the temperature to 34-35°C, maintain for 3-4 hours, then distill, wash, and dry to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates directly into enhanced operational agility and reduced total cost of ownership for steroid intermediate production. The elimination of sodium silicate and other complexing agents removes an entire unit operation from the manufacturing line, specifically the difficult filtration and extensive washing steps required to remove silica residues. This simplification not only reduces the consumption of water and utilities but also shortens the overall batch cycle time, allowing facilities to increase their annual output capacity without requiring additional capital investment in new reactors. Furthermore, the high yield and purity reduce the need for reprocessing or recrystallization, ensuring a more predictable and reliable supply of high-quality material for downstream API synthesis.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic reduction in reaction time and the simplification of the workup procedure. By cutting the reaction duration from nearly a day to just a few hours, manufacturers can significantly lower energy costs associated with heating, cooling, and agitation over extended periods. Additionally, the absence of expensive or difficult-to-handle complexing agents reduces raw material costs, while the simplified isolation process decreases labor hours and waste disposal fees associated with silica sludge. These factors combine to create a leaner manufacturing process that offers substantial cost savings per kilogram of finished product compared to legacy technologies.
- Enhanced Supply Chain Reliability: Shorter cycle times inherently improve supply chain responsiveness, enabling producers to react more quickly to fluctuations in market demand for steroid hormones. The robustness of the methanol-based system, which does not rely on sensitive low-temperature control or specialized reagents, ensures consistent production runs with minimal risk of batch failure due to process deviations. This reliability is crucial for maintaining continuous supply lines to API manufacturers, reducing the risk of stockouts and ensuring that downstream production schedules for critical medications like corticosteroids remain uninterrupted.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns well with modern green chemistry principles by minimizing waste generation and avoiding the use of persistent inorganic additives. The straightforward aqueous workup generates a waste stream that is easier to treat compared to silicate-containing effluents, facilitating compliance with increasingly stringent environmental regulations. Moreover, the process has been demonstrated to scale effectively, with examples showing successful execution at multi-kilogram scales, proving its viability for transition from pilot plants to hundred-ton annual production capacities without loss of yield or quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this epoxidation technology, derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the specific performance metrics and operational parameters validated in the patent examples.
Q: What are the key advantages of this synthesis method over traditional processes?
A: This method significantly reduces reaction time from 18 hours to just 3-4 hours and eliminates the need for complexing agents like sodium silicate, simplifying purification and increasing yield to over 98.5%.
Q: What is the purity and melting point of the resulting epoxy pregnenolone?
A: The product achieves a high purity of 99.3% with a stable melting point range of 189.5-191.2°C, meeting stringent quality standards for downstream steroid synthesis.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses common solvents like methanol, avoids difficult-to-remove additives, and features a short cycle time, making it highly scalable and cost-effective for factory production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3β-hydroxy-16α,17α-epoxy-5-pregnene-20-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain, and we possess the technical expertise to bring advanced synthesis routes like CN102219825B to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of rapid reaction times and high purity are fully realized in large-scale manufacturing. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch of epoxy pregnenolone meets the exacting melting point and assay requirements necessary for downstream steroid synthesis, providing our partners with absolute confidence in material consistency.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis method can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic impact of switching to this efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our capabilities can support your long-term supply security and product quality goals.
