Revolutionizing Venlafaxine Intermediate Production with Safe Amidine Catalysis Technology
Revolutionizing Venlafaxine Intermediate Production with Safe Amidine Catalysis Technology
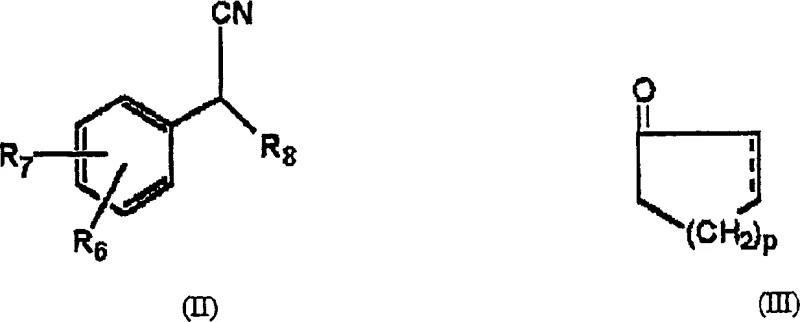
The pharmaceutical industry is constantly seeking robust, scalable, and safe methodologies for the production of critical active pharmaceutical ingredient (API) intermediates. Patent CN1267410C introduces a transformative approach to synthesizing cyclohexanol derivatives, specifically 1-[cyano(4-methoxyphenyl)methyl]cyclohexanol, which serves as a pivotal precursor for the antidepressant drug Venlafaxine. This innovation addresses the longstanding limitations of traditional organometallic chemistry by employing non-organometallic base catalysts, such as amidines and guanidines. For R&D directors and procurement specialists, this shift represents a move away from hazardous, cryogenic processes toward a more sustainable, cost-effective, and operationally simple manufacturing paradigm that ensures consistent supply chain continuity for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key cyclohexanol intermediates has relied heavily on the use of strong organometallic bases like n-butyllithium or lithium diisopropylamide (LDA). As detailed in prior art such as USP 4,535,186 and USP 5,043,466, these reagents necessitate extreme reaction conditions, typically requiring temperatures below -50°C to maintain stability and control reactivity. The logistical burden of maintaining such cryogenic environments is substantial, demanding specialized equipment and high energy consumption. Furthermore, organometallic bases are notoriously sensitive to moisture and air, posing significant safety risks including fire and explosion hazards during large-scale handling. Perhaps most critically for commercial viability, these conventional two-step processes often suffer from poor atom economy and inconsistent yields, frequently falling below 50%, which drastically inflates the cost of goods sold and complicates waste management protocols.
The Novel Approach
In stark contrast, the methodology disclosed in CN1267410C utilizes non-organometallic bases, specifically amidines and guanidines, to facilitate the coupling of phenylacetonitrile derivatives with cyclohexanones. This novel approach eliminates the need for stoichiometric amounts of dangerous metal reagents, replacing them with catalytic quantities of stable organic bases like DBU, DBN, or TBD. The reaction can proceed efficiently at much milder temperatures, ranging from -20°C to 80°C, with a preferred window of 10-30°C, effectively allowing for ambient temperature processing. By consolidating the anion generation and coupling steps into a single pot, the process simplifies the operational workflow, removes the complexity of endpoint determination for anion formation, and significantly enhances the overall yield profile, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations aiming for industrial scale-up.
Mechanistic Insights into Amidine-Catalyzed Nucleophilic Addition
The core mechanistic advantage of this technology lies in the unique basicity and nucleophilic properties of the amidine and guanidine catalysts. Unlike traditional strong bases that irreversibly deprotonate substrates and generate reactive metal salts, these organic superbases function through a reversible proton transfer mechanism. The catalyst, represented generally by Formula IV or Formula V in the patent data, abstracts the acidic alpha-proton from the phenylacetonitrile derivative to generate a stabilized carbanion intermediate. This anion then acts as a potent nucleophile, attacking the carbonyl carbon of the cyclohexanone derivative. The use of cyclic amidines like DBU or bicyclic guanidines like TBD provides a steric environment that favors deprotonation while minimizing side reactions such as self-condensation of the ketone. This precise control over the reaction pathway is crucial for R&D teams focused on impurity profiling, as it inherently limits the formation of complex organometallic by-products that are difficult to remove in downstream purification.

Furthermore, the impurity control mechanism is significantly enhanced by the absence of metal ions in the reaction matrix. In conventional organolithium routes, quenching steps often generate lithium salts and other inorganic residues that require extensive aqueous workups and chelation treatments to meet stringent purity specifications for API intermediates. The organic nature of the catalysts in this patent allows for a cleaner workup procedure, typically involving simple acidification and filtration. The catalyst itself, being an organic amine, can often be removed or neutralized easily during the acidic quench step, resulting in a crude product with a much simpler impurity profile. This mechanistic cleanliness translates directly to higher isolated yields and reduced solvent consumption during recrystallization, addressing key concerns for both quality assurance and environmental compliance in modern chemical manufacturing.
How to Synthesize 1-[cyano(4-methoxyphenyl)methyl]cyclohexanol Efficiently
The practical implementation of this synthesis route is designed for ease of adoption in existing multipurpose chemical plants. The process begins with the direct mixing of the nitrile and ketone starting materials in the presence of a catalytic amount of the selected base, such as 1,8-diazabicyclo[5,4,0]undec-7-ene (DBU). Detailed standard operating procedures for scaling this reaction from laboratory benchtop to multi-ton production are critical for ensuring batch-to-batch consistency. The following guide outlines the fundamental steps derived from the patent examples to achieve optimal conversion and purity, serving as a foundational reference for process engineers looking to implement this technology.
- Mix p-methoxyphenylacetonitrile and cyclohexanone with a catalytic amount of a non-organometallic base such as DBU or TBD.
- Stir the reaction mixture at a mild temperature ranging from 0°C to 30°C for a period of 10 to 48 hours depending on the specific catalyst loading.
- Quench the reaction with dilute hydrochloric acid to adjust pH to acidic, filter the precipitated solid, and wash with water and organic solvents to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this catalytic process offers profound strategic benefits beyond mere technical feasibility. The elimination of pyrophoric organometallic reagents fundamentally alters the risk profile of the manufacturing site, leading to substantial cost savings in terms of safety infrastructure, insurance premiums, and hazardous waste disposal. By removing the dependency on cryogenic cooling systems, the process becomes significantly more energy-efficient, reducing the utility burden associated with maintaining sub-zero reaction temperatures. These operational efficiencies contribute to a more competitive pricing structure for the final intermediate, allowing buyers to secure high-purity pharmaceutical intermediates at a more sustainable cost point without compromising on quality or regulatory compliance standards.
- Cost Reduction in Manufacturing: The shift from stoichiometric organometallic reagents to catalytic organic bases dramatically reduces raw material costs. Organometallic bases like n-butyllithium are expensive and must be used in excess, whereas the new method utilizes catalysts in minute quantities (as low as 0.005 equivalents). Additionally, the ability to run the reaction without organic solvents in certain embodiments further slashes solvent procurement and recovery costs. This leaner material usage profile directly impacts the bottom line, enabling significant cost reduction in API manufacturing by minimizing waste generation and maximizing the throughput of valuable starting materials into the final product.
- Enhanced Supply Chain Reliability: Reliance on hazardous, moisture-sensitive reagents often introduces fragility into the supply chain, where minor deviations in storage or transport can render batches unusable. The robust nature of amidine and guanidine catalysts, which are stable solids or liquids at room temperature, ensures a much more resilient supply chain. This stability reduces the risk of production delays caused by reagent degradation or safety incidents. Consequently, suppliers can offer more reliable lead times and consistent availability of critical intermediates, mitigating the risk of stockouts for downstream drug manufacturers who depend on uninterrupted raw material flow for their own production schedules.
- Scalability and Environmental Compliance: Scaling exothermic reactions involving organometallics presents significant engineering challenges due to heat management and safety concerns. The milder thermal profile of this new process facilitates easier scale-up from pilot plant to commercial production volumes. Furthermore, the reduction in hazardous waste and the potential for solvent-free operation align perfectly with increasingly stringent global environmental regulations. This green chemistry approach not only simplifies permitting and compliance but also enhances the corporate sustainability profile of the manufacturing partner, a factor that is becoming increasingly important for multinational pharmaceutical companies evaluating their vendor networks.
Frequently Asked Questions (FAQ)
Understanding the nuances of this new catalytic technology is essential for stakeholders evaluating its integration into their supply chains. The following questions address common inquiries regarding catalyst selection, reaction conditions, and product quality, providing clarity on how this method compares to established industry practices. These insights are derived directly from the experimental data and technical disclosures within the patent documentation to ensure accuracy and relevance for technical decision-makers.
Q: What are the advantages of using DBU over n-butyllithium in this synthesis?
A: Unlike n-butyllithium, which requires cryogenic conditions below -50°C and poses severe fire and explosion hazards, DBU operates safely at room temperature (10-30°C) and eliminates the need for stoichiometric organometallic reagents, significantly reducing safety risks and operational costs.
Q: Can this process be performed without organic solvents?
A: Yes, the patent explicitly states that the reaction can be carried out with or without organic solvents. Operating under solvent-free conditions simplifies the workup procedure, reduces waste generation, and enhances the environmental profile of the manufacturing process.
Q: What yields can be expected with the catalytic amidine method?
A: The process demonstrates superior efficiency compared to conventional methods, with experimental examples showing yields ranging from 70% up to 90.5% when optimized catalyst loadings and reaction times are utilized, whereas traditional organometallic routes often struggle to exceed 50% yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-[cyano(4-methoxyphenyl)methyl]cyclohexanol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic methodologies like the one described in CN1267410C is key to maintaining a competitive edge in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this catalytic process are fully realized in a GMP-compliant manufacturing environment. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity, assay, and impurity profiles for every batch produced.
We invite you to collaborate with us to leverage this safer, more efficient synthesis route for your Venlafaxine supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise in organocatalysis can drive value and reliability for your organization.
